Abstract
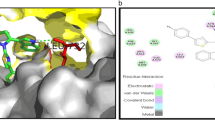
A series of new 3-(2-substituted)-4-(oxothiazolidin-3-yl)-2-methylquinazolin-4(3H)-ones 1a–1k is synthesized by using the hybridization approach via one pot multicomponent reaction of 3-amino-2-methylquinazolin-4(3H)-one with substituted benzaldehyde, thioglycolic acid and N, N-dicyclohexylcarbodiimide in DMF media. Structures of the synthesized compounds are elucidated from the spectral data. Antidiabetic activity of the products is tested against streptozotocin induced diabetic rats at a dose of 200 mg/kg compared with standard Pioglitazone (15 mg/kg). Compounds 1b, 1d, 1f, and 1i demonstrate significant antidiabetic activity. Compounds 1b, 1d, 1f, and 1i are evaluated in vitro are tested for serum insulin, cholesterol, triglycerides, total protein, lipoprotein, and enzymes factors. Significant lowering of glycated hemoglobin level is induced by the compounds after 21 days of treatment. Mean±S.E.M. data accumulated are subjected to one-way analysis of variance (ANOVA) followed by Dunnett’s t-test. p < 0.001 was considered statistically significant. Histopathological results accumulated for the rats treated by compounds 1b, 1d and 1f confirm the significant recovery of pancreas destruction. Free energy of binding for all synthesized compounds is calculated using AutoDock 1.5.6 with peroxisome proliferator-activated receptor γ (PPAR γ; PDB ID: 4PRG). Among the synthesized compounds, 1d demonstrates significant binding energy value of −11.46 kcal/mol. The current study is expected to provide useful insight into the design of potential agents that can act as a platform for the development of future antidiabetic drugs.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Deshmukh, A.R., Bhosle, M.R., Khillare, L.D., Dhumal, S.T., Mishra, A., Srivastava, A.K., and Mane, R.A., Res. Med. Intermed., 2017, vol. 43, p. 1107. doi https://doi.org/10.1007/s11164-016-2686-5
Moller, D.E., Nature, 2001, vol. 414, p. 821. doihttps://doi.org/10.1038/414821a
Sahay, R.K., Reddy, V.S., Bhadada, S.K., Agrawal, J.K., and Agrawal, N.K., J. Indian. Acad. Clin. Med., 2000, vol. 1, p. 245.
Mahapatra, M.K., Saini, R., and Kumar, M., Res. Chem. Intermed., 2016, vol. 42, p. 8239. doi https://doi.org/10.1007/s11164-016-2592-x
Day, C., Diabet. Med., 1999, vol. 16, p. 179. doi https://doi.org/10.1046/j.1464-5491.1999.00023.x
Kim, B.Y., Ahn, J.B., Lee, H.W., Kang, S.K., Lee, J.H., Shin, J.S., Ahn, S.K., Hong, C I., and Yoon, S.S., Eur. J. Med. Chem., 2004, vol. 39, p. 433. doi https://doi.org/10.1016/j.ejmech.2004.03.001
Laresn, T.M., Toubro, S., and Astrup, A., Int. J. Obes. Relat. Metab. Disord., 2003, vol. 27, p. 147. doi https://doi.org/10.1038/sj.ijo.802223
Vigorita, M.G., Ottana, R., Monforte, F., Maccari, R., Trovato, A., Monforte, M.T., and Taviano, M.F., Bioorg. Med. Chem. Lett., 2001, vol. 11, p. 2791. doi https://doi.org/10.1016/S0960-894X(01)00476-0
Goel, B., Ram, T., Tyagi, R., Bansal, E., Kumar, A., Mukherjee, D., and Sinha, J.N., Eur. J. Med. Chem., 1999, vol. 34, p. 265. doi https://doi.org/10.1016/S0223-5234(99)80060-9
Geronikaki, A.A., Pitta, E.P., and Liaras, K.S., Curr. Med. Chem., 2013, vol. 20, p. 4460. doi https://doi.org/10.2174/09298673113209990143
Babaoglu, K., Page, M.A., Jones, V.C., McNeil, M.R., Dong, C., Naismith, J.H., and Lee, R.E., Bioorg. Med. Chem. Lett., 2003, vol. 13, p. 3227. doi https://doi.org/10.1016/S0960-894X(03)00673-5
Dayam, R., Aiello, F., Deng, J., Wu, Y., Garofalo, A., Chen, X., and Neamati, N., J. Med. Chem., 2006, vol. 49, p. 4526. doi https://doi.org/10.1021/jm051296s
Chaudhari, M A., Gujar, J.B., Kawade, D.S., Shinde, P.V., and Shingare, M.S., Res. Chem. Intermed., 2015, vol. 41, p. 10027. doi https://doi.org/10.1007/s11164-015-2010-9
Maccari, R., Vitale, R.M., Ottana, R., Rocchiccioli, M., Marrazzo, A., Cardile, V., Graziano, A.C., Amodeo, P., Mura, U., and Del Corso, A., Eur. J. Med. Chem., 2014, vol. 81, p. 1. doi https://doi.org/10.1016/j.ejmech.2014.05.003
Archana, Srivastava, V.K. and Kumar, A., Eur. J. Med. Chem., 2002, vol. 37, p. 873. doi https://doi.org/10.1016/S0223-5234(02)01389-2
Chapleo, C.B., Myers, M., Myers, P.L., Saville, J.F., Smith, A.C.B., Stillings, M.R., Tulloch, I.F., Walter, D.S., and Welbourn, A.P., J. Med. Chem., 1986, vol. 29, p. 2273. doi https://doi.org/10.1021/jm00161a024
Srivastava, V K., Archana, and Kumar, A., Bioorg. Med. Chem., 2004, vol. 12, p. 1257. doi https://doi.org/10.1016/j.bmc.2003.08.035
Aziza, M.A., Ibrahim, M.K., and El-Helpy, A.G., Al-Azhar. J. Pharm. Sci., 1994, vol. 14, p. 193.
Bekhit, A.A. and Khalil, M.A., Pharmazie, 1998, vol. 53, p. 539.
Bedi, P.M.S., Kumar, V., and Mahajan, M.P., Bioorg. Med. Chem. Lett., 2004, vol. 14, p. 5211. doi https://doi.org/10.1016/j.bmcl.2004.07.065
Kung, P.P., Casper, M.D., Cook, K.L., Wilson-Lingardo, L., Risen, L.M., Vickers, T.A., Ranken, R., Blyn, L.B., Wyatt, J.R., Cook, P.D., and Ecker, D.J., J. Med. Chem., 1999, vol. 42, p. 4705. doi https://doi.org/10.1021/jm9903500
Mohamed, M.S., Kamel, M.M., Kassem, E.M., Abotaleb, N., El-moez, S.I.A., and Ahmed, M.F., Eur. J. Med. Chem., 2010, vol. 45, p. 3311. doi https://doi.org/10.1016/j.ejmech.2010.04.014
Ram, V.J., Farhanullah, Tripathi, B.K., and Srivastava, A.K., Bioorg. Med. Chem., 2003, vol. 11, p. 2439. doi https://doi.org/10.1016/S0968-0896(03)00142-1
Kavitha, C.V., Basappa, Swamy, S.N., Mantelingu, K., Doreswamy, S., Sridhar, M.A., Prasad, J.S., and Rangappa, K.S., Bioorg. Med. Chem., 2006, vol. 14, p. 2290. doi https://doi.org/10.1016/j.bmc.2005.11.017
Suthar, S.K., Jaiswal, V., Lohan, S., Bansal, S., Chaudhary, A., Tiwari, A., Alex, A.T., and Joseph, A., Eur. J. Med. Chem., 2013, vol. 63, p. 589. doi https://doi.org/10.1016/j.ejmech.2013.03.011
Lipinski, C.A., Lombardo, F., Dominy, B.W., and Feeney, P.J., Adv. Drug. Deliv. Rev., 2001, vol. 46, p. 3. doi https://doi.org/10.1016/S0169-409X(00)00129-0
Monday, O.M. and Uzoma, A.I., Asian. Pac. J. Trop. Biomed., 2013, vol. 3, p. 628. doi https://doi.org/10.1016/S2221-1691(13)60127-6
Bala, A., Kar, B., Haldar, P.K., Mazumder, U.K., and Bera, S., J. Ethnopharmacol., 2010, vol. 129, p. 131. doi https://doi.org/10.1016/j.jep.2010.03.010
Murtaza, B., Abbas, A., Aslam, A., Akhtar, M.S., Bashir, S., Khalid, M., and Naseer, M.M., Res. Chem. Intermed., 2016, vol. 42, p. 4161. doi https://doi.org/10.1007/s11164-015-2266-0
Andrarde-Cetto, A., Wiedenfeld, H., J. Ethnopharmacol., 2001, vol. 78, p. 145. doi https://doi.org/10.1016/S0378-8741(01)00335-X
Shirwaikar, A., Rajendran, K., and Barik, R., J. Ethnopharmacol., 2006, vol. 107, p. 285. doi https://doi.org/10.1016/j.jep.2006.03.012
Mariappan, G., Saha, B.P., Datta, S., Kumar, D., and Haldar, P.K., J. Chem. Sci., 2011, vol. 123, p. 335. doi https://doi.org/10.1007/s12039-011-0079-2
Jarrett, R.J., Keen, H., and Hardwick, C., Diabetes, 1970, vol. 19, p. 724. doi https://doi.org/10.2337/diab.19.10.724
Latner, A., Clinical Biochemistry, in Carbohydrate Metabolism: Abnormalities of Post Absorptive Blood Sugar Level, Philadelphia: WB Saunders Co., 1958, p. 48.
Ghosh, S. and Suryawanshi, S.A., Indian. J. Exp. Biol., 2001, vol. 39, p. 748.
Hall, P.M., Cook, J.G., Sheldon, J., Rutherford, S.M., and Gould, B.J., Diabetes Care, 1984, vol. 7, p. 147. doi https://doi.org/10.2337/diacare.7.2.147
Kishore, A., Nampurath, G.K., Mathew, S.P., Zachariah, R.T., Potu, B.K., Rao, M.S., Valiathan, M., and Chamallamudi, M.R., Chem.-Biol. Interaction., 2009, vol. 177, p. 242. doi https://doi.org/10.1016/j.cbi.2008.10.032
Strate, T., Mann, O., Kleinhans, H., Rusani, S., Schneider, C., Yekebas, E., Freitag, M., Stand, T., Bloechle, C., and Lzbicki, J.R., Pancreas, 2005, vol. 30, p. 254. doi https://doi.org/10.1097/01.mpa.0000157481.22155.2d
Oberfield, J.L., Collins, J.L., Holmes, C.P., Goreham, D.M., Cooper, J.P., Cobb, J.E., Lenhard, J.M., Hull-Ryde, E.A., Mohr, C.P., Blanchard, S.G., Parks, D.J., Moore, L.B., Lehmann, J.M., Plunket, K., Miller, A.B., Milburn, M.V., Kliewer, S.A., and Wilson, T.M., Proc. Natl. Acad. Sci. USA, 1999, vol. 96, p. 6102. doi https://doi.org/10.1073/pnas.96.11.6102
Willson, T.M. and Wahli, W., Curr. Opin. Chem. Biol., 1997, vol. 1, p. 235. doi https://doi.org/10.1016/S1367-5931(97)80015-4
Houssen, M., Shahin, D., Eltoraby, E., and Mesbah, A., Clin. Biochem., 2010, vol. 43, p. 661. doi https://doi.org/10.1016/j.clinbiochem.2010.01.012
Eschwege, E. and Fontbonne, A., Transplant. Proc., 1992, vol. 24, p. 767.
Macdonald, R.P., Standard Methods of Clinical Chemistry, New York: Academic Press, 1970, p. 215.
Balakrishnan, S., Pandhare, R., Malays. J. Pharm. Sci., 2010, vol. 8, p. 13.
Friedewald, W.T., Levy, R.I., and Fredrickson, D.S., Clin. Chem., 1972, vol. 18, p. 499.
Assmann, G., Schriewer, H., Schmitz, G., and Hagele, E.O., Clin. Chem., 1983, vol. 29, p. 2026.
Donga, S., Shukla, V.J., Ravishankar, B., Ashok, B.K., and Mishtry, I.U., Ayu., 2011, vol. 32, p. 120. doi https://doi.org/10.4103/0974-8520.85743
Kumar, S., Malhotra, R., and Kumar, D., Ind. J. Pharm. Sci., 2010, vol. 72, p. 533. doi https://doi.org/10.4103/0250-474X.73921
Bonner-Weir, S., Diabetes, 1988, vol. 37, p. 616. doi https://doi.org/10.2337/diab.37.5.616
Masiello, P., Broca, C., Gross, R., Roye, M., Manteghetti, M., Hillaire-Buys, D., Novelli, M., and Ribes, G., Diabetes, 1998, vol. 47, p. 224. doi https://doi.org/10.2337/diab.47.2.224
Sekar, N., Kanthasamy, A., William, S., Subramanian, S., and Govindasamy, S., Pharmacol. Res., 1990, vol. 22, p. 207. doi https://doi.org/10.1016/1043-6618(90)90717-R
Morris, G.M., Huey, R., Lindstrom, W., Sanner, M.F., Belew, R.K., Goodsell, D.S., and Olson, A.J., J. Comput. Chem., 2009, vol. 30, p. 2785. doi https://doi.org/10.1002/jcc.21256
Solis, F.J. and Wets, R.J.B., Math. Oper. Res., 1981, vol. 6, p. 19. doi https://doi.org/10.1287/moor.6.1.19
Acknowledgments
The authors gratefully acknowledge Head, Central Instrumentation Facility (CIF) department, Savitribai Phule Pune University, Pune, India for providing the facilities for Spectral Analysis of compounds.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jangam, S.S., Wankhede, S.B. Synthesis, Molecular Docking, and Biological Evaluation of the New Hybrids of 4-Thiazolidinone and 4(3H)-Quinazolinone Against Streptozotocin Induced Diabetic Rats. Russ J Gen Chem 89, 1029–1041 (2019). https://doi.org/10.1134/S1070363219050256
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363219050256