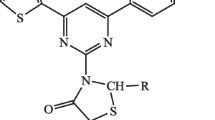
Abstract
5-Phenyl-2-[(3,4,5-trimethoxybenzylidene)hydrazino]-thiazole and 3-[(3,4,5-trimethoxybenzylidene)amino]-4-oxoimidazolidin-2-thione were prepared by cyclization of 1-[(3,4,5-trimethoxybenzyliden)amino]-thiourea with phenacyl bromide and ethyl chloroacetate in the presence of fused sodium acetate. Acetylation of the synthesized compounds with acetic anhydride gave corresponding N-acetyl derivatives. Condensation of the synthesized thione with aromatic aldehydes yielded two 3-substituted 5-arylidene-4oxo-imidazolidin-2-thiones. Acetylation of the latter compounds with acetic anhydride afforded the corresponding N-acetyl-4-oxo-imidazolidin-2-thiones. Some of the synthesized compounds exhibited antimicrobial activity. The cytotoxic activity of the prepared thiazole and imidazolidin-2-thione derivatives was studied on several tumor cell lines.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Wilson, L.J., Li, M., and Portlock, D.E., Tetrahedron Lett., 1998, vol. 39, no. 29, pp. 5135–5138.
Belai, I., Tetrahedron Lett., 2003, vol. 44, no. 40, pp. 7475–7477.
Brouilette, W.J., Jestkov, V.P., Brown, M.L., Akhtar,M.S., Delorey, T.M., and Brawn, G.B., J. Med. Chem., 1994, vol. 37, pp. 3289–3293.
Erik, C.S., Ottosen, R., Petersen, H.J., Bjorking, F., Latini, S., Hjarnaa, V., Bramm, E., and Binderup, L., Bioorg. Med. Chem. Lett., 1997, vol. 7, pp. 3095–3100.
Hjarnaa, P.J., Jonsson, E., Latini, S., Dhar, S., Larsson, R., Bramm, E., Skov, T., and Binderup, L., Cancer Res., 1999, vol. 59, pp. 5751–5757.
Olsen, L.S., Hijamaa, P.J., Latini, S., Holm, P.K., Larsson, R., Bramm, E., Binderup, I., and Madsen, M.W., Inc. Cancer, 2004, vol. 3, pp. 98–105.
Mader, M.M., Curr. Opin. Drug Discov. Devel., 2005, vol. 8, pp. 613–618.
Ibrahim, H.K., Hasanen, J.A., Zein, M.A., and ElDeen, I.M., Mens Agitat., 2008, vol. 3, pp. 59–72.
Aboubshit, S.A. and Aboubshit, H.A., J. Chem. Pharm. Res., 2012, vol. 4(1), pp. 568–579.
Abd El-Moneim, M., IOSR JAC, 2014, vol. 7, no. 1, pp. 67–76.
El-Sakka, S., Mens Agitat., 2008, vol. 2, pp. 57–68.
Schelz, Z., Molnar, J., and Hohmann, J., Fitoterapia, 2006, vol. 77, no. 4, pp. 279–285.
National Committee for Clinical Laboratory Standards. Performance Standards for Antimicrobial Susceptibility Testing, Informational Supplement M100. S13, 12th ed., NCCLS: Wayne, PA,USA, 2003.
Gillespie, S.H., Antimicrobial Susceptibility in Medical Microbiology, Illustrated, London: Butterworth Heinemann, 1994, pp. 234–247.
Chatterjee, S. and Das, S.N., Ind. J. Pharmacol., 1996, vol. 28, pp. 116–119.
Mosmann, T., J. Immunol. Methods, 1983, vol. 65, pp. 55–63.
Vijayen, P., Raghu, C., Ashok, G., Dhanaraj, S.A., and Suresh, B., Indian J. Med. Res., 2004, vol. 120, no. 1, pp. 9–24.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
El-Shenawy, A.I. Synthesis and in vitro antimicrobial and antitumor activity of some nitrogen heterocycles. Russ J Bioorg Chem 42, 100–105 (2016). https://doi.org/10.1134/S1068162016010040
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1068162016010040