Abstract
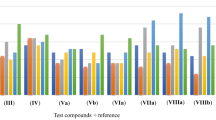
As a part of systematic study, new series of 6-{[5-(5-substituted 2-phenyl-1H-indol-3-yl)- methyleneamino]-1,3,4-oxadiazol-2-yl}-8-substituted-6H-indolo[3,2-c]isoquinolin-5(11H)-ones and their derivatives are synthesized and evaluated for their biological activities. Compound 8a displays potent antimicrobial activity against bacteria E. coli, K. penumoniae, and S. aureus, and fungi A. niger, A. flavus, and A. fumigates. Compounds 7a and 8a demonstrate promising radical scavenging and chelating with ferrous ions (Fe2+). According to the accumulated data the designed motifs exhibit higher biological activities than the reference compounds.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Saxena, V.-C., Bapat, S.-K., Dhawan, B.-N., Jpn. J. Pharmacol., 1969 vol. 19, p. 477, Chem. Abstr. 1970, vol. 72, 65152.
Cohen, A., US Patent 1967, no. 3 316 271. Chem Abstr., vol. 67, 21900s
Garmaise, D.-L. and Parks, G.V., US Patent 1973, no. 3 705 901; Chem Abstr., vol. 78, 72103.
Wang, Y.-H., Tang, J.-G., Wang, R.-R., Yang, L.-M., Dong, Z.-J., Shen, L. D.-X., Liu, J.-K., and Zheng, T., BioChem. Bioph. Res. Comm. 2007, vol. 355(4), p. 1091. doi https://doi.org/10.1016/j.bbrc.2007.02.081
Ishida, J., Wang, H.-K., Oyama, M., Cosentino, M.-L., Hu, C.-Q., and Lee, K.-H., J. Nat. Prod., 2001, vol. 6(7), p. 958. doi https://doi.org/10.1021/np0101189
Anelise, S., Formagio, N., Tonin, L.T.-D., Foglio, M.A., Madjarof, C., de Carvalho, J.E., da Costa, W.F., Cardoso, F-P., and Sarragiotto, M.-H., Bioorg. Med. Chem. 2008, vol. 16, p. 9660. doi https://doi.org/10.1016/j.bmc.2008.10.008
Hadjaz, F., Besret, S., and Martin-Nizard, F., Eur. J. Med. Chem. 2011, vol. 46(6), p. 2575. doi https://doi.org/10.1016/j.ejmech.2011.03.048
Winter, G., Dimola, N., Berti, M., and Ariali, V., Farmaco. Ed. Sci. 1979, vol. 34(6), p. 507.
Ishizumi, K. and Katsube, J., Brit. Patent, 1980, 2 025 932, Chem. Abstr., vol. 93, 186322e
Sumitomo Chemical Co. Ltd and Jpn. Kokai Tokkyo Hoho, Jap. Pantent, 1983, 5869 882 (8369 882); Chem. Abstr. 1983, vol. 99, 88182p
Kosuge, T., Zenda, H., Tamamoto, H., and Torigoe, Y., Jap. Pantent, 1973, 7391 210, Chem Abstr., 19780, 112616p.
Hiremath, S.-P., Saundane, A.-R., and Mruthyunjayaswamy, B.H.-M., J. Heterocycl. Chem., 1993, vol. 30, p.603.
Saundane, A.-R., Ranganath, S.-H., Prayagrai, G., Rudresh, K., and Satyanayana, N.D., Orient. J. Chem. 1998, vol. 14(2), p. 251.
Hoorocks, P., Fallon, S., Denman, L., Devine, O., Duffy, L.J., Harper, A., Meredith, E. L., Hasenkam, P.-S., Sidaway, A., Monnery, D., and Phillips, T.-R., Bioorg. Med. Chem. Lett. 2011, vol. 22(4), p. 1770. doi https://doi.org/10.1016/j.bmcl.2011.12.071
Zheng, X., Li, Z.-Y., and Wang, J. Flu. Chem., 2003, vol. 123(2), p. 163.
Amir, M.-S. and Shahani., Ind. J. Het. Chem. 1998, vol. 8, p. 107.
Shah, H.-P., Shah, B.-R., Bhatt, J.-J., Desai, N.-C., Trivedi, P.-B., and Undavia, N.-K., Ind. J Chem. 1998, vol. 37B, p.180.
Hazarika, J. and Kataky, J.C.-S., Indian. J. Heterocycl. Chem., 1998, vol. 7, p. 83.
Liszkiewicz, H., Kowalska, M.-W., Wietrzyk, J., and Opolski, A., Ind. J. Chem. 2003, vol. 42B, p. 2846.
Mallick, S.-K., Martin, A.-R., and Lingard, R.-G., J. Med. Chem., 1971, vol. 14(6), p. 528.
Freddy, H., Havaldar Sushil, K., and Mishra, J., Ind. J. Heterocycl. Chem. 2004, vol. 13, p. 197
Saundane, A.-R., Vaijinath, A.-V., and Vijaykumar, K., Heterocyclic. Lett. 2012, vol. 2(3), p. 333.
Sies, H., Am. J. Med. 1991, vol. 91(3C), p. 31S. doi https://doi.org/10.1016/0002-9343(91)90281-2
Stocker, R., Curr. Opi. Lip. 1999, vol. 10(6), p. 589.
Saundane, A.-R., Vaijinath, A.-V., and Vijaykumar, K., Med. Chem. Res., 2013,vol. 22, p. 3787.
Hiremath, S.-P., Biradar, J.-S., and Purohit, M.-G., Ind. J. Chem B. 1982, vol. 21, p. 249.
Indian Pharmacopoeia, Government of India, New Delhi Appendix IV, 3rd ed. 1985, vol. 90
Janovska, D., Kubikova, K., and Kokoska, L., Czech. J. Food. Sci. 2003, vol. 21, p. 107.
Bishnu, J., Sunil, L., and Anuja, S., J. Sci. Eng and Tech., 2009, vol. 5, p. 143.
Hatano, H., Kanawa, T., and Yasuhara, O., Chem. Pharm. Bull. 1988, vol. 36, p. 2090.
Oyaizu, M., Jap. J. Nutr. 1986, vol. 44(6), p. 307.
Dinis, T. C.-P., Madeira, V. M.-C., and Almeida, L.-M., Arch. Biochem and Biophys. 1994, vol. 315 (1), p. 161.
Author information
Authors and Affiliations
Corresponding author
Additional information
The text was submitted by the author in English.
Rights and permissions
About this article
Cite this article
Verma, V.A. Synthesis, Antimicrobial, and Antioxidant Studies of Some New Indolo[3,2-c]isoquinoline Derivatives. Russ J Gen Chem 88, 2628–2645 (2018). https://doi.org/10.1134/S1070363218120265
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363218120265