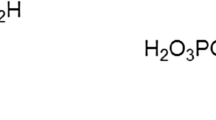Abstract
It was found that introduction of polyhexamethylguanidines modified with carboxy and phosphonic groups into a saturated solution of calcium carbonate affects the formation rate, morphology, and phase composition of the deposits being formed, with crystals in the deposit becoming coarser and rhombic structures appearing. These phenomena were manifested to a greater extent when zinc complexes of polyelectrolytes are present in solution. An analysis of the phase composition of the deposit demonstrated that the presence of polyelectrolytes and their zinc complexes in solution leads to an increase in the amount of the calcite phase. The reason why the phase composition and morphology of the deposits changes is that polyelectrolytes and their complexes are adsorbed on the surface of carbonate deposits. The adsorption of polyelectrolytes on a calcium carbonate powder is described by the Langmuir isotherm and the formation of their zinc complexes leads to an increase in the adsorption equilibrium constant by more than a factor of 2, which is due to the change in the hydrophilic-lipophilic balance of macromolecules in complexation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Chausov, F.F., Ekolog. Prom-st’ Rossii, 2008, no. 9, pp. 28–33.
Fedorenko, V.I., Kritich. Tekhnol., Membrany, 2003, vol. 18, no. 2, pp. 23–30.
Potapov, S.A., Ezhov, A.Yu., and Martynova, I.A., Korroz.: Mater., Zashch., 2006, no. 12, pp. 28–31.
Kuznetsov, Y.I., Russ. Chem. Rev., 2004, vol. 73, no. 1, pp. 75–87.
Chirkunov, A.A., Kuznetsov Yu.I., Tomin V.P. Korroz.: Mater. Zashch., 2007, no. 4, pp. 18–23.
Antonik, L.M., Lopyrev, V.A., and Korchevin, N.A., Russ. J. Appl. Chem., 2002, vol. 75, no. 2, pp. 257–260.
USSR Inventor’s Certificate no. 513995; Bull. Izobret., 1976, no. 18.
Obraztsov, V.B., Rublova, E.D., and Amirulloeva, N.V., Mater. Sci., 2013, vol. 49, no. 3, pp. 326–333.
Dyatlova, N.M., Temkina, V.Ya., and Popov, K.I., Kompleksony i kompleksonaty metallov (Complexones and Complexonates of Metals), Moscow: Khimiya, 1988.
GOST (State Standard) 4151-72, Method for Determining the General Hardness.
Zhikharev, Yu.P., Perekupka, A.G., and Zakirova, A.I., Vestn. Tyumen. Gos. Univ., 2007, no. 3, pp. 129–138.
Shestak, I.V., Vorob’ev, P.D., Cherednichenko, D.V., et al., Russ. J. Inorg. Chem., 2011, vol. 56, no. 2, pp. 176–180.
Adamson, A.A., Physical Chemistry of Surfaces, New York: Wiley, 1976, 3rd ed.
Damaskin, B.B., Petrii, O.A., and Batrak, V.V., Adsorbtsiya organicheskikh soedinenii na elektrodakh (Adsorption of Organic Compounds on Electrodes), Moscow: Nauka, 1968.
Powder Diffraction File, International Centre for Diffraction Data, Swarthmore Pennsylvania, USA, Sets 1-45.
Gorelik, S.S., Skakov, Yu.A., and Rastorguev, L.N., Rentgenograficheskii i elektronno-opticheskii analiz (X-ray Diffraction and Electron-Optical Analyses), Moscow: Mosk. Inst. Stali Splavov, 1994.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © E.D. Rublova, V.B. Obraztsov, F.I. Danilov, 2014, published in Zhurnal Prikladnoi Khimii, 2014, Vol. 87, No. 12, pp. 1756–1761.
Rights and permissions
About this article
Cite this article
Rublova, E.D., Obraztsov, V.B. & Danilov, F.I. Effect of adsorption of polyhexamethyleneguanidine derivatives on the formation rate, morphology, and phase composition of carbonate deposits. Russ J Appl Chem 87, 1836–1841 (2014). https://doi.org/10.1134/S1070427214120076
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070427214120076