Abstract
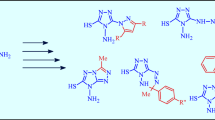
The reaction of 1-alkyl-2-chloro-1H-indole-3-carbaldehydes with 4-amino-5-alkyl(aryl)-4H-1,2,4-triazole-3-thiols afforded new heterocyclic compounds, namely triazolo(thiadiazepino)indoles. Structure of the latter was established by single crystal X-ray diffraction method. This heterocyclic system is formed upon cyclization of the intermediate 5-alkyl-4-[indol-3-yl(methylideneamino)]-4H-1,2,4-triazole-3-thiols.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Corey, E.J., Czako, B., and Kurti, L., Molecules and Medicine, Hoboken: Wiley, 2007. https://doi.org/10.1002/bmb.20179
Pozharskii, A.F., Soldatenkov, A.T., and Katritzky, A.R., Heterocycles in Life and Society: An Introduction to Heterocyclic Chemistry, Biochemistry and Applications, Chichester: Wiley, 2011. https://doi.org/10.1002/9781119998372
Chitra, G., Franklin, D.S., and Guhanathan, S., J. Macromol. Sci. (A), 2017, vol. 54, p. 151. https://doi.org/10.1080/10601325.2017.1265401
Abdel-Gawad, H., Mohamed, H.A., and Dawood, K.M., Chem. Pharm. Bull., 2010, vol. 58, p. 1529. https://doi.org/10.1248/cpb.58.1529
Dandia, A., Singh, R., Khaturia, S., Mérienne, C., Morgant, G., and Loupy, A., Bioorg. Med. Chem., 2006, vol. 14, p. 2409. https://doi.org/10.1016/j.bmc.2005.11.025
Winter, C.A., Risley, E.A., and Nuss, G.W., J. Pharmacol. Exp. Ther., 1963, vol. 141, p. 369.
Karthikeyan, S.V., Perumal, S., Shetty, K.A., Yogeeswari, P., and Sriram, D., Bioorg. Med. Chem. Lett., 2009, vol. 19, p. 3006. https://doi.org/10.1016/j.bmcl.2009.04.029
Jha, M., Edmunds, M., Lund, K.-I., and Ryan, A., Tetrahedron Lett., 2014, vol. 55, p. 5691. https://doi.org/10.1016/j.tetlet.2014.08.100
Zhao, N., Qiu, L., Wang, X., An, Z., and Wan, X., Tetrahedron Lett., 2014, vol. 55, p. 1040. https://doi.org/10.1016/j.tetlet.2013.12.076
Jakobsen, P., Kanstrup, A., Faarup, P., and Olesen, P.H., US Patent 5536721, 1996.
Suzdalev, K.F., Vikrischuk, N.I., Prikhodko, K.A., Shasheva, E.Yu., Kurbatov, S.V., Bogus, S.K., and Galenko-Yaroshevsky, P.A., Chem. Heterocycl. Compd., 2016, vol. 5, p. 303. https://doi.org/10.1007/s10593-016-1882-y
Bazian, A., Taheri, M., and Alavi, H., Russ. J. Gen. Chem., 2014, vol. 84, no. 3, p. 586. https://doi.org/10.1134/S107036321403030X
Sheldrick, G.M., SHELXTL, Bruker AXS Inc., Madison, Wisconsin, USA, 2000.
Acknowledgments
IR and NMR spectra were recorded at the Center for Collective Use “Molecular Spectroscopy” of the Southern Federal University and the Educational Research Laboratory of Resonance Spectroscopy, Department of Chemistry of Natural and High Molecular Compounds of the Southern Federal University.
Funding
This work was supported by the Southern Federal University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
No conflict of interest was declared by the authors.
Additional information
Russian Text © The Author(s), 2019, published in Zhurnal Obshchei Khimii, 2019, Vol. 89, No. 12, pp. 1848–1853.
Rights and permissions
About this article
Cite this article
Vikrishchuk, N.I., Suzdalev, K.F., Dranikova, A.Y. et al. Reactions of 1-Alkyl-2-chloro-1H-indole-3-carbaldehyde with 4-Amino-5-alkyl(aryl)-4H-triazole-3-thioles. Russ J Gen Chem 89, 2369–2373 (2019). https://doi.org/10.1134/S1070363219120077
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363219120077