Abstract
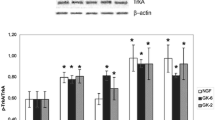
Previously, we prepared dimeric dipeptide mimetics of the first and the fourth loops of the nerve growth factor (NGF): hexamethylenediamides of bis(N-aminocaproyl-glycyl-L-lysine) (GK-6) and bis(N-monosuccinyl-L-glutamyl-L-lysine) (GK-2). Both mimetics activated TrkA-receptors, but induced different postreceptor signal pathways. GK-2 selectively activated PI3K/AKT, whereas GK-6 activated both PI3K/AKT and MAPK/ERK. Both mimetics exhibited a neuroprotective activity. In this study, we continued the investigation of a contribution of separate loop-like structures in the NGF functions and created and studied dimeric dipeptide mimetics based on a beta-turn of the NGF third loop: hexamethylenediamides of bis(N-gamma-hydroxybutyryl-L-lysyl-L-histidine) (GTS-115) and bis(N-acetyl-L-lysyl-L-histidine) (GTS-113). GTS-115 was shown to exhibit the neuroprotective activity in the concentration range from 10–5 to 10–7 М towards the HT-22 cell culture under the conditions of oxidative stress. The acetyl-containing GTS-113 mimetic proved to be inactive. GTS-115 (1 mg/kg/day intraperitoneally, for 7 days, the administration was started 4 h after the operation) exhibited the neuroprotective properties and decreased the infarction volume by 25% on the model of a stroke that was induced by a transient occlusion of the medial cerebral artery of rats. The action mechanism of GTS-115 was studied by Western-blot analysis and this mimetic in a concentration of 10–6 М was shown to activate the TrkA-receptor and both MAPK/ERK and PI3K/AKT basic postreceptor signal pathways. The inhibitory analysis revealed different contributions of these pathways into the GTS-115 neuroprotective effect. The LY294002 selective inhibitor of PI3K completely blocked the neuroprotective effect of GTS-115 in vitro, whereas the PD98059 specific inhibitor of MEK1 and MEK2 decreased this effect only by 10–15%. GTS-115 peptide stimulated a differentiation of the PC12 cells and caused a hyperalgesia in rats. These facts were in a good agreement with the literature data on the participation of the MAP-kinase pathway in these effects. Thus, the third NGF loop and the neighboring first NGF loop activated the postreceptor pathways in a similar way and exhibited the similar activities.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- BuONO:
-
n-butyl nitrite
- DCС:
-
1,3-dicyclohexylcarbodiimide
- DCU:
-
1,3-dicyclohexylurea
- DIEA:
-
N,N-diisopropylethylamine
- DMAPA:
-
3-(dimethylamino)propylamine
- GHB:
-
gamma–hydroxybutyric acid
- MAPK/ERK:
-
the mitogen-activated proteinkinase pathway
- NGF:
-
the nerve growth factor
- PI3K/AKT:
-
phosphatidyl inositol-3-kinase pathway
- TEA:
-
triethylamine
- TFA:
-
trifluoroacetic acid
References
Levi-Montalcini, R., Science, 1987, vol. 237, pp. 1154–1162.
Aloe, L., Rocco, M.L., Bianchi, P., and Manni, L., J. Transl. Med., 2012, vol. 10, no. 11, pp. 239–252.
Kaplan, D.R. and Miller, F.D., Curr. Opin. Neurobiol., 2000, vol. 10, no. 3, pp. 381–391.
Obata, K. and Noguchi, K., Life Sci., 2004, vol. 74, no. 21, pp. 2643–5263.
Gudasheva, T.A., Antipova, T.A., and Seredenin, S.B., Dokl. Biochem. Biophys., 2010, vol. 434, pp. 262–265.
Seredenin, S.B. and Gudasheva, T.A., Dipeptide mimetics of neurotrophins NGF and BDNF, RF Patent no. 2410392, Byul. Izobret., 2011, no. 3.
Gudasheva, T.A., Povarnina, P.Yu., Antipova, T.A., and Seredenin, S.B., Neurosci. Med., 2014, vol. 5, no. 2, pp. 101–108.
Seredenin, S.B. and Gudasheva, T.A., Zh. Nevrol. Psikhiatr. im. S.S. Korsakova, 2015, vol. 115, no. 6, pp. 63–70.
Gudasheva, T.A., Povarnina, P.Y., Antipova, T.A., Firsova, Y.N., Konstantinopolsky, M.A., and Seredenin, S.B., J. Biomed. Sci., 2015, vol. 22, p. 106.
Jackson, G.R., Werrbach-Perez, K., Ezell, E.L., Post, J.F., and Perez-Polo, J.R., Brain Res., 1992, vol. 592, nos. 1–2, pp. 239–248.
Virdee, K. and Tolkovsky, A.M., J. Neurochem., 1996, vol. 67, no. 5, pp. 1801–1805.
Chang, J.H., Mellon, E., Schanen, N.C., and Twiss, J.L., J. Biol. Chem., 2003, vol. 278, no. 44, pp. 42877–42885.
Longa, E.Z., Weinstein, P.R., Carlson, S., and Cummins, R., Stroke, 1989, vol. 20, no. 1, pp. 84–91.
De Ryck, M., Van Reempts, J., Borgers, M., Wauquier, A., and Janssen, P.A., Stroke, 1989, vol. 20, no. 10, pp. 1383–1390.
Jolkkonen, J., Puurunen, K., Rantakomi, S., Harkonen, A., Haapalinna, A., and Sivenius, J., Eur. J. Pharmacol., 2000, vol. 400, nos. 2–3, pp. 211–219.
Hathway, G.J. and Fitzgerald, M., J. Pain, 2006, vol. 7, pp. 57–61.
Taglialatela, G., Foreman, P.J., and Perez-Polo, J.R., Int. J. Dev. Neurosci., 1997, vol. 15, no. 6, pp. 703–710.
Khan, S.A., Chatterjee, S.S., and Kumar, V., Life Sci., 2016, vol. 148, pp. 53–62.
Tarasyuk, A.V., Pomogaibo, S.V., Kurilov, D.V., and Gudasheva, T.A., Pharm. Chem. J., 2013, no. 1, pp. 20–27.
Jaouadi, M., Martinez, J., and Castro, B., Org. Chem., 1987, vol. 52, pp. 2364–2367.
Ueda, Y., Walsh, E., Nakanishi, H., and Yoshida, K., Neurosci. Lett., 1994, vol. 165, nos 1–2, pp. 203–207.
Alam, A., Anal. Biochem., 1992, vol. 203, no. 1, pp. 121–126.
Towbin, H., Staehelin, T., and Gordon, J., Proc. Natl. Acad. Sci. U. S. A., 1979, vol. 76, no. 9, pp. 4350–4354.
Kukhtina, V.V., Tsetlin, V.I., Utkin, Y.N., Inozemtseva, L.S., and Grivennikov, I.A., J. Nat. Toxins, 2001, vol. 10, no. 1, pp. 9–16.
Brown, R.F., Hui, K.Y., Matter, W.F., and Vlahos, C.J., J. Biol. Chem., 1994, vol. 269, no. 7, pp. 5241–5248.
Alessi, D.R., Cuenda, A., Cohen, P., Dudley, D.T., and Saltiel, A.R., J. Biol. Chem., 1995, vol. 270, no. 46, pp. 27489–27494.
Siren, A.L., Fratelli, M., Brines, M., Goemans, C., Casagrande, S., Lewczuk, P., et al., Proc. Natl. Acad. Sci. U. S. A., 2001, vol. 98, no. 7, pp. 4044–4049.
Hathway, G.J. and Fitzgerald, M., J. Pain, 2006, vol. 7, no. 1, pp. 57–61.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © T.A. Gudasheva, A.V. Tarasiuk, N.M. Sazonova, S.V. Pomogaibo, A.N. Shumskiy, I.O. Logvinov, S.V. Nikolaev, P.Yu. Povarnina, M.A. Konstantinopolsky, T.A. Antipova, S.B. Seredenin, 2017, published in Bioorganicheskaya Khimiya, 2017, Vol. 43, No. 3, pp. 236–249.
Rights and permissions
About this article
Cite this article
Gudasheva, T.A., Tarasiuk, A.V., Sazonova, N.M. et al. Design, synthesis, and neuroprotective effects of a dimeric dipeptide mimetic of the third loop of the nerve growth factor. Russ J Bioorg Chem 43, 235–247 (2017). https://doi.org/10.1134/S1068162017030050
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1068162017030050