Abstract
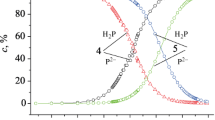
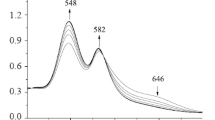
2,3,7,8,12,13,17,18-Ocatbromo-5,10,15,20-tetra-(4-chloroprienyl)porphyrin and 2,3,7,8,12,13,17,18-octachloro-5,10,15,20-tetra-(4-bromophenyl)porphyrin have been synthesized. The obtained compounds have been identified by electronic absorption and 1H NMR spectroscopy as well as mass spectrometry. The complex-forming properties of the synthesized porphyrins in the zinc acetate (II)-acetonitrile system at 278–298 K have been studied. Kinetic parameters of the formation of the corresponding zinc complexes in acetonitrile have been determined.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Berezin, B.D., Coordination Compounds of Porphyrins and Phthalocyanines, New York: John Wiley and Sons, 1981, p. 286.
Senge, M.O., MacGowan, S.A., and O’Brien, J.M., Chem. Commun., 2015, vol. 51, no. 96, p. 17031. doi https://doi.org/10.1039/C5CC06254C
Xie, Y., Hill, J.P., Charvet, R., and Ariga, K., J. Nanosci. Nanotechnol., 2007, vol. 7, no. 9, p. 2969. doi https://doi.org/10.1166/jnn.2007.910
Röder, B., Büchner, M., Rückmann, I., and Senge, M.O., Photochem. Photobiol. Sci., 2010, vol. 9, no. 8, p. 1152. doi https://doi.org/10.1039/C0PP00107D
Porphyrins and Metalloporphyrins, Smith, K.M., Ed., Amsterdam: Elsevier Sci., 1975, p. 317.
The Porphyrins, Dolphin, D., Ed., New York: Academic Press, 1979, p. 463.
Marri, R., Grenner, D., Meyes, P., and Rodwell, V., Human Biochemistry, Moscow: Mir, 1993, vol. 1, p. 384.
Askarov, K.A., Berezin, B.D., and Bystritskaya, E.V., Porfiriny: spektroskopiya, elektrokhimiya, primenenie (Porphyrins: Spectroscopy, Electrochemistry, Application), Moscow: Nauka, 1987, p. 384.
Da Silva, V.S., Teixeira, L.I., do Nascimento, E., Idemori, Y.M., and De Freitas-Silva, G., Appl. Catal. (A), 2014, vol. 469, p. 124. doi https://doi.org/10.1016/j.apcata.2013.09.033
De Freitas Castro, K.A., de Lima, F.H.C., Simoes, M.M.Q., Neves, M.G.P.M.S., Almeida Paz, F.A., Mendes, R.F., Nakagaki, S., and Cavaleiro, J.A.S., Inorg. Chim. Acta, 2017, vol. 455, p. 575. doi https://doi.org/10.1016/j.ica.2016.05.038
Ali, B.B., Belkhiria, M.S., Giorgi, M., and Nasri, H., Open J. Inorg. Chem., 2011, vol. 1, p. 39. doi https://doi.org/10.4236/ojic.2011.13006
Ivanova, Yu.B., Pukhovskaya, S.G., Syrbu, S.A., and Mamardashvili, N.Zh., Abstracts of Papers, XIII Mezhdunar. konf. “Problemy sol’vatatsii i kompleksoobrazovaniya v rastvorakh” (XIII Int. Conf. “Problems of Solvation and Complexation in Solutions”), Suzdal, 2018, p. 68.
Ivanova, Yu.B., Churakhina, Yu.I., and Mamardashvili, N.Zh., Russ. J. Gen. Chem. 2008, vol. 78, no. 4, p. 673. doi https://doi.org/10.1134/S1070363208040269
Andrianov, V.G. and Malkova, O.V., Macroheterocycles, 2009, no. 2, p. 130. doi https://doi.org/10.6060/mhc2009.2.130
Pukhovskaya, S.G., Ivanova, Yu.B., Nam, D.T., and Vashurin, A.S., Russ. J. Phys. Chem., 2014, vol. 88, no. 10, p. 1670. doi https://doi.org/10.1134/S0036024414100288
Nam, D.T., Ivanova, Yu.B., Puhovskaya, S.G., Kruk, M.M., and Syrbu, A.S., RSC Adv., 2015, vol. 5, no. 33, p. 26125. doi https://doi.org/10.1039/C5RA01323B
Ivanova, Yu.B., Chizhova, N.V., Mamardashvili, N.Zh., and Pukhovskaya, S.G., Russ. J. Gen. Chem., 2014, vol. 84, p. 939. doi https://doi.org/10.1134/S1070363214050260
Senge, M.O., Chem. Commun., 2006, no. 3, p. 243. doi https://doi.org/10.1039/b511389j
Pukhovskaya, S.G., Ivanova, Yu.B., Dao The Nam, Vashurin, A.S., and Golubchikov, O.A., J. Porph. Phthal., 2015, vol. 19, p. 858. doi https://doi.org/10.1142/S1088424615500649
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ivanova, Y.B., Chizhova, N.V., Khrushkova, Y.V. et al. Synthesis, Spectral, and Coordination Properties of Halogen-Substituted Tetraarylporphyrins. Russ J Gen Chem 89, 459–465 (2019). https://doi.org/10.1134/S1070363219030150
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363219030150