Abstract
In muscle cells of the motor muscle of earthworm Lumbricus terrestris, the dystrophin and actin proteins and fast and slow isoforms of myosin heavy chains were determined using fluorescence microscopy. It can be thought that the expression of these proteins occurred at the earliest stages of the evolutionary formation of the intracellular contractile apparatus of the motor tissue in both invertebrates and vertebrates. This study will complement the picture of the evolutionary development of motor muscle tissue.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
INTRODUCTION
Dystrophin protein of 427 kDa is widely represented in both muscle and nervous tissues of vertebrates (Florczyk-Soluch et al., 2021; Sadoulet-Puccio, Kunkel, 1996). A similar protein with a mass of 140 kDa is present in similar tissues of sea leech Pontobdella muricata (Royuela et al., 1999, 2001). Dystrophin stabilizes the sarcolemma by attaching the extracellular matrix to the cytoskeleton through F-actin and a number of other dystrophin-associated proteins (Wilson et al., 2022). It has been shown that dystrophin-associated proteins are involved in the intracellular signaling processes involving neuronal NO synthase, phosphoinositol triphosphate 2, and calmodulin (Pilgram et al., 2010). In addition, dystrophin-associated proteins are required for clustering of neurotransmitter receptors and ion channels, as well as maintaining intracellular Ca2+ homeostasis (Pilgram et al., 2010).
The main motor proteins in somatic muscles that ensure their contractile function are actin and myosin molecules (Sweeney, Holzbaur, 2018). The latter consist of light and heavy chains (Lowey et al., 1993). Marine polychaete Urechis unicinctus exhibits myosin light and heavy chains (Kanzawa et al., 1991). Myosin light chains belong to a large family of Ca2+-binding proteins (Nieznanski et al., 2003). In invertebrates, an increase in the intracellular concentration of Ca2+ results in the binding of these ions to myosin light chains and triggering a cascade of biochemical reactions. Thus, actin interaction with myosin ATPase induces ATP hydrolysis, which triggers the contraction of myosin heavy chains (Fromherz and Szent-Györgyi, 1995). In this case, the main contractile proteins of thick muscle filaments are myosin heavy chains (Wells et al., 1996). It is known that fast and slow isoforms of myosin heavy chains are present in somatic muscles (Hooper and Thuma, 2005).
Actin plays an important role in physiological processes associated with intracellular Ca2+. The actin cytoskeleton modulates Ca2+ entry through membrane ligand- and voltage-activated Ca2+ channels. The release of Ca2+ from the endoplasmic reticulum with the participation of IP3 and ryanodine receptors is modulated by the polymerization and depolymerization of actin (Wang et al., 2002). F-actin of the muscular system is well characterized in representatives of the class of polychaetes (Filippova et al., 2006, 2010; Rüchel and Müller, 2007; Han et al., 2020). It should be noted that direct evidence of the presence of dystrophin, actin and myosin in muscle cells of the somatic muscle of oligochaetes is currently absent.
Taking into account the above, the purpose of this work was the immunofluorescent identification of the dystrophin and actin proteins and light and heavy isoforms of myosin in muscle cells of the evolutionarily primary motor muscle of the oligochaete Lumbricus terrestris.
MATERIALS AND METHODS
To prepare samples, earthworm Lumbricus terrestris was cut from the side along its entire length, the head and tail ends were cut off, the worm was opened and the internal organs and partitions between the segments were removed (Volkov et al., 2000). Next, fragments of the earthworm body wall 10–15 segments long were fixed with needles on the bottom of Petri dishes filled with Sylgard resin and perfused with Drewes-Pax solution (composition in mM: 77 NaCl, 4 KCl, 43 Na2SO4, 6 CaCl2, 2 Tris, 167 sucrose, pH 7.4) for about 30 min at room temperature (22 ± 1°C). Then, the samples were fixed for 30 min in 2% solution of p-formaldehyde and washed three times for 30 min in the phosphate-buffered saline (PBS). Samples were sequentially incubated in 0.5% Triton X-100 for 30 min; 15 min in a solution containing 5% normal goat serum, 1% bovine serum albumin, and 0.5% Triton X-100; and another 15 min in a solution of 1% bovine serum albumin and 0.5% Triton X-100 (solution A). All these solutions were prepared in PBS.
Next, the samples were incubated for 12 h at 4°C in solution A with poly- and monoclonal antibodies to dystrophin and fast and slow isoforms of myosin heavy chains (all diluted 1 : 100). Antibodies to the fast and slow isoforms of myosin heavy chains developed in rabbit and mouse, which allowed double immunolabeling of the proteins under study. The samples were washed in solution A three times for 30 min each and incubated for 1 h at room temperature with the corresponding secondary antibodies conjugated to Alexa 488 or 647 (1 : 200 dilution) in solution A. To confirm the specificity of antibody binding to the corresponding proteins, control experiments were performed. For negative controls, the samples were incubated with secondary antibodies without prior incubation with primary antibodies. For positive control, a preliminary 1-h incubation of primary antibodies was carried out with an immunogenic peptide (at a ratio of 1 : 10, respectively), to which primary antibodies were produced. Then, the samples were incubated in the resulting solution of primary antibodies with the immunogenic peptide (Li et al., 2016). The absence of antibody labeling in control experiments indicates the specificity of antibody binding to the corresponding peptides.
To stain F-actin, we used the toxin phalloidin conjugated with tetramethylrhodamine (TRITC) at a concentration of 10 μM. Labeling of cell nuclei was carried out in the presence of the dye DAPI (4',6-diamidino-2-phenylindole) at a concentration of 5 μM.
After washing in PBS, the samples were placed in the solution of phosphate-buffered saline with glycerol (1 : 1) and mounted on a glass slide for microscopic examination on a Leica TCS SP5 MP laser scanning confocal microscope (Leica Microsystems, United States) using a 63×/1.4 oil immersion objective. Multiphoton, Ar, and He–Ne lasers were used to excite fluorophore emission. Excitation wavelengths were 488 nm for Alexa fluorophores, 543 nm for TRITC, 633 nm for Alexa 647, and 349 nm DAPI. Analysis of the obtained confocal images was carried out using the ImageJ software (NIH, United States).
The following reagents were used: p-formaldehyde, Tris, phosphate-buffered saline (137 NaCl, 2.7 KCl, 4.3 Na2SO4, 1.4 KH2PO4, pH 7.2), Triton X-100, normal goat serum, bovine serum albumin, phalloidin-TRITC, DAPI, glycerol (Sigma-Aldrich, United States), primary rabbit polyclonal antibodies to dystrophin (ab85302; Abcam, United Kingdom), primary rabbit polyclonal antibodies to the fast isoform of a myosin heavy chain (ab91506; Abcam, United Kingdom), primary mouse monoclonal antibodies to the slow isoform of a myosin heavy chain (ma1064; Bosterbio, United States), immunogenic peptides corresponding to poly- and monoclonal antibodies (Abcam, Bosterbio), and secondary antibodies conjugated to Alexa 488 or Alexa 647 (Invitrogen, United States).
RESULTS AND DISCUSSION
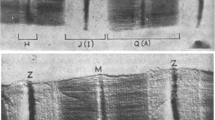
It has been established that antibodies produced against the vertebrate dystrophin protein are also capable of labeling dystrophin in the somatic muscles of annelids (Royuela et al., 2001). In our experiments, we stained earthworm muscle samples with antibodies against vertebrate dystrophin. It is seen (Fig. 1, downward-pointing arrows) that dystrophin is present in all areas of muscle tissue, with areas of pale and more intense (Fig. 1, upward-pointing arrows) staining. Thus, the protein dystrophin is widely represented in the muscle cells of the earthworm motor muscle. One might think that it is structurally and functionally close to its counterpart in the muscle tissue of higher chordates and invertebrates. The latter suggests that this protein is an obligatory and functionally important component of motor muscle cells starting from the earliest stages of the phylogenetic development of animal motor muscles.
The toxin phalloidin isolated from fungus Amanita phalloides specifically binds to fibrillar F-actin (Dancker et al., 1975). We stained muscle samples with phalloidin labeled with a fluorescent dye, as well as DAPI dye (with affinity for nuclear DNA). Application of fluorescently labeled phalloidin revealed continuous staining of muscle filaments along their entire length (Fig. 2a). Thus, the actin cytoskeleton is present in all parts of muscle cells. DAPI staining revealed typical interphase cell nuclei with hetero- and euchromatin (Fig. 2b). Our images show that each muscle cell has one or two cell nuclei (Figs. 2c, 2d). This fact confirms that the somatic musculature of the earthworm consists of muscle cells (Fig. 2), in contrast to the musculature of vertebrates containing muscle fibers, which is consistent with literature data (David, 1990; Cadot et al., 2015).
Earthworm samples labeled with antibodies to fast and slow isoforms of myosin heavy chains exhibit common oblique striation staining of muscle cells was observed (Fig. 3). Both myosin isoforms were detected (Figs. 3a, 3b). Moreover, this staining largely coincided, but in some areas the staining for the slow isoform was localized between the areas stained for the fast isoform (Fig. 3c). The high specificity of antibody binding when tagging the fast isoform of myosin heavy chains allowed us to take a more detailed image. It can be seen that the myofibril blocks are composed of smaller filaments (Fig. 4).
Detection of fast and slow isoforms of myosin heavy chains in earthworm muscle cells using double fluorescent staining with antibodies. (a) Staining with antibodies for the fast isoform of myosin heavy chains (green), (b) staining with antibodies for the slow isoform of myosin heavy chains (red), (c) composite of a and b images. Scale bar is 20 µm.
Thus, the studies have shown the presence of dystrophin and actin proteins and fast and slow isoforms of myosin heavy chains in muscle cells of the motor muscle of the earthworm. According to the literature data, the expression of dystrophin, actin, and myosin is found in a wide range of representatives of the animal world, including chordates, arthropods, and nematodes (Meedel, 1983; Miller et al., 1983; Roberts and Bobrow, 1998; Giugia et al., 1999; Lovato et al., 2001; Mercer et al., 2011; Ono and Pruyne, 2012). One might think that the expression of these proteins occurred at the earliest stages of the evolutionary formation of the intracellular contractile apparatus of the motor tissue in both invertebrates and vertebrates. The research that has been conducted will complement the picture of the evolutionary process of motor muscle tissue.
ABBREVIATIONS AND NOTATION
TRITC | tetramethylrhodamine |
PBS | phosphate buffer saline |
REFERENCES
Cadot, B., Gache, V., and Gomes, E.R., Moving and positioning the nucleus in skeletal muscle—one step at a time, Nucleus, 2015, vol. 6, p. 373. https://doi.org/10.1080/19491034.2015.1090073
Dancker, P., Löw, I., Hasselbach, W., and Wieland, T., Interaction of actin with phalloidin: polymerization and stabilization of F-actin, Biochim. Biophys. Acta, 1975, vol. 400, p. 407. https://doi.org/10.1016/0005-2795(75)90196-8
David, O.F., Morfofiziologicheskie osnovy lokomotsii annelid (Morphophysiological Basics of Locomotion of Annelids), Akad. Nauk SSSR, Inst. Evol. Fiziol. Biokhim. im. I.M. Sechenova, Leningrad: Nauka, 1990.
Filippova, A., Purschke, G., Tzetlin, A.B., and Müller, M.C.M., Three-dimensional reconstruction of the F-actin musculature of Dorvillea kastjani (Dorvilleidae, Polychaeta) by means of phalloidin-labelling and cLSM, Sci. Mar., 2006, vol. 70, no. S3, p. 293. https://doi.org/10.3989/scimar.2006.70s3293
Filippova, A., Pürschke, G., Tzetlin, A.B., and Müller, M.C.M., Musculature in polychaetes: comparison of Myrianida prolifera (Syllidae) and Sphaerodoropsis sp. (Sphaerodoridae), Invertebr. Biol., 2010, vol. 129, p. 184. https://doi.org/10.1111/j.1744-7410.2010.00191.x
Florczyk-Soluch, U., Polak, K., and Dulak, J., The multifaceted view of heart problem in Duchenne muscular dystrophy, Cell. Mol. Life. Sci., 2021, vol. 78, p. 5447. https://doi.org/10.1007/s00018-021-03862-2
Fromherz, S. and Szent-Györgyi, A.G., Role of essential light chain EF hand domains in calcium binding and regulation of scallop myosin, Proc. Natl. Acad. Sci. U. S. A., 1995, vol. 92, p. 7652. https://doi.org/10.1073%2Fpnas.92.17.7652
Giugia, J., Gieseler, K., Arpagaus, M., and Ségalat, L., Mutations in the dystrophin-like dys-1 gene of Caenorhabditis elegans result in reduced acetylcholinesterase activity, FEBS Lett., 1999, vol. 463, p. 270. https://doi.org/10.1016/s0014-5793(99)01651-8
Han, Y.H., Ryu, K.B., Medina Jiménez, B.I., Kim, J., Lee, H.Y., and Cho, S.J., Muscular development in Urechis unicinctus (Echiura, Annelida), Int. J. Mol. Sci., 2020, vol. 21, p. 1. https://doi.org/10.3390/ijms21072306
Hooper, S.L. and Thuma, J.B., Invertebrate muscles: muscle specific genes and proteins, Physiol. Rev., 2005, vol. 85, p. 1001. https://doi.org/10.1152/physrev.00019.2004
Kanzawa, N., Kawamura, Y., Matsuno, A., and Maruyama, K., Characterization of myosin isolated from bodywall smooth muscle of the annelid, Urechis unicinctus, Proc. Jpn. Acad. Ser. B, 1991, vol. 67, p. 176. https://doi.org/10.2183/pjab.67.176
Li, Y., Hu, H., Butterworth, M.B., Tian, J.B., Zhu, M.X., and O’Neil, R.G., Expression of a Diverse array of Ca2+-activated K+ channels (SK1/3, IK1, BK) that functionally couple to the mechanosensitive TRPV4 channel in the collecting duct system of kidney, PLoS One, 2016, vol. 11, p. e0155006. https://doi.org/10.1371/journal.pone.0155006
Lovato, T.L., Meadows, S.M., Baker, P.W., Sparrow, J.C., and Cripps, R.M., Characterization of muscle actin genes in Drosophila virilis reveals significant molecular complexity in skeletal muscle types, Insect. Mol. Biol., 2001, vol. 10, p. 333. https://doi.org/10.1046/j.0962-1075.2001.00270.x
Lowey, S., Waller, G.S., and Trybus, K.M., Function of skeletal muscle myosin heavy and light chain isoforms by an in vitro motility assay, J. Biol. Chem., 1993, vol. 268, p. 20414. https://doi.org/10.1016/S0021-9258(20)80744-3
Meedel, T.H., Myosin expression in the developing ascidian embryo, J. Exp. Zool., 1983, vol. 227, p. 203. https://doi.org/10.1002/jez.1402270205
Mercer, R.C., Mudalige, W.A., Ige, T.O., and Heeley, D.H., Vertebrate slow skeletal muscle actin—conservation, distribution and conformational flexibility, Biochim. Biophys. Acta, 2011, vol. 1814, p. 1253. https://doi.org/10.1016/j.bbapap.2011.06.009
Miller, D.M., 3rd, Ortiz, I., Berliner, G.C., and Epstein, H.F., Differential localization of two myosins within nematode thick filaments, Cell, 1983, vol. 34, p. 477. https://doi.org/10.1016/0092-8674(83)90381-1
Nieznanski, K., Nieznanska, H., Skowronek, K., Kasprzak, A.A., and Stepkowski, D., Ca2+ binding to myosin regulatory light chain affects the conformation of the N-terminus of essential light chain and its binding to actin, Arch. Biochem. Biophys., 2003, vol. 417, p. 153. https://doi.org/10.1016/s0003-9861(03)00382-5
Ono, S. and Pruyne, D., Biochemical and cell biological analysis of actin in the nematode Caenorhabditis elegans, Methods, 2012, vol. 56, p. 11. https://doi.org/10.1016/j.ymeth.2011.09.008
Pilgram, G.S., Potikanond, S., Baines, R.A., Fradkin, L.G., and Noordermeer, J.N., The roles of the dystrophin-associated glycoprotein complex at the synapse, Mol. Neurobiol., 2010, vol. 41, p. 1. https://doi.org/10.1007/s12035-009-8089-5
Roberts, R.G. and Bobrow, M., Dystrophins in vertebrates and invertebrates, Hum. Mol. Genet., 1998, vol. 7, p. 589. https://doi.org/10.1093/hmg/7.4.589
Royuela, M., Paniagua, R., Rivier, F., Hugon, G., Robert, A., and Mornet, D., Presence of invertebrate dystrophin-like products in obliquely striated muscle of the leech, Pontobdella muricata (Annelida, Hirudinea), Histochem. J., 1999, vol. 31, p. 603. https://doi.org/10.1023/A:1003855108802
Royuela, M., Hugon, G., Rivier, F., Paniagua, R., and Mornet, D., Dystrophin-associated proteins in obliquely striated muscle of the leech Pontobdella muricata (Annelida, Hirudinea), Histochem. J., 2001, vol. 33, p. 135. https://doi.org/10.1023/A:1017979623095
Rüchel, J. and Müller, M.C.M., F-actin framework in Spirorbis cf. spirorbis (Annelida: Serpulidae): phalloidin staining investigated and reconstructed by cLSM, Invertebr. Biol., 2007, vol. 126, p. 173. https://doi.org/10.1111/j.1744-7410.2007.00087.x
Sadoulet-Puccio, H.M. and Kunkel, L.M., Dystrophin and its isoforms, Brain Pathol., 1996, vol. 6, p. 25. https://doi.org/10.1111/j.1750-3639.1996.tb00780.x
Sweeney, H.L. and Holzbaur, E.L.F., Motor proteins, Cold Spring Harb. Perspect. Biol., 2018, vol. 10, p. a021931. https://doi.org/10.1101/cshperspect.a021931
Volkov, E.M., Nurullin, L.F., Svandová, I., Nikolsky, E.E., and Vyskocil, F., Participation of electrogenic Na+, K+-ATPase in the membrane potential of earthworm body wall muscles, Physiol. Res., 2000, vol. 49, p. 481. http:// www.biomed.cas.cz/physiolres/pdf/49/49_481.pdf
Wang, Y., Mattson, M.P., and Furukawa, K., Endoplasmic reticulum calcium release is modulated by actin polymerization, J. Neurochem., 2002, vol. 82, p. 945. https://doi.org/10.1046/j.1471-4159.2002.01059.x
Wells, L., Edwards, K.A., and Bernstein, S.I., Myosin heavy chain isoforms regulate muscle function but not myofibril assembly, EMBO J., 1996, vol. 15, p. 4454. https://doi.org/10.1002/j.1460-2075.1996.tb00822.x
Wilson, D.G.S., Tinker, A., and Iskratsch, T., The role of the dystrophin glycoprotein complex in muscle cell mechanotransduction, Commun. Biol., 2022, vol. 5, p. 1022. https://doi.org/10.1038/s42003-022-03980-y
Funding
The work was supported by the Russian Science Foundation, project no. 23-24-00239 (https://rscf.ru/project/23-24-00239/).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Animal experiments were conducted in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals (http://oacu.od.nih.gov/ regs/index.htm). Protocols using animals were approved by the bioethics commission of the Kazan Scientific Center of the Russian Academy of Sciences, Kazan, Russia, protocol no. 23/5 of December 5, 2023.
CONFLICT OF INTEREST
The authors of this work declare that they have no conflicts of interest.
Additional information
Translated by I. Fridlyanskaya
Publisher’s Note.
Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nurullin, L.F., Volkov, E.M. Immunofluorescent Identification of Dystrophin, Actin, and Light and Heavy Myosin Chains in Somatic Cells of Earthworm Lumbricus terrestris. Cell Tiss. Biol. 18, 341–346 (2024). https://doi.org/10.1134/S1990519X24700287
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990519X24700287