Abstract
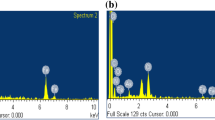
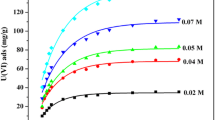
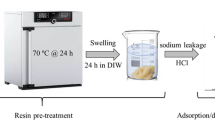
A static-batch technique was used to demonstrate the adsorption behavior of Re (VII) and Mo ions onto Dowex 21K at equilibrium in single and binary component systems. The single equilibrium adsorption data were modeled through a linear form of four widely used equilibrium isotherm equations. The results indicated that Freundlich and D-R models for Re, and Temkin and D–R isotherms for Mo fitted the obtained data satisfactorily. Binary adsorptions of Re and Mo ions onto Dowex 21K were also analyzed using Extended Langmuir, Modified Langmuir, Extended Freundlich and Langmuir–Freundlich models. The competitive Extended Freundlich model fitted the binary adsorption equilibrium data adequately. Studies on mutual interference effects of Mo ions on Re adsorption capacity indicated that the adsorption of perrhenate ions is always suppressed. In this perspective, the results from EDX studies confirmed the rhenium atom decrease in the simulated Re–Mo adsorption. However, under the studied conditions the affinity of the Dowex 21K for rhenium ions is marginally greater than that of molybdenum ions.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bryce, C.W. and Shaw, M., Mining Eng., 2014, vol. 9, no. 66, p.6.
Blokhin, A., Amosov, A., Murashkin, Y.V., Evdoshenko, S., Mikhailenko, M. and Nikitin, N., Russ. J. Appl. Chem., 2005, vol. 78, no. 9, p. 1411–1415.
Ma, H.Z., Lan, X.Z., and Wang, Y.N., Adv. Mater. Res., 2011.
Mal’tseva, E., Blokhin, A. and Murashkin, Y. V., Russ. J. Appl. Chem., 2012b, vol. 85, no. 7, p. 1034–1040.
Kholmogorov, A., Kononova, O., Kachin, S., Ilyichev, S., Kryuchkov, V., Kalyakina, O. and Pashkov, G., Hydrometallurgy, 1999, vol. 51, no. 1, p. 19–35.
Leszczynska-Sejda, K., Benke, G., Chmielarz, A., Krompiec, S., Michalik, S. and Krompiec, M., Hydrometallurgy, 2007, vol. 89, no. 3, p. 289–296.
Liu, W., Zhang, P., Borthwick, A.G., Chen, H. and Ni, J., J. Coll. & Int. Sci., 2014, vol. 423, p. 67–75.
Van Deventer, J., Solvent Extraction and Ion Exchange, 2011, vol. 29, nos. 5, 6, p. 695–718.
Senthilkumar, G., Murugappan, A., Int. J. Eng. Res. & Techn. (IJERT), 2015a, vol. 4, no.08.
Sitaram, R. and Khopkar, S., Microchimica Acta, 1976, vol. 65, no. 1, p. 1–10.
Sitaram, R. and Khopkar, S., Chromatographia, 1973, vol. 6, no. 4, p. 198–201.
Chakravorty, M. and Khopkar, S., Chromatographia, 1976, vol. 9, no. 5, p. 230–232.
Lopes, C.B., Lito, P.F., Cardoso, S.P., Pereira, E., Duarte, A.C. and Silva, C.M., Ion Exchange Technology II, Springer, 2012, p. 237–322.
Joo, S.-H., Kim, Y.-U., Kang, J.-G., Kumar, J.R., Yoon, H.-S., Parhi, P. and Shin, S. M., Materials Transactions, 2012, vol. 53, no. 11, p. 2034–2037.
Lou, Z., Zhao, Z., Li, Y., Shan, W., Xiong, Y., Fang, D., Yue, S. and Zang, S., Biores. Techn., 2013, vol. 133, p. 546–554.
Kadirvelu, K., Goel, J. and Rajagopal, C., J. Hazar. Mat., 2008, vol. 153, no. 1, p. 502–507.
Shahmohammadi-Kalalagh, S., Caspian J. Env. Sci., 2011, vol. 9, no. 2, p. 243–255.
Günay, A., Arslankaya, E. and Tosun, I., J. Hazar. Mat., 2007, vol. 146, no. 1, p. 362–371.
Hobson, J.P., J. Phys. Chem., 1969, vol. 73, no. 8, p. 2720–2727.
Singha, B. and Das, S.K., Col. & Sur., B: Biointerfaces, 2013, vol. 107, p. 97–106.
Senthilkumar, G. and Murugappan, A, International J. ChemTech Research, 2015, vol. 8, p.19.
Sukpreabprom, H., Arqueropanyo, O.-A., Naksata, W., Sooksamiti, P., and Janhom, S., Korean J. Chem. Eng., 2015, vol. 32, no. 5, p. 896–902.
Fouladgar, M., Beheshti, M., and Sabzyan, H., J. Mol. Liq., 2015, vol. 211, p. 1060–1073.
Chen, Y.-G., He, Y., Ye, W.-M., and Jia, L.-Y., J. Ind. & Eng. Chem., 2015, vol. 26, p. 335–339.
Zhu, Y., Hu, J., and Wang, J., J. Hazar. Mat.,, 2012, vol. 221, p. 155–161.
Sigma Aldrich Co., 2017.
Rahmani, A., Mousavi, H.Z., and Fazli, M., Desalination, 2010, vol. 253, no. 1, p. 94–100.
Mikhaylenko, M. and Blokhin, A., Preprint 12–156, SME Annual Meeting, Feb. 19–22, 2012, Seattle, Washington, 2012.
Hubicki, Z. and Kolodynska, D., Selective Removal of Heavy Metal Ions from Waters and Waste Waters Using Ion Exchange Methods, INTECH Open Access Publisher, 2012.
Coutinho, F., Carvalho, D., Aponte, M.L.T., and Barbosa, C., Polymer, 2001, vol. 42, no. 1, pp. 43–48.
Author information
Authors and Affiliations
Corresponding author
Additional information
The text was submitted by the authors in English.
Rights and permissions
About this article
Cite this article
Fathi, M.B., Rezai, B., Alamdari, E.K. et al. Mechanism and equilibrium modeling of Re and Mo adsorption on a gel type strong base anion resin. Russ J Appl Chem 90, 1504–1513 (2017). https://doi.org/10.1134/S1070427217080208
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070427217080208