Abstract
The root soil interaction affects the fractionation and bioavailability of nutrients in the rhizosphere and, in turn, the uptake and accumulation of nutrients by plants. In this study, a greenhouse experiment using Thomson Navel Orange (Citrus sinensis (L.) Osbeck.) grafted on three rootstocks [Swingle Citrumelo (SC), Sour Orange (SO), and Troyer Citrange (TC)] was conducted in mildly acidic soil. Eighteen months after transplanting in the pots, the i) P acquisition efficiency (PAE) in these plants; ii) rhizosphere effects on some biological characteristics, including dissolved organic Carbon (DOC), fungi and bacteria populations, microbial biomass P (MBP), and alkaline (AlP) phosphatase, and iii) the fractions of P in the rhizosphere soils were determined. Results of the study showed that the TC and SO promoted scion P uptake and increased PAE. The rhizospheric condition of SO rootstock induced significant promotion (P < 0.05) in all biological properties compared to other rootstocks. Living roots of SO markedly modified biological properties in the rhizosphere soil and therefore affected P bioavailability. Moreover, the rhizosphere effects on the chemical fractions of P were under rootstock control. The lowest organic P was measured in SO-grafting, whereas the lowest amounts of exchangeable P and residual P in the rhizosphere soils were measured in TC- grafting. The contents of P associated with Fe and Al compounds were lower in the SO and TC rhizosphere than in the SC rhizosphere soil. This study provides insights into the adaptive mechanisms of Thomson Navel Orange grafted on three rootstocks to phosphorus deficiency.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
INTRODUCTION
Thomson Navel Orange trees (Citrus sinensis (L.) Osbeck.) are widely cultivated in many countries, and Iran is the ninth-largest orange producer in the world [10]. Citrus cultivars are grafted on the rootstocks of other citrus species and new hybrids [24]. Citrus rootstocks have pivotal roles in nutrient and water uptake and translocations because they develop the root systems of these trees [11]. Moreover, rootstocks can affect citrus growth, yield, fruit quality [11, 15, 21] and tolerance to different biotic and abiotic stresses [24].
Phosphorus (P) contributes in energy storage, root development, and early maturity of crops which was explained in previous studies [18]. Despite the relatively high amount of total-P in most agricultural soils, it is the least accessible element for plants because of its low availability and poor recovery from the applied fertilizers [4]. The development of agriculture has been restricted by the low availability of P. Recently, increased fertilizer input has led not only to increase yields but also to a series of environmental problems such as groundwater pollution and eutrophication of surface waters [30]. Thus, understanding how to minimize the inefficient use of P-inputs to ensure the sustainable development of agriculture has become an international focus. To this end, screening of the genotypes with high efficiency of P acquisition has been considered as a cost-effective and environmentally-friendly technology to minimize inefficient use of P-inputs and environmental pollution [5].
In low-P environments, more P-efficient genotypes can take up more P from the soil and transport it to the shoot (uptake efficiency) [37] and thus can reduce mineral P fertilizer input requirement in agricultural production. Some plants developed several adaptive strategies to maximize the acquisition of P, particularly when resources are limiting [7]. More P‑efficient plant species can increase the bioavailability of P in the soil by exuding: (1) protons into the rhizosphere, which solubilize less soluble forms of soil P by lowering the rhizosphere soil pH [12] (2) large quantities of carboxylates to aid mobilization unavailable soil Ca, Fe and Al phosphates [22], (3) acid phosphatase that mineralizes organic P [27] (4) and or can alter root characteristics under P deficiency condition (e.g. rate of growth, specific root length, and density and length of root hairs) [7]. Therefore, an increased plant capacity to efficiently use the poorly available pools will be helpful in promoting sustainable development of agriculture [28].
In addition, the roots can secrete 5–21% of all photosynthetically fixed carbon into the rhizosphere soil through root exudates [18], which can supply the important energy source for rhizosphere microorganisms [35]. Microorganisms can make a significant contribution to nutrient availability, turnover and retention [26]. Therefore, the dynamics and availability of P in soil are controlled by a combination of biological processes (mineralization–immobilization) and chemical processes (adsorption–desorption and dissolution–precipitation).
Plant roots affect rhizospheric reaction and, in turn, the fractionation and bioavailability of nutrients in the rhizosphere [19]. The genetic potential of the rootstocks for nutrient acquire plays a pivotal role in the nutrient efficiency of grafted plants. Hence, the key to the success of citrus production is finding a suitable rootstock. To achieve a sustainable citrus production, screening of citrus rootstocks for high P-efficiency is important to improve P utilization through better matching of plant demand with supply. The most important rootstock resources in the world and especially in northern Iran include Swingle Citrumelo, Sour orange, and Troyer Citrange. However, studies on the adaptive mechanism of the perennial woody plants to P deficiency are still limited. Therefore, this work aimed to gain better insight into how various citrus rootstocks can acquire soil P and/or alter the availability of P in their root surroundings soil. In the present study, we focused on evaluating the P acquisition efficiency (PAE) as a relative measurement of the capacity of Thomson Navel Orange (Citrus sinensis L. Osbeck.) plants grafted on three rootstocks for acquiring and transporting P to the scion part and the adaptive mechanisms developed by citrus rootstocks to improve their efficiency in acquiring P from a mildly acidic soil.
MATERIAL AND METHODS
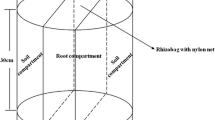
Soil materials. The soil used in this experiment was collected from one citrus orchard (0–60 cm horizon) located in the east of Mazandaran province (Khoramabad), in northern Iran (36°46′ N, 50°52′ E). After air-drying, the soil sample was sieved through a 2-mm sieve to determine some of its physicochemical properties (Table 1). The studied soil was mildly acidic with sandy clay loam texture and low in initial available–P.
Plant material and growth conditions. In this study, the three rootstocks employed for Thomson Navel Orange (Citrus sinensis (L.) Osbeck) were Swingle citrumelo (SC) (Citrus paradisi Macf.×Poncirus trifoliata (L.) Raf.), Sour orange (SO) (Citrus aurantium L.), and Troy er citrange (TC) (Citrus sinensis (L.) Osbeck.× Poncirus trifoliata (L.) Raf.). In budding time, year-old uniform seedlings from SC, SO and TC rootstocks were budded via T-budding technique using the scions of Thomson Navel Orange in September 2016. Disease-free scion and rootstocks were obtained from research greenhouse of Citrus and Subtropical Fruits Research Center of Ramsar city (36°54′ N, 50°39′ E), Mazandaran Province, Iran. The seedlings (various rootstock-scion combinations) were initially grown for 7 months at the same nutrient medium, and then transplanted into plastic pots containing 15 kg of the soil.
To ensure a sufficient supply of mineral nutrients, chemical fertilizers at the rates of 500 mg K kg–1 soil (K2SO4), 500 mg N kg–1 soil (urea), 10 mg Fe kg–1 soil (Fe-EDDHA), and 5 mg Zn kg–1 soil (ZnSO4) were mixed with the soil thoroughly in each 15 kg-holding pots before transplanting. Then, uniform Thomson Navel Orange were transplanted on SO or SC, and or TC rootstocks (April 2017), one per pot. Seedlings were grown for approximately 18 months in a greenhouse with natural light, a day/night temperature of 30/18 ± 3°C. Plant irrigation was daily performed to field capacity.
Plant harvest and measurements. After about 18 months from the transplanting date, in October 2018, the plants were harvested. Then, samples were divided into scion part (plant parts above the graft junction) and rootstock part (plant parts below the graft junction). The fresh materials were oven-dried at 70°C for 72 h, and then weighted. The dry ashing method was used for P analysis in all of the dried plant samples [14]. The P concentration was determined by colorimetric analysis by the ascorbic acid method [20]. The plant indices including dry weight (DW), and P uptake were measured. In addition, PAE (Efficiency of P absorption and transport to the shoot) was calculated based on the method proposed by Zambrosi et al. [37], with some modifications [28].
Soil chemical and biological properties. At the end of the experiment (18 months after planting), soil adhering tightly to the root surface was recognized as the rhizosphere soils [34]. The soil samples were then sieved through a 2 mm mesh sieve and divided into two portions. One portion of the soil, used for the analysis of concentrations of dissolved organic C (DOC) [2, 8], fungi and bacteria populations [36], microbial biomass P (MBP) [6], alkaline phosphatase (AlP) [33] and acid phosphatase (AcP) [9] activities was stored in a fridge at 4°C, and the other portion was air-dried for analysis of the P-organic, and the various fractions of P in soil. The ignition method was used to determine the organic P content [16].
Phosphorus Fractionation in soil. Soil P fractions were sequentially extracted using a modified version of Hedley’s method [1, 3]. The phosphorus was partitioned into four fractions: (1) exchangeable (EXCH-P: 2 M KCl for 2 h); (2) bound to Fe-Al oxides (Fe-Al-P: 0.1 M NaOH for 17 h); (3) bound to carbonates (CARB-P: 0.5 M HCl for 24 h); and (4) residual (RES-P: HNO3–HClO4 mixture). The steps were separated through decantation of the sample supernatant after centrifugation at 5.000 rpm for 30 min. Finally, P concentrations in the extracts were determined by colorimetric analysis using Murphy and Riley’s [20] method.
In the studied soil prior to plant growth, the initial contents of P in various fractions including EXCH-P, Fe-Al-P, CARB-P, organic-P, and RES-P were equal to 8.0, 78, 543, 136 and 205 mg kg–1, respectively. In addition, the change percentage of various P fractions in the rhizosphere soil of plants grown for 18 months with reference to P fractions of the original soil (before growing plant) was calculated for each P fractions using the following equation:
Data analysis. The experiment was set up in a completely randomized design with three replications. Statistical analyses of the data were performed by one-way ANOVA using the SAS software package. Means were compared using Duncan’s post-hoc test (significant at P ≤ 0.05). In addition, data were assessed by principal component analysis (PCA) using Minitab software (version 17.0).
RESULTS AND DISCUSSION
Phosphorus efficiency acquisition (PAE). The growth of Thomson Navel Orange (g DW/plant) is affected by the rootstock-scion combinations (Table 2). The scion DW produced by TC and SO rootstocks was, respectively, about 1.6 and 1.4 times more than SC under low-P conditions. However, the highest rootstock biomass was measured in SC and TC-grafting.
Respectively, about 35, 57 and 61% of the total amount of P absorbed in SC, SO, and TC-grafting, was allocated to the scion part. This suggests that the SO and TC rootstocks have preferentially allocated adsorbed P to the scion part compared to the rootstock part.
PAE is a relative index for the capacity of a plant to acquire and transport of P to the scion part. The rootstocks showed significant differences in the PAE. The PAE of the Thomson scions grafted on the TC and SO rootstocks, increased 1.8- and 2.0-fold respectively, compared with the scion grafted on the SC rootstock.
Generally, the results showed that the status of P uptake, and thus PAE of Thomson scion was improved by SO- and TC-grafting compared to SC-grafting. Some previous studies have reported that grafting combinations and rootstock types influence plant growth and nutrient uptake [15, 37]. Kumar et al. [15] found that P absorption efficiency was higher on Sour orange than rough lemon, Carrizo and Troyer citrange, and Rangpur lime. In other study, it was reported that P acquisition efficiency of Sweet orange scion is improved by Rangpur lime-grafting in compared to Cleopatra mandarin-grafting [37]. In this study, different tolerance levels of the citrus rootstocks to P deprivation (different PAE) may be associated with the various strategies of each rootstock for obtaining adequate P for their growth under P limiting conditions.
Soil chemical and biological properties. Scions grafted on TC showed lower DOC content in the rhizosphere than those grafted on SO and SC (Fig. 1). In contrast, pH in the rhizosphere soils was not significantly affected by the rootstock.
Bacteria population in the rhizosphere soils varied from 6.9 × 103 (TC) to 14.0 × 103 g–1 soil (SO). Furthermore, the results showed that fungi count has the highest populations in the SO rhizosphere (2.0 × 102 g–1 soil) and the lowest population was recorded for the SC and TC (1.33 × 102 g–1 soil) rhizosphere soils (Fig. 2). In addition, The MBP contents ranged from 2.3 (TC) to 12.4 mg kg–1 (SO) (Fig. 3).
Effect of different citrus rootstocks on the rhizosphere fungal and bacterial population (CFU g–1 soil) in the rhizosphere soils. Bars with the different letter are significantly different (Duncan test, P ≤ 0.05) [the means were separately compared for Fungi and Bacteria in the different rootstocks].
Effect of different citrus rootstocks on the status of microbial biomass P (mg kg–1) and microbial biomass P to organic-P (%) in the rhizosphere soils. Bars with the different letter are significantly different (Duncan test, P ≤ 0.05) [the means were separately compared for microbial biomass P and microbial biomass P to organic-P in the different rootstocks].
In this study, the maximum and the minimum activity of AlP were obtained in the rhizosphere of the scions grafted on SO (401 PNP-P \({\text{g}}_{{{\text{soil}}}}^{{ - 1}}\))) and SC (250 PNP-P \({\text{g}}_{{{\text{soil}}}}^{{ - 1}}\))), respectively. In contrast, AcP activity in the rhizosphere soils was not significantly affected by the rootstocks (Fig. 4).
Effect of different citrus rootstocks on the activity of phosphatase enzymes (PNP-P \({\text{g}}_{{{\text{soil}}}}^{{ - 1}}\)) in the rhizosphere soils. Bars with the different letter are significantly different (Duncan test, P ≤ 0.05) [the means were separately compared for Acid phosphatase and Alkaline phosphatase in the different rootstocks].
In the rhizosphere, the mutual demand for P results in competition between plants and microorganisms; however, carbon availability strongly affects microbial competitiveness [17]. In this study, rootstock-mediated regulation of the quantity of DOC) can induce alternation in the microbial community’s structure associated with root. Indeed, it has been established that DOC in mineral soils originates largely from root exudates and root residues [13]. Comparison of DOC and microbial populations between the rhizosphere soils of the studied rootstocks showed that these traits significantly increased in the SO rhizosphere compared with the other rootstocks. According to Ngullie et al. [21] rootstocks mediate variation in microbial community composition of citrus rhizosphere. In the SO rhizosphere, due to readily available C resources, bacteria and fungi can rapidly mineralize P from soil organic matter and or directly uptake inorganic P and incorporate it into their biomass. Hence, MBP was also higher in SO rhizosphere compared with the other rootstocks. In addition, the highest MBP/PO ratio was observed in the rhizosphere soil of the scions on SO, while the lowest MBP/PO ratio was observed in the rhizosphere of SC and TC-grafting. The immobilization of soil P into the microbial biomass, while temporarily protecting the P from reacting with the soil and maintaining it in labile forms [23], can also be considered as a mechanism for regulating the supply of P in the solution phase [32]. In addition, directly uptake of inorganic P from the solution phase by microorganisms can alter sorption equilibria in SO rhizosphere that may result in an enhanced net transfer of orthophosphate ions into the soil solution. Therefore, it is obvious that microorganisms can enhance the capacity of the scions on SO rootstock to acquire P from sparingly available forms, through the mentioned mechanisms (Table 3).
In addition, plants and microorganisms can mineralize organic P by releasing various phosphatase enzymes [17]. Based on the results, AlP activity in the rhizosphere soil of SO-grafting was higher than the AlP activity in each of the rhizosphere soil of TC- and SC-grafting. The stimulation of microbial activity via the enhanced production of DOC in the SO rhizosphere probably results in the promotion of phosphatase activity in this soil. The decrease in organic P along with the increase in AlP activity in the SO-grafting (Fig. 5) suggest that AlP activity may be involved in regulating the P balance in this particular soil and organic P can be an important source for plant growth grafted on SO rootstock. It has been suggested that microorganisms can facilitate the mobility of organic P either directly or indirectly through microbial turnover and result in enhanced inorganic P in the soil solution [29]. Hence, an increase in microbial P mobilization capacity, increase in solubilizing and mineralization of P from sparingly available forms of soil inorganic and organic P are possible mechanisms by which SO-grafting might increase P acquisition efficiency from rhizosphere soil. According to previous studies, microbial biomass P can play an important role in the supply of plant-available P [26].
Principal component analysis (PCA) biplot of the biological and chemical attributes in the soil rhizosphere of three citrus rootstocks with different PAE. Image show score variations in these traits along the first two main component axes, with the percentage of variation explained [▲: Troyer Citrange; ◼: Swingle Citrumelo; ⚫: Sour Orange].
Generally, compared with the other rootstocks, it is clear that SO-grafting had the greatest impact on biological features in the rhizosphere of young orange plants in response to deficient P in this soil.
Phosphorus fractions. EXCH-P represents the most biologically available form of P. The lowest content of all chemical fractions belonged to the EXCH-P fraction. The concentration of EXCH-P varied between the rootstocks. The descending order of EXCH-P concentrations in the rhizosphere soils was as follows: SC-grafting (7.0 mg kg–1) > SO-grafting (6.0 mg kg–1) > TC-grafting (5.3 mg kg–1). Among the studied rootstock-scion combinations, the highest EX-P depletion in the rhizosphere soil and the highest P accumulation in the scion part were found for TC-grafting. Although plants can only uptake the available P, solid phase soil P (other P fractions) can also be transformed to available P due to plant growth [25].
For all the rootstocks, Fe–Al–P had the second lowest content among the chemical fractions. The Fe-Al-P concentration was lower for TC and SO-grafting (45 and 46 mg kg–1, respectively) than the SC-grafting (53 mg kg–1). In addition, CARB-P ranged from 461 to 488 mg kg–1, with an average of 473 mg kg–1. There was a significant difference (P < 0.05) in the CARB-P concentration between the rootstocks. CARB-P was the most abundant P form in this soil.
Organic-P content in the rhizosphere soils varied from 96 to 126 mg kg–1 soil. Furthermore, the results showed that the lowest organic-P was measured in the rhizosphere soil of the scions grafted on SO. The ratio of MBP to PO in the rhizosphere soils varied from 1.9–13% (data not shown). The highest MBP/PO ratio was observed in the rhizosphere soil of the scions grafted on SO, while the lowest MBP/PO ratio was measured in the rhizosphere of SC- and TC-grafting. RES-P also ranged from 141 to 195 mg kg–1, with the average of 173 mg kg–1. The lowest concentration of the RES-P was found in the rhizosphere of TC-grafting (Table 3).
Generally, compared to SC-grafting, SO, and TC-grafting resulted in lower levels of Fe-Al-P in the rhizosphere soil. In addition, the lowest CARB-P and RES-P contents were measured in the rhizosphere soil of TC rootstock. It is concluded that the decrease of RES-P concentration in the TC-grafting may be due to its transformation into other P forms in the rhizosphere soil As was stated before, the pH was lower for TC rootstock compared to other rootstocks; although this difference was not significant. Indeed, the greater involvement of H+ ions in the dissolution of calcium carbonates in the TC rhizosphere could be one of the main reasons for not observing the pH reduction in the TC rhizosphere. The pH buffering capacity is a chemical parameter that plays a key role in root induced pH changes in the rhizosphere, as stressed by Schaller [31]. Moreover, the results showed that TC-grafting resulted in lower levels of DOC in the rhizosphere soil than SO-grafting, implying that compared to SO-grafting, scions grafted on TC allocated lower amounts of the photo-assimilated carbon to root exudates. Furthermore, plants on this rootstock seem to avoid direct competition with micro-organisms by reducing the amount of DOC that they release into the rhizosphere. In addition, root dry weight in TC- grafting was higher than in SO-grafting. Hence, plants on TC rootstock can escape microbial competition in the rhizosphere by increasing the volume of soil that is explored by roots and P mobilization of poorly available pools in this soil. The mentioned strategies are considered as vital mechanisms for P acquisition by scions grafted on TC in this soil.
Principal component analysis (PCA). Based on the multi-correlation analysis of 12 biological and chemical attributes in the rhizosphere soils, we performed principal component analysis (PCA) to determine the biological and chemical attributes in the soil rhizosphere responding more to P deficiency among citrus rootstocks. The first two main components (PC1 and PC2) explained 69.0% of the total data variation for the citrus rootstocks with different PAE. It was observed that the variables bacterial pollution, DOC, Olsen-P, pH, MBP made a higher contribution to PC1. Furthermore, it was observed that the variables organic-P, CARB-P, EXCH-P positively and the variables AlP negatively made a higher contribution to PC2 (Fig. 5). PCA can fully reflect various strategies of SO and TC rootstocks to obtain adequate P for their growth under P limiting conditions. Based on the PCA, TC improves PAE mainly by modifying chemical reactions in its root surroundings soil and altering P fractionation in its rhizosphere soil. However, SO improves PAE mainly by inducing promotion in all biological properties, and mineralization of organic P. Therefore, various mechanisms (modification of biological and chemical properties in the rhizosphere) used for P acquisition by each rootstock can lead to the different PAE of the rootstocks. Collectively, selecting the rootstocks with high efficiency of P acquisition can minimize inefficient use of P-inputs and environmental pollution.
CONCLUSIONS
This study showed some biological features in the rhizosphere soil after cultivating with various rootstock-scion combinations, including Thomson Navel grafted on rootstocks of Swingle citrumelo, Sour orange and Troyer citrange, as well as the P fractionation in the rhizosphere. Collectively, the results indicated that SO root-induced the greatest changes in the biological features in the rhizosphere. The MBP, fungal and bacterial population, DOC, AlP in the SO rhizosphere soils were greater than the SC and TC rhizosphere soils (P < 0.05). The obvious changes in the P fractions in the soil were caused by the root activities of rootstocks. The lowest organic P was measured in SO-grafting, whereas the lowest amount of exchangeable P and residual P in the rhizosphere soils was measured in TC- grafting. The contents of P associated with Fe and Al compounds were lower in the SO and TC rhizosphere compared to the SC rhizosphere. Furthermore, the results of this study showed that the P acquisition efficiency of Thomson Navel scion was improved by TC- and SO-grafting in this soil. Hence, mechanisms such as increasing of microbial P mobilization capacity, mineralization of organic P, and uptake of sparingly available pools (in SO-grafting) or escaping microbial competition by decreasing the root exudates amount, and increasing P mobilization of poorly available pools (in the TC-grafting) were chief strategies mediated by SO and TC rootstocks for P acquisition in this soil.
REFERENCES
J. Alvarez-Rogel, F. J. Jimenez-Carceles, and C. Egea-Nicolas, “Phosphorus retention in a coastal salt marsh in SE Spain,” Sci. Total Environ. 378, 71–74 (2007).
J. M. Anderson and J. S. I. Ingram, Tropical Soil Biology and Fertility: A Handbook of Methods (CAB Int., Wallingford, 1994).
Y. Ann, K. R. Reddy, and J. J. Delfino, “Influence of chemical amendments on phosphorus immobilization in soils from a constructed wetland,” Ecol. Eng. 14, 157–167 (2000).
T. Balemi and K. Negisho, “Management of soil phosphorus and plant adaptation mechanisms to phosphorus stress for sustainable crop production: a review,” J. Soil Sci. Plant Nutr. 12, 547–561 (2012).
H. M. Bilal, T. Aziz, M. A. Maqsood, M. Farooq, and G. Yan, “Categorization of wheat genotypes for phosphorus efficiency,” PLoS One 13, e0205471 (2018).
P. C. Brookes, D. S. Powlson, and D. S. Jenkinson, “Measurement of microbial biomass phosphorus in soil,” Soil Biol. Biochem. 14, 319–329 (1982).
W. F. Cong, L. D. B. Suriyagoda, and H. Lambers, “Tightening the phosphorus cycle through phosphorus-efficient crop genotypes,” Trends Plant Sci. 25, 967–975 (2020).
M. D. Corre, R. R. Schnabel, and J. A. Shaffer, “Evaluation of soil organic carbon under forests, cool-season and warm-season grasses in the north-eastern US,” Soil Biol. Biochem. 31, 1531–1539 (1999).
F. Eivazi and M. A. Tabatabai, “Phosphatase in soils,” Soil Biol. Biochem. 9, 167–172 (1977).
FAOSTAT, Food and Agriculture Organization, 2019. http://www.fao.org.
J. Grace, K. L. Sharma, K. V. Seshadri, C. Ranganayakulu, K. V.Subramanyam, G. Bhupal, S. H. K. Sharma, G. Ramesh, P. N. Gajbhiye, and M. Madhavi, “Evaluation of sweet orange (Citrus sinensis L. Osbeck) cv. Sathgudi budded on five rootstocks for differential behaviour in relation to nutrient utilization in alfisol,” Commun. Soil Sci. Plant Anal. 43, 985–1014 (2012).
P. Hinsinger, L. Herrmann, D. Lesueur, A. Robin, J. Trap, K. Waithaisong, and C. Plassard, “Impact of roots, microorganisms and microfauna on the fate of soil phosphorus in the rhizosphere,” Annu. Plant Rev. 48, 377–407 (2015).
V. Jensen, “Decomposition of angiosperm tree leaf litter,” in Biology of Plant Litter Decomposition, Ed. by C. H. Dickinson and G. J. F. Pugh (Academic, New York, 1974), pp. 69–104.
Y. P. Kalra, Handbook of Reference Methods for Plant Analysis (CRC Press, London, 1998).
S. Kumar, O. P. Awasthi, A. K. Dubey, R. Pandey, V. K. Sharma, A. K. Mishra, and R. M. Sharm, “Root morphology and the effect of rootstocks on leaf nutrient acquisition of Kinnow mandarin (Citrus nobilis Loureiro × Citrus reticulate Blanco),” J. Hortic. Sci. Biotechnol. 93, 100–106 (2018).
S. Kuo, “Phosphorus,” in Methods of Soil Analysis, Part 3: Chemical Methods, Ed. by D. L. Sparks, et al. (Soil Science Society of America, American Society of Agronomy, Madison, WI, 1996), pp. 869–920.
P. Marschner, D. Crowley, and Z. Rengel, “Rhizosphere interactions between microorganisms and plants govern iron and phosphorus acquisition along the root axis e model and research methods,” Soil Biol. Biochem. 43, 883–894 (2011).
P. Marschner, Mineral Nutrition of Higher Plants, 3rd ed. (Academic, London, 2012).
S. M. Mousavi, B. Motesharezadeh, H. Mirseyed Hosseini, H. Alikhani, and A. A. Zolfaghari, “Root-induced changes of Zn and Pb dynamics in the rhizosphere of sunflower with different plant growth promoting treatments in a heavily contaminated soil,” Ecotoxicol. Environ. Saf. 147, 206–216 (2018).
J. Murphy and J. P. Riley, “A modified single solution method for the determination of phosphate in natural waters,” Anal. Chim. Acta 27, 31–36 (1962).
E. Ngullie, A. K. Singh, A. Sema, and A. K. Srivastava, “Citrus growth and rhizosphere properties,” Commun. Soil Sci. Plant Anal. 46, 1540–1550 (2015).
M. Nuruzzaman, H. Lambers, M. D. A. Bolland, and E. J. Veneklaas, “Distribution of carboxylates and acid phosphatase and depletion of different phosphorus fractions in the rhizosphere of a cereal and three grain legumes,” Plant Soil 281, 109–112 (2006).
L. P. Olander and P. M. Vitousek, “Biological and geochemical sinks for phosphorus in soil from a wet tropical forest,” Ecosystems 7, 404–419 (2004).
P. Ollitrault and L. Navarro, “Citrus,” in Fruit Breeding, Part 3: Tree Fruits, Ed. by M. L. Badenes and D. H. Byrne (Springer-Verlag, Boston, 2012), pp. 623–662.
T. Raiesi, A. R. Hosseinpur, and H. Raiesi, “The influence of bean rhizosphere on the biological properties and phosphorus fractionation in the calcareous soils amended with municipal sewage sludge,” J. Arid Land 7, 644–652 (2015).
T. Raiesi and A. R. Hosseinpur, “Phosphorus availability and some biological properties in the bean (Phaseolus vulgaris) rhizosphere,” Commun. Soil Sci. Plant Anal. 48, 501–510 (2017).
T. Raiesi, A. Hosseinpur, and B. Moradi, “The influence of the wheat rhizosphere on phosphorus desorption kinetics in calcareous soils amended with municipal sewage sludge,” S. Afr. J. Plant Soil 34, 283–290 (2017).
T. Raiesi and B. Moradi, “Young navel orange rootstock improves phosphorus absorption from poorly soluble pools through rhizosphere processes,” Rhizosphere 17, 100316 (2021).
E. Richardson and R. J. Simpson, “Soil microorganisms mediating phosphorus availability,” Plant Physiol. 156, 989–996 (2011).
S. Savci, “Investigation of effect of chemical fertilizers on environment,” APCBEE Procedia 1, 287–292 (2012).
G. Schaller, “pH changes in the rhizosphere in relation to the pH-buffering of soils,” Plant Soil 97, 444–449 (1987).
B. Seeling and R. J. Zasoski, “Microbial effects in maintaining organic and inorganic solution phosphorus concentrations in a grassland topsoil,” Plant Soil 148, 277–284 (1993).
M. A. Tabatabai and J. M. Bremner, “Use of p-nitrophenyl phosphate for assay of soil phosphatase activity,” Soil Biol. Biochem. 1, 301–307 (1969).
G. Visioli, M. Lauro, T. Vamerali, C. Dal Cortivo, A. Panozzo, S. Folloni, C. Piazza, and R. A. Ranieri, “Comparative study of organic and conventional management on the rhizosphere microbiome, growth and grain quality traits of tritordeum,” Agronomy 10, 1717 (2020).
T. S. Walker, H. P. Bais, E. Grotewold, and J. M. Vivanco, “Root exudation and rhizosphere biology,” Plant Physiol. 132, 44–51 (2003).
G. Wollum II, “Cultural methods for soil microorganism,” in Methods of Soil Analysis, Part 2: Chemical and Microbiological Properties, Ed. by A. Page, et al. (Soil Science Society of America, American Society of Agronomy, Madison, WI, 1982), pp. 781–802.
F. B. Zambrosi, D. Mattos, R. M. Boaretto, J. A. Quaggio, T. Muraoka, and J. P. Syvertsen, “Contribution of phosphorus (32P) absorption and remobilization for citrus growth,” Plant Soil 355, 353–362 (2012).
ACKNOWLEDGMENTS
The authors wish to thank Citrus and Subtropical Fruits Research Centre for providing research facilities.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
CONFLICT OF INTEREST
The authors declare that they have no conflict of interest.
ADDITIONAL INFORMATION
AcP: Acid phosphatase, AlP: Alkaline phosphatase, CARB-P: Phosphorus bound to carbonates, DOC: Dissolved organic carbon, DW: Dry weight, EXCH-P: Exchangeable phosphorus, Fe-Al-P: Phosphorus bound to Fe-Al oxides, MBP: Microbial biomass phosphorus, P: Phosphorus, PAE: Phosphorus acquisition efficiency, PCA: Principal component analysis, PO: Organic phosphorus, RES-P: Residual phosphorus, SC: Swingle citrumelo, SO: Sour orange, TC: Troyer citrange.
Rights and permissions
About this article
Cite this article
Tahereh Raiesi, Moradi, B. & Mousavi, S.M. Alterations of P Fractions and Some Biochemical Features in Rhizosphere Soil Induced by the Root Activities of Citrus Rootstocks with Different P Acquisition Efficiency. Eurasian Soil Sc. 55, 212–220 (2022). https://doi.org/10.1134/S1064229322020107
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1064229322020107