Abstract
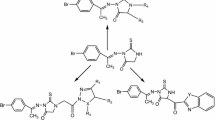
A novel series of thiourea, carbamimidothioic acid, 4, 5-dihydrooxazole-2-thiol, oxazolidine-2thine, and 2-amino-1-phenylpropyl-2-chloroacetate derivatives was designed and synthesized using 2-amino-1-phenylpropan-1-ol (L-norephedrine) as a strategic starting material. The structures of the newly synthesized compounds were established by elemental analyses, IR, and 1H NMR and 13C NMR spectral data. The compounds were evaluated for their in vitro anticancer activity against various cancer cell lines. The corresponding acetamide, carbamimidothioic acid, and 2-2-amino-1-phenylpropyl-2-chloroacetate derivatives showed almost the same activity as the standard drug doxorubicin against human breast cancer cell line (MCF-7). Also, the acetamide and 2-thioxoimidazolidin-4-one derivatives exhibited higher activity than the reference drug doxorubicin against human colon cancer cell line (HCT 116).
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Gupta, S.P., Chem. Rev., 1994, vol. 94, pp. 1507–1551.
Keri, G. and Ullrich, A., Cancer as a communication disorder, in Molecular Pathomechanisms and New Trends in Drug Research, Keri, G. and Toth, I., Eds., London: Taylor and Francis, 2003, pp. 227–248.
Vega-Pérez, J.M., Periñán, I., Argandoña, M., Vega-Holm, M., Palo-Nieto, C., Burgos-Morón, E., López-Lázaro, M., Vargas, C., Nieto, J.J., and Iglesias-Guerra, F., Eur. J. Med. Chem., 2012, vol. 58, pp. 591–612.
Yao, J., Chen, J., He, Z., Sun, W., and Xu, W., Bioorg. Med. Chem., 2012, vol. 20, pp. 2923–2929.
Shantharam, C.S., Suyoga, V.D.M., Suhas, R., Sridhara, M.B., and Channe, G.D., Eur. J. Med. Chem., 2013, vol. 60, pp. 325–332.
Yang, W., Hu, Y., Yang, Y.S., Zhang, F., Zhang, Y.B., Wang, X.L., Tang, J.F., Zhong, W.Q., and Zhu, H.L., Bioorg. Med. Chem., 2013, vol. 21, pp. 1050–1063.
Keche, A.P., Hatnapure, G.D., Tale, R.H., Rodge, A.H., and Kamble, V.M., Bioorg. Med. Chem., 2012, vol. 22, pp. 6611–6615.
Burgeson, J.R., Moore, A.L., Boutilier, J.K., Cerruti, N.R., Gharaibeh, D.N., Lovejoy, C.E., Amberg, S.M., Hruby, D.E., Tyavanagimatt, S.R., Allen, R.D., and Daisar, D., Bioorg. Med. Chem. Lett., 2012, vol. 22, pp. 4263–4272.
Kazimierczuk, Z., Chalimoniuk, M., Laudy, A.E., Moo-Puc, R., Cedillo-Rivera, R., Starosciak, B.J., and Chrapusta, S.J., Arch. Pharm. Res., 2010, vol. 33, pp. 821–830.
Al-Dosari, M.S., Ghorab, M.M., Alsaid, M.S., Nissan, Y.M., and Ahmed, A.B., Eur. J. Med. Chem., 2013, vol. 16, pp. 373–383.
Fang-Bin, H., Ze-Mei, G., Tie-Ming, Ch., and RunTao, L., SYNLETT, 2009, vol. 4, pp. 0648–0650.
Kumar, V., Kumar, A., Sureshbabu, V.V., and Chimni, S.S., Med. Chem., 2014, vol. 14, pp. 910–920.
Sen-Lin, W., Yu-Jun, S., Hai-Bing, H., Yu, L., Yang, L., and Hong, D., Chinese Chem. Lett., 2015, vol. 26, pp. 672–674.
Tian, Y., Jin, J., Wang, X., Hu, J., Xiao, Q., Zhou, W., Chen, X., and Yin, D., Eur. J. Med. Chem., 2014, vol. 85, pp. 1–15.
Martins, P., Jesus, J., Santos, S., Raposo, L.R., RomaRodrigues, C., Viana Baptista, P., and Fernandes, A.R., Molecules, 2015, vol. 20, pp. 16852–16891.
Kumar, V., Mudgal, M.M., Rani, N., Jha, A., Jaggi, M., Singh, A.T., Sanna, V.K., Singh, P., Sharma, P.K., Irchhaiya, R., and Burman, A.C., J. Enzy. Inhib. Med. Chem., 2009, vol. 24, pp. 763–770.
Rodriguez-Fernandez, E., Manzano, J.L., Benito, J.J., Hermosa, R., Monte, E., and Criado, J.J., J. Inorg. Biochem., 2005, vol. 99, pp. 1558–1572.
Rauf, M.K., Ebihara, M., and Imtiaz-ud-Din, B.A., Acta. Cryst. Sect. E Online, 2012, vol. 68, p. o119.
Koca, I., Özgür, A., Coşkun K.A., and Tutar, Y., Bioorg. Med. Chem., 2013, vol. 21, pp. 3859–3865.
Lv, P.C., Li, H., Sun, J., Zhou, Y., and Zhu, H., Bioorg. Med. Chem., 2010, vol. 18, pp. 4606–4614.
Crank, G., Neville, M., and Ryden, R., J. Med. Chem., 1973, vol. 16, pp. 1402–1405.
Jianwen, Y., Zuopeng, H., Jing, C., Daquan, C., Wei, S., and Wenfang, X., Chin. J. Chem., 2012, vol. 30, pp. 2423–2430.
Skehan, P., Storeng, R., Scudiero, D., Monks, A., McMahon, J., Vistica, D., Warren, J.T., Bokesch, H., Kenney, S., and Boyd, M.R., J. Natl. Cancer Inst., 1990, vol. 82, pp. 1107–1112.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Abdel-Kader, M.S., Ghorab, M.M., Alsaid, M.S. et al. Design, synthesis, and anticancer evaluation of some novel thiourea, carbamimidothioic acid, oxazole, oxazolidine, and 2-amino-1-phenylpropyl-2-chloroacetate derived from L-norephedrine. Russ J Bioorg Chem 42, 434–440 (2016). https://doi.org/10.1134/S1068162016040026
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1068162016040026