Abstract
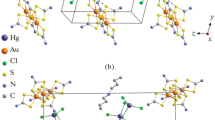
Novel pseudo-polymeric complexes of gold(III)-mercury(II) with cyclic alkylene dithiocarbamate ligands: ([Au{S2CN(CH2)6}2][HgCl3])n, ([Au{S2CN(CH2)6}2]2[HgCl4]·H2O)n, and ([Au{S2CN(CH2)4O}2]2[Hg2Cl6])n have been obtained and structurally characterized. The sophisticated supramolecular structure of the obtained compounds is realized due to secondary non-valent Au⋯S interactions and includes mononuclear and binuclear cations and anions as structural units as well as cationic and anionic polymer chains. The thermal behavior of the obtained complexes has been studied by means of simultaneous thermal analysis. The products of thermal transformations of the complexes are reduced elemental gold and HgCl2.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Kloppers, L., Maree, W., Oyekola, O., and Hangone, G., Miner. Engin., 2016, vol. 87, p. 54. https://doi.org/10.1016/j.mineng.2015.12.003
Nieuwenhuizen, P.J., Appl. Catal. (A), 2001, vol. 207, nos. 1–2, p. 55. https://doi.org/10.1016/S0926-860X(00)00613-X
Onwudiwe, D.C. and Ajibade, P.A., Mater. Lett., 2011, vol. 65, no. 21–22, p. 3258. https://doi.org/10.1016/j.matlet.2011.07.012
Sathiyaraj, E. and Thirumaran, S., Spectrochim. Acta (A), 2012, vol. 97, p. 575. https://doi.org/10.1016/j.saa.2012.06.052
Srinivasan, N. and Thirumaran, S., Superlatt. Microstruct., 2012, vol. 51, no. 6, p. 912. https://doi.org/10.1016/j.spmi.2012.03.006
Nami, S.A.A., Husain, A., and Ullah, I., Spectrochim. Acta (A), 2014, vol. 118, p. 380. https://doi.org/10.1016/j.saa.2013.08.064
Prakasam, B.A., Lahtinen, M., Peuronen, A., Muruganandham, M., Kolehmainen, E., Haapaniemi, E., and Sillanpää, M., Mater. Lett., 2015, vol. 144, p. 19. https://doi.org/10.1016/j.matlet.2014.12.128
Hrubaru, M., Onwudiwe, D.C., and Hosten, E., J. Sulfur Chem., 2016, vol. 37, no. 1, p. 37. https://doi.org/10.1080/17415993.2015.1080707
Chesman, A.S.R., van Embden, J., Duffy, N.W., Webster, N.A.S., and Jasieniak, J.J., Cryst. Growth Des., 2013, vol. 13, p. 1712. https://doi.org/10.1021/cg4000268
Ronconi, L., Giovagnini, L., Marzano, C., Bettìo, F., Graziani, R., Pilloni, G., and Fregona, D., Inorg. Chem., 2005, vol. 44, no. 6, p. 1867. https://doi.org/10.1021/ic048260v
Ronconi, L., Marzano, C., Zanello, P., Corsini, M., Miolo, G., Maccà, C., Trevisan, A., and Fregona, D., J. Med. Chem., 2006. Vol.49, no. 5, p. 1648. https://doi.org/10.1021/jm0509288
Milacic, V., Chen, D., Ronconi, L., Landis-Piwowar, K.R., Fregona, D., and Dou, Q.P., Cancer Res., 2006, vol. 66, no. 21, p. 10478. https://doi.org/10.1158/0008-5472.CAN-06-3017
Mansour, M.A., Connick, W.B., Lachicotte, R.J., Gysling, H.J., and Eisenberg, R., J. Am. Chem. Soc., 1998, vol. 120, no. 6, p. 1329. https://doi.org/10.1021/ja973216i
Han, S., Jung, O.-S., and Lee, Y.-A., Trans. Met. Chem., 2011, vol. 36, no. 7, p. 691. https://doi.org/10.1007/s11243-011-9521-z
Ivanov, A.V., Korneeva, E.V., Bukvetskii, B.V., Goryan, A.S., Antzutkin, O.N., and Forsling, W., Russ. J. Coord. Chem., 2008, vol. 34, no. 1, p. 59. https://doi.org/10.1007/s11173-008-1010-3
Loseva, O.V., Rodina, T.A., Antzutkin, O.N., and Ivanov, A.V., Russ. J. Gen. Chem., 2018, vol. 88, no. 12, p. 2540. https://doi.org/10.1134/S1070363218120149
Loseva, O.V., Rodina, T.A., Smolentsev, A.I., and Ivanov, A.V., Polyhedron, 2017, vol. 134, p. 238. https://doi.org/10.1016/j.poly.2017.06.021
Loseva, O.V., Rodina, T.A., and Ivanov, A.V., Russ. J. Coord. Chem., 2019, vol. 45, no. 1, p. 22. https://doi.org/10.1134/S1070328419010068
Jotani, M.M., Tan, Y.S., and Tiekink, E.R.T., Z. Kristallogr., 2016, vol. 231, no. 7, p. 403. https://doi.org/10.1515/zkri-2016-1943
Howie, R.A., Tiekink, E.R.T., Wardell, J.L., and Wardell, S.M.S.V., J. Chem. Crystallogr., 2009, vol. 39, no. 4, p. 293. https://doi.org/10.1007/s10870-008-9473-0
Loseva, O.V., Rodina, T.A., Smolentsev, A.I., and Ivanov, A.V., Russ. J. Coord. Chem., 2016, vol. 42, no. 11, p. 719. https://doi.org/10.1134/S1070328416110063
Byr’ko, V.M., Ditiokarbamaty (Dithiocarbamates), Moscow: Nauka, 1984.
Bellamy, L.J., The Infrared Spectra of Complex Molecules, New York: Wiley, 1958.
Casas, J.S., Sanchez, A., Bravo, J., Soledad, G.F., Castellano, E.E., and Jones, M.M., Inorg. Chim. Acta, 1989, vol. 158, no. 1, p. 119. https://doi.org/10.1016/S0020-1693(00)84021-9
Ehsan, M.A., Khaledi, H., Tahir, A.A., Ming, H.N., Wijayantha, K.G.U., and Mazhar, M., Thin Solid Films, 2013, vol. 536, p. 124. https://doi.org/10.1016/j.tsf.2013.03.092
Yin, H., Li, F., and Wang, D., J. Coord. Chem., 2007, vol. 60, no. 11, p. 1133. https://doi.org/10.1080/00958970601008846
Khitrich, N.V. and Seifullina, Russ. J. Coord. Chem., 2000, vol. 26, no. 11, p. 798.
Kazitsyna, L.A. and Kupletskaya, N.B., Primenenie UF-, IK-, YaMR- i mass-spektroskopii v organicheskoi khimii (The Use of UV, IR, NMR, and Mass Spectroscopy in Organic Chemistry), Moscow: Mosk. Univ., 1979.
Pauling, L., The Nature of the Chemical Bond and the Structure of Molecules and Crystals, London: Cornell Univ. Press, 1960.
Bondi, A., J. Phys. Chem., 1964, vol. 68, no. 3, p. 441. https://doi.org/10.1021/j100785a001
Bocian, D.F., Pickett, H.M., Rounds, T.C., and Strauss, H.L., J. Am. Chem. Soc., 1975, vol. 97, no. 4, p. 687. https://doi.org/10.1021/ja00837a001
Yang, L., Powel, D.R., and Houser, R.P., Dalton Trans., 2007, no. 9, p. 955. https://doi.org/10.1039/b617136b
Alcock, N.W., Adv. Inorg. Chem. Radiochem., 1972, vol. 15, no. 1, p. 1. https://doi.org/10.1016/S0065-2792(08)60016-3
Bruker, APEX2 (version 1.08), SAINT (version 7.03), SADABS (version 2.11), SHELXTL (version 6.12). Madison (WI, USA): Bruker AXS Inc., 2004.
Angeloski, A., Rawal, A., Bhadbhade, M., Hook, J.M., Schurko, R.W., and McDonagh, A.M., Cryst. Growth Des, 2019, vol. 19, no. 2, p. 1125. https://doi.org/10.1021/acs.cgd.8b01619
Loseva, O.V., Rodina, T.A., Ivanov, A.V., Smolentsev, A.I., and Antzutkin, O.N., Russ. Chem. Bull, 2019, vol. 68, no. 4, p. 782. https://doi.org/10.1007/s11172-019-2486-3
Acknowledgments
Electron microscopy studies, determination of gold content in the solutions, and recording of energy-dispersive spectra were performed at the Analytical Center for Mineralogy and Geochemical Studies, Institute of Geology and Nature Management, Far Eastern Branch of the RAS. IR spectra were recorded at the Center For Collective Usage, Kurnakov Institute of General and Inorganic Chemistry, Russian Academy of Sciences.
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of Interest
No conflict of interest was declared by the authors.
Russian Text © The Author(s), 2019, published in Zhurnal Obshchei Khimii, 2019, Vol. 89, No. 11, pp. 1766–1779.
Rights and permissions
About this article
Cite this article
Loseva, O.V., Rodina, T.A. & Ivanov, A.V. Double Pseudo-Polymeric Gold(III)-Mercury(II) Complexes [Au(S2CNR2)2]nX [R2 = (CH2)6, (CH2)4O] Containing ([HgCl3]−)n, [HgCl4]2− and [Hg2Cl6]2− Anions: Chemisorption Synthesis, Principles of Supramolecular Self-Assembly, and Thermal Behavior. Russ J Gen Chem 89, 2273–2284 (2019). https://doi.org/10.1134/S1070363219110185
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363219110185