Abstract
To understand what happens in the serum after methamphetamine (MA) administration, nuclear magnetic resonance-based metabolomics was performed to investigate the characteristic metabolite profile in the serum of rats with MA-induced conditioned place preference. The results showed that tyrosine concentration was significantly higher in the MA-induced group (1.68 × 10–5 ppm) than in the control group (0.99 × 10–5 ppm) (p = 0.004), uncharacterized lipid concentration was significantly higher in the control group (1.28 × 10–3 ppm) than in the MA-induced group (0.78 × 10–3 ppm) (p = 0.008), acetone concentration was significantly higher in the control (1.65 × 10–4 ppm) than in the and MA-induced group (1.10 × 10–4 ppm) (p = 0.005), and leucine concentration was significantly higher in the control (5.20 × 10–4 ppm) than in the MA-induced group (4.37 × 10–4) (p-value = 0.03). This decrease in acetone concentration in the MA-induced group may help elucidate why ketogenic diet has shown promise for many neurological disorders. Tyrosine is the precursor of dopamine and γ-aminobutyric acid, and leucine is the major nitrogen donor for glutamate synthesis. These results suggest that a disturbance in the substrate supply in the circulatory system partly explains MA toxicity in the central nervous system.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Methamphetamine (MA) is a widely abused psychostimulant classified as an amphetamine-type stimulant (ATS). MA abuse is a serious public crisis. In the 2018 World Drug Report, amphetamine (including both amphetamine and MA) was deemed to be used by approximately 34 million people globally. “Despite the trends in the respective shares of each drug in seizure cases shows a decline in the share of global cannabis seizure cases over the past decade, by contrast, the share of seizure cases of ATS (mostly methamphetamine) rose over the same period.” Many countries consider MA use to be one of the most worrying threats of drug use. Chronic heavy MA use likely causes dopaminergic neurotoxicity and results in cognitive deficits, such as disturbances in working memory, attention problems, and impaired impulse control and decision making [1]. Both acute and chronic use of MA could generate an imbalance in the release and reuptake of dopamine, norepinephrine, and epinephrine. MA could have an effect on not only the central but also the peripheral nervous system, resulting in both physical and psychological alterations. Continuous consumption of large doses of MA is related to a heightened risk of developing cardiovascular problems [2]. Different hypotheses regarding the mechanism responsible for the neurotoxicity caused by MA have been proposed, and the most predominant one is focused on dopamine release and the function of dopaminergic nerve terminals [3].
Different pharmacological approaches have been established in the treatment of MA addiction, but most clinical studies have not found meaningful benefits [4]. Thus, to determine the molecular mechanism leading to MA addiction, a method that could provide a complete picture reflecting what happens in the body would be useful. Metabolomics analysis seemed to be a prospective approach. In this study, nuclear magnetic resonance (NMR)-based metabolomics was utilized to analyze the metabolomic changes in the serum of rats with MA-induced conditioned place preference (CPP).
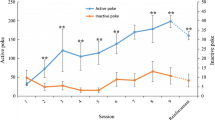
First, the animal model was set up. MA-induced CPP was conducted as previously described, with minor modification [5]. There were 8 rats in the control group and 22 in the MA-induced CPP group. After treatment with MA for 7 sessions, a significant preference was established. The MA-treated group spent 282 s of their time in the MA-paired, naturally unpreferred white compartment, while the control group spent 198 s in the white compartment. The p‑value of time spent in MA-paired chamber between the two groups was 0.038 (Fig. 1).
Development of CPP induced by repeated administration of MA (a) and PLS-DA model generated by SIMCA-P software for comparison of the MA-induced CPP and control groups (b). (a) Data are presented as mean ± SEM of time spent in the MA-paired chamber (chamber a) during the post-conditioning tests. Asterisk indicates statistically significant difference between the MA-induced CPP and control groups, p < 0.05. (b) R2X = 39.4%, R2Y = 93.7%, Q2 = 0.676. Ellipse was obtained by Hotteling’s T2 (0.95); n = 8 for the control group and n = 22 for the MA-induced CPP group.
The NMR spectra of serum samples were recorded as previously mentioned [6]. The metabolites observed in the 1H NMR spectra of the serum were mainly amino acids (isoleucine, leucine, valine, alanine, lysine, glutamate, glutamine, histidine, tyrosine, and phenylalanine), metabolites of glycolysis and TCA (TriCarboxylic Acid cycle) cycle (α-glucose, succinate, fumarate, and formate), ketone bodies (acetone, acetoacetate, and 3-hydroxybutyrate), and lipids (phosphocholine and glycerol phosphocholine).
To visualize the clustering of the tested samples, a principal component analysis (PCA) model was constructed. The two first principal components showed that the two groups could not be separated. Then, we attempted to perform partial least squares-discriminate analysis (PLS-DA), and the score plot based on PLS-DA showed obvious categorization between the two groups (Fig. 1). Q2 (cum) obtained from the cross-validation of PLS-DA was 0.676, which was >0.5, and for metabolomics data, Q2 (cum) >0.5 indicates good predictability.
To further identify altered metabolites in the MA‑induced CPP group, orthogonal projections to latent structures discriminant analysis was conducted. If metabolites that differ between the two groups met the cutoff value (|r| > 0.666 (df = 7)), they were recorded as being significantly different at a p-value <0.05. Of 27 metabolites, 2 were identified (Fig. 2). To find more differences between the two groups, we attempted to use another criterion, that is VIP >1 and p-value <0.05 (VIP: Variable Important in Projection). Four metabolites were recorded as being significantly different between the MA-induced CPP and control groups. Metabolites with significant difference are presented in Table 1.
OPLS-DA score plot (a) and corresponding coefficient plot (b) derived from the 1H NMR spectra of extractions of the serum obtained from the control (n = 8) and MA-induced CPP (n = 22) groups. A correlation coefficient of |r| > 0.666 (df = 7) was used as the cutoff value that showed a statistically significant result at a p-value < 0.05. Loading maps show the significance of metabolite variations between the two groups. Peaks in the positive and negative direction indicate the metabolites that are more abundant in the MA-induced CPP and control groups, respectively.
Furthermore, to assess the major metabolic functions of the characteristic metabolites, the four altered metabolites were invoked as the input into the Kyoto Encyclopedia of Genes and Genomes database (http://www.kegg.jp/), excluding lipids as they were loosely defined. As shown in Fig. 3, impaired metabolic pathways were mostly involved in ketone bodies and amino acid metabolism.
Metabolic reaction networks of characteristic metabolites found differentially expressed in the serum between the control and MA-induced CPP groups. Metabolites in red indicate that the metabolite has a higher concentration in the MA-induced CPP group, and metabolites in blue indicate that the metabolite has a lower concentration in MA-induced CPP group.
Acetone is a ketone body, which is generated from fatty acids in the liver during periods of prolonged intense exercise, starvation, low food intake, or untreated type 1 diabetes mellitus as a result of intense gluconeogenesis. Three ketone bodies that exist in the body, i.e., acetoacetate, 3-β-hydroxybutyrate (3-HB), and acetone. Acetone is converted into lactic acid in the liver, which can be oxidized into pyruvic acid, and only then turn into acetyl-CoA, which then enters the citric acid cycle and is oxidized in the mitochondria to produce ATP. Ketone bodies are a more efficient energy source, generating more ATP per unit oxygen than glucose. Disturbance of ketone bodies was reported in other metabolomic studies of drug addiction. A decrease in β-hydrobutyrate concentration was found in morphine-addicted rats [7] and MA-treated rats [8]. In this study, acetoacetate and 3-HB were detected, but there was no significant difference. Recently, ketogenic diets (KDs) are shown to be effective in normalizing behaviors and neural processes induced by cocaine [9]. Administration of acetoacetate reduces quantal size of hippocampal synapses and extracellular glutamate levels and seizures evoked by 4-aminopyridine infusions [10]. KD-fed rats showed decreased cocaine-induced stereotyped response compared with control diet-fed rats [6]. The reduction in ketone bodies may partially explain why KDs could normalize or recover behaviors or neural processes.
Leucine acts as the most important nitrogen donor for glutamate synthesis in the brain and is one of the three branched-chain amino acids (BCAAs) existing in the mammal body; the other two are isoleucine and valine. A decrease in the levels of these three BCAAs was found in rat plasma collected after 16 days of MA self-administration [11]. In our results, isoleucine and valine were also detected, but there was no difference. Leucine could readily pass through the blood–brain barrier (BBB), and in astrocytes, it is transaminated by branched-chain aminotransferases (BCATs) to produce glutamate and β-keto-isocaproate. Approximately 25% of nitrogen transfer was derived from leucine in the rat brain. Deprivation of nitrogen source for a long period can deprive the brain of necessary amino acids for the synthesis of glutamate, which served as the precursor of γ-aminobutyric acid [5]. In the urine of rats with MA-induced CPP, the relative concentration of β-keto-isocaproate increased (data not shown). As the BCATs catalyzed the conversion of leucine to β-keto-isocaproate, the increase in keto acid concentration and decrease in leucine concentration may the activity of BCATs was downregulated, but further study should be conducted to confirm this speculation. Mutation of BCAT could result in neurological dysfunction [12]. Unrestricted BCAA oxidation could result in an autism-spectrum disorder with epilepsy, and this disorder can be addressed with BCAA supplementation [13]. The decrease in leucine concentration in this case may help explain why dietary supplementation could serve as effective treatment for neurological disorders.
Another role of leucine relevant to brain function is the activation of mammalian target of rapamycin (mTOR). mTOR is a cellular serine/threonine protein kinase that plays an essential role in regulating synaptic protein synthesis [14]. mTOR participates in multiple brain functions, such as neuron differentiation and dendrite development. The role of mTOR in drug addiction was further confirmed by studies reporting that infusion of mTOR-shRNA into the shell region of the nucleus accumbens inhibited MA sensitization [14]. Comparable effects were obtained by administration of rapamycin in cocaine-induced rats [15, 16]. An in-depth study is needed to substantiate the role of mTOR in MA addiction.
L-Tyrosine is converted to l-DOPA (l-3,4-dihydroxyphenylalanine) by tyrosine hydroxylase (TH) after passing through the BBB, and TH is the rate-limiting enzyme in dopamine synthesis. Subsequently, DOPA decarboxylase converts l-DOPA into dopamine. MA elicits dopamine neurotoxicity, and MA treatment could increase extracellular levels of dopamine in the striatum [17, 18]. Now, based on the critical role of dopamine neurotransmission in the addictive properties of MA, dopamine D2 receptor antagonists were regarded as the most promising target for ATS abuse [4]. The upregulation of tyrosine in the serum indicated that a disturbance in the substrate supply in the circulatory system could perhaps partly explain MA toxicity in the central nervous system.
A change in lipid concentration was frequently observed in drug addiction models, which indicates that lipids could be considered markers for drug addiction [19]. Another study showed that repeated MA use significantly modified the lipidome in the brain [20]. Such results demonstrate that lipid profiling in the serum may be a potential indication of cell membrane integrity associated with drug addiction. Recent studies showed that serum glycerophospholipids could predict cognitive impairment in 2–3 years with 90% accuracy in Alzheimer’s disease, which bolstered this viewpoint to a greater extent [21]. However, as lipids were loosely defined, targeted lipid metabolomics (lipidomics) is needed to identify the different types of lipids, and several meaningful efforts were already made [21, 22].
Therefore, in this study, we attempted to discover dysregulations in the serum when rats were treated with MA by NMR-based metabolomics and CPP models. The results showed that the concentration of tyrosine (the precursor of dopamine) increased and that of acetone (one of ketone bodies) and leucine (one of BCAAs) decreased in rats were treated with MA. Our data may help in elucidating the molecular mechanism of MA addiction.
REFERENCES
Panenka, W.J., Procyshyn R.M., Lecomte T., MacEwan G.W., Flynn S.W., Honer W.G., and Barr A.M., Drug Alcohol Depend., 2013, vol. 129, no. 3, pp. 167–79.
Darke, S., Duflou J., and Kaye S., Drug Alcohol Depend., 2017. Vol.179, pp. 174–179.
Yu, S., Zhu L., Shen Q., Bai X., and Di X., Behav. Neurol., 2015, vol. 2015, p. 103969.
Cumming, C., Troeung L., Young J.T., Kelty E., and Preen D.B., Drug Alcohol Depend., 2016, vol. 168, pp. 263–273.
Sperringer, J.E., Addington A., and Hutson S.M., Neurochem. Res., 2017, vol. 42, no. 6, pp. 1697–1709.
Martinez, L.A., Lees M.E., Ruskin D.N., and Masino S.A., Neuropharmacology, 2019, vol. 149, pp. 27–34.
Zaitsu, K., Miyawaki, I., Bando, K., Horie, H., Shima, N., Katagi, M., Tatsuno, M., Bamba, T., Sato, T., Ishii, A., Tsuchihashi, H., Suzuki, K., and Fukusaki, E., Anal. Bioanal. Chem., 2014, vol. 406, no. 5, pp. 1339–1354.
Shima, N., Miyawaki, I., Bando, K., Horie, H., Zaitsu, K., Katagi, M., Bamba, T., Tsuchihashi, H., and Fukusaki, E., Toxicology, 2011, vol. 287, nos. 1–3, pp. 29–37.
Simeone, T.A., Simeone, K.A., Stafstrom, C.E., and Rho, J.M., Neuropharmacology, 2018, vol. 133, pp. 233–241.
Juge, N., Gray, J.A., Omote, H., Miyaji, T., Inoue, T., Hara, C., Uneyama, H., Edwards, R.H., Nicoll, R.A., and Moriyama, Y., Neuron, 2010, vol. 68, no. 1, pp. 99–112.
Kim, S., Jang W.J., Yu H., Ryu I.S., Jeong C.H., and Lee S., J. Proteome. Res., 2019, vol. 18, no. 11, pp. 3913–3925.
Garcia-Cazorla, A., Oyarzabal, A., Fort, J., Robles, C., Castejon, E., Ruiz-Sala, P., Bodoy, S., Merinero, B., Lopez-Sala, A., Dopazo, J., Nunes, V., Ugarte, M., Artuch, R., Palacin, M., Rodriguez-Pombo, P., Alcaide, P., Navarrete, R., Sanz, P., Font-Llitjos, M., Vilaseca, M.A., Ormaizabal, A., Pristoupilova, A., and Agullo, S.B., Hum. Mutat., 2014, vol. 35, no. 4, pp. 470–477.
Novarino, G., El-Fishawy, P., Kayserili, H., Meguid, N.A., Scott, E.M., Schroth, J., Silhavy, J.L., Kara, M., Khalil, R.O., Ben-Omran, T., Ercan-Sencicek, A.G., Hashish, A.F., Sanders, S.J., Gupta, A.R., Hashem, H.S., Matern, D., Gabriel, S., Sweetman, L., Rahimi, Y., Harris, R.A., State, M.W., and Gleeson, J.G., Science, 2012, vol. 338, no. 6105, pp. 394–397.
Huang, S.H., Wu, W.R., Lee, L.M., Huang, P.R., and Chen, J.C., Prog. Neuropsychopharmacol Biol. Psychiatry, 2018, vol. 86, pp. 331–339.
Wang, X., Luo, Y.X., He, Y.Y., Li, F.Q., Shi, H.S., Xue, L.F., Xue, Y.X., and Lu, L., J. Neurosci., 2010, vol. 30, no. 38, pp. 12632-1241.
Cui, Y., Zhang, X.Q., Cui, Y., Xin, W.J., Jing, J., and Liu, X.G., Neuroscience, 2010, vol. 171, no. 1, pp. 134-143.
McFadden, L.M., Vieira-Brock, P.L., Hanson, G.R., and Fleckenstein, A.E., Int. J. Neuropsychopharmacol, 2014, vol. 17, no. 8, pp. 1315-1320.
McFadden, L.M., Hadlock, G.C., Allen, S.C., Vieira-Brock, P.L., Stout, K.A., Ellis, J.D., Hoonakker, A.J., Andrenyak, D.M., Nielsen, S.M., Wilkins, D.G., Hanson, G.R., and Fleckenstein, A.E., J. Pharmacol. Exp. Ther., 2012, vol. 340, no. 2, pp. 295–303.
Zheng, T., Liu, L., Shi, J., Yu, X., Xiao, W., Sun, R., Zhou, Y., Aa, J., and Wang, G., Mol. Biosyst., 2014, vol. 10, no. 7, pp. 1968–1977.
Jiang, L., Gu, H., Lin, Y., Xu, W., Zhu, R., Kong, J., Luo, L., Long, H., Liu, B., Chen, B., Zhao, Y., and Cen, X., Toxicol. Lett., 2017, vol. 279, pp. 67–76.
Mapstone, M., Cheema, A.K., Fiandaca, M.S., Zhong, X., Mhyre, T.R., MacArthur, L.H., Hall, W.J., Fisher, S.G., Peterson, D.R., Haley, J.M., Nazar, M.D., Rich, S.A., Berlau, D.J., Peltz, C.B., Tan, M.T., Kawas, C.H., and Federoff, H.J., Nat. Med., 2014, vol. 20, no. 4, pp. 415–418.
Shi, X., Yao, D., Gosnell, B.A., and Chen, C., J. Lipid. Res., 2012, vol. 53, no. 11, pp. 2318–2330.
ACKNOWLEDGMENTS
We thank Xing Yan at Wuhan Institute of Biotechnology, for his help with the NMR analysis. We are grateful to BioNovogene Company for its help with the data analysis.
Funding
This work was supported by the project of the Education Department of Hubei Province (B2016300).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest. The authors declare that they have no conflict of interest.
Ethical approval. All procedures involving animals and their care were conducted according to governmental guidelines on animal experimentation, namely, the National Institutes of Health’s “Principles of Laboratory Animal Care” and the “Guide for the Care and Use of Laboratory Animals.” The experiment was approved by Jianghan University experimental animal ethical review committee and the approval number is 2016100006.
Informed consent. This article does not contain any studies involving human participants performed by any of the authors.
Availability of data and materials. The data sets generated during the current study are available from the corresponding author on reasonable request.
Author’s contributions. JZ was responsible for the study concept and design. XKG constructed the self-administration animal model. JZ and TTN drafted the manuscript. All authors critically reviewed content and approved the final version for publication.
Rights and permissions
About this article
Cite this article
Zeng, C., Gong, X., Dai, X. et al. Metabolomic Analysis of the Serum in Rats with Methamphetamine-Induced Conditioned Place Preference. Neurochem. J. 14, 424–428 (2020). https://doi.org/10.1134/S1819712420040091
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1819712420040091