Abstract
Vegetation restoration projects have been implemented in China’s degraded karst ecosystems (rocky desertification areas) for more than 20 years. There is an urgent need to evaluate the impact of different vegetation restoration modes on the ecological environment. The status of the soil environment under different vegetation restoration modes in degraded karst ecosystems was determined in the Guanling-Zhenfeng demonstration area of rocky desertification control in Guizhou, China. The differences in soil microbial quantity; soil microbial biomass carbon (MBC), nitrogen (MBN) and phosphorus (MBP); and soil nutrients were compared among five typical vegetation restoration modes (Hylocereus undatus 'Foo-Lon’ (HUF), Lonicera japonica Thunb. (LJT), Zanthoxylum bungeanum Maxim. (ZBM), artificially accelerated forest regeneration (AFR), and hillclosing afforestation (HA)) implemented continuously for 13–15 years. Pearson correlation analysis and redundancy analysis (RDA) were utilized to analyse the environmental factors affecting the soil microbial quantity and biomass under the five vegetation restoration modes. The results showed that the soil organic carbon (SOC), total nitrogen (TN), total phosphorus (TP), calcium (Ca), and ammonium nitrogen contents under HA were significantly higher than those under the other vegetation restoration modes (P < 0.05). There were no significant differences in the soil microbial quantity or biomass among the different vegetation restoration modes, and the other microbial indicators, except MBP, reached their highest values at the HA site. The correlation analysis and RDA showed that TP, TN, vegetation type, and SOC were the main factors affecting the soil microbial quantity and biomass under the 5 vegetation restoration modes and that TP was the limiting factor for vegetation restoration in degraded karst ecosystems. The results of our study provide insights into the selection of appropriate vegetation reconstruction measures and modes for degraded karst ecosystems.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
INTRODUCTION
Karst ecosystems in South China are fragile, with slow soil formation rates, shallow soil, and a low carrying capacity. In the context of intense human-land conflicts, irrational land use has led to ecosystem degradation, habitat fragmentation, serious soil erosion, and high bedrock exposure rates; these factors have resulted in rocky desertification, which has become one of the main obstacles to the sustainable development of karst areas [4, 49]. Due to the fragility of the foundational environment, positive succession in karst ecosystems occurs only very slowly and can be terminated or reversed due to external disturbances; this makes recovery after the destruction of karst ecosystems extremely difficult [13, 25]. Vegetation recovery plays an essential role in ecological restoration and has implications for the ability of the environment to support socioeconomic activities [53]. In 2018, statistics showed that grass was growing in artificial afforestation areas and that forest-grass vegetation protection played a leading role (contribution rate of 65.5%) in the reversal of rocky desertification [10]. This area is characterized by high-intensity agricultural activities, little arable land per capita, low socioeconomic development, and a lack of coordination between ecological benefits and socioeconomic benefits [44, 46]. Therefore, ecological restoration must be considered with regard to socioeconomic factors. Artificial economic forests and their related industries can alleviate the conflicts between humans and land in this area.
Rocky desertification control efforts typically have problems such as low stability in planted vegetation communities, a lack of regional suitability, and weak sustainability of the ecological restoration projects implemented for desertification control [39]. Evaluations of whether vegetation restoration modes are scientifically appropriate and have long-term effects are urgently needed. Artificial afforestation and natural restoration have their own advantages and disadvantages [5, 23]. Artificial forests have a simple structure and a single dominant species, and it is difficult for artificial forests to form ecosystems that are self-sustaining and self-stabilizing. These factors can lead to a decrease in the ability of the ecosystem to resist outside interference, which can easily cause a large-scale reduction in crop production as well as soil quality degradation [2, 8]. The economic benefits produced by some artificial economic forests have emerged gradually, effectively alleviating human-land conflicts; this result has confirmed the effectiveness of stony desertification control to a certain extent. In contrast, natural restoration vegetation communities develop towards complex, complete and stable structures, and their species diversity, soil nutrients, microbial biomass, and microbial diversity are significantly higher than those of artificial forests [20, 40]. Changes in vegetation communities are always related to changes in the soil [35]; that is, any change process in the vegetation community is also a process of mutual influence and interaction between vegetation and soil. Therefore, the effects of vegetation restoration modes can be evaluated through the soil-vegetation system.
Soil is the foundation of plant growth and the main source of plant nutrients, and it plays an important role in regulating and driving plant growth and activity [6]. Soil microorganisms are sensitive indicators and early warning systems for soil ecosystem change [14, 34] and play an important role in the mineralization of soil organic matter, nutrient transformation, material circulation, etc. [50]. Studies of these processes are of great value to the restoration and reconstruction of karst ecosystems [19]. Vegetation and soil interact with and influence each other [41, 47]. Positive succession in vegetation communities can improve the stability and interference resistance of karst forest ecosystems and is beneficial to the accumulation of soil nutrients [19]. There is an obvious relationship between the diversity of vegetation and the grade of rocky desertification in karst areas; with the increase in the grade of rocky desertification, the physical and chemical properties of soil degrade at first and then improve gradually [37]. Different restoration modes can significantly increase soil carbon and nitrogen fixation in karst areas, and natural restoration is the best restoration mode for this purpose [42]. In the process of vegetation restoration, with the gradual restoration of surface vegetation, biodiversity also increases gradually [22].
At present, studies on the relationship between vegetation restoration and the soil environment in degraded karst ecosystems focus mainly on different successional stages or individual vegetation community types and adopt the “space-for-time substitution” method [19, 22]. However, there are few comparative studies on the relationship between different vegetation restoration modes and the soil environment among vegetation restoration projects of similar durations. Therefore, in this study, five vegetation restoration modes (artificial afforestation modes: (1) Hylocereus undatus 'Foo-Lon’, (2) Lonicera japonica Thunb., (3) Zanthoxylum bungeanum Maxim; natural restoration modes: (4) artificially accelerated forest regeneration and (5) hillclosing afforestation) applied in degraded karst ecosystems were taken as the object of study. The physical and chemical properties and microbial quantity and biomass of the topsoil (0~20 cm) were studied. The main purposes of this study are to determine 1) the soil physical and chemical properties and soil microbial quantity and biomass under the different vegetation restoration modes, 2) which environmental factors have important effects on microbial quantity and biomass, and 3) which environmental factors are the limiting factors for vegetation restoration. Our study is expected to provide a basis for vegetation restoration and sustainable development in degraded karst ecosystems.
MATERIALS AND METHODS
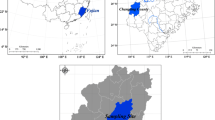
Study site. The study area is located in the Guanling-Zhenfeng Demonstration Area of Rocky Desertification Control (25°37′40″~25°42′30″ N, 105°35′00″~105°43′20″ E, 530~1473 m above sea level) in the Beipan River basin at the border between Zhenfeng County and Guanling County, Guizhou Province, Southwest China (Fig. 1a). This area is dominated by a subtropical humid monsoon climate. The mean annual temperature is 18.4°C, the mean annual precipitation is 1100 mm, and the rainfall from May to October accounts for approximately 83% of the annual rainfall. High-intensity hillside ploughing and deforestation activities have occurred in this area. The vegetation coverage is low, and the land is dominated by moderate and heavy rocky desertification (bedrock outcropping rate is 20~80%). According to the WRB classification standard, the soils in the study area are mainly cambisols. The soil types in this area are yellow limestone soil and brown limestone soil since their parent rock is Triassic carbonate. Due to the lack of a transition layer between the rock interface and the soil, soil loss and block slip easily occur in rainstorms, resulting in thin and discontinuous soil layers, and the soil is mainly distributed in karst depressions and lapiaz, grike, and rhegmalypt formations.
Location of the study site. Study site (A) in the South China Karst; location of sampling plots (B) in the study area; landscape view of the sample plot from above (C): (a) Hylocereus undatus 'Foo-Lon’ (HUF), (b) Lonicera japonica Thunb. (LJT), (c) Zanthoxylum bungeanum Maxim. (ZBM), (d) artificially accelerated forest regeneration (AFR), (e) hillclosing afforestation (HA), and (f) Zea mays L. (CK).
Rocky desertification control has been implemented in the study area for more than 20 years. Several typical vegetation restoration species have been planted in the study area (Fig. 1c): a) Hylocereus undatus 'Foo-Lon’ began to be popularized and planted on a large scale in 2007. It is mainly planted on sloping farmland with a continuous soil distribution. b) Lonicera japonica Thunb. is one of the early vegetation restoration species and is mainly planted in lapiaz or near exposed bedrock. c) Zanthoxylum bungeanum Maxim. is one of the early vegetation restoration species because it has economic value; it is popular with local farmers, and an industry has developed around its production. d) Artificially accelerated forest regeneration areas are planted with native trees and shrubs. Natural vegetation succession processes are allowed to occur in these areas without interference from human activities. e) Hillclosing afforestation areas are established on thin soils (soil depth 0~15 cm). The vegetation roots grow along grikes, and the dominant vegetation type is low trees.
Experimental design. Before sampling, a survey was conducted on the different vegetation restoration modes in the study area. Combined with the plots monitored by the team for a long time, plots with the same or similar vegetation restoration durations were selected for this study. Due to the different life cycles of the artificial economic forests, the three HUF, LJT, and ZBM plots were replanted many times during the study period, but no other species were planted in the study plots. In the end, 5 plots (Fig. 1B) in the study area were selected. Three standard sampling squares of 10 × 10 m were established in each plot. The average soil chemical properties at the initial stage (2008) of the five restoration modes are shown in Table 1, and detailed information on the sample plots is shown in Table 2.
Applicable scope and main features of the 5 restoration modes. HUF: Hylocereus undatus 'Foo-Lon’ is drought resistant and tolerant of low fertility. Under the condition of sufficient water, the root system can maintain a relatively vigorous growth state; this species has a beneficial role in maintaining water and soil and has produced good economic benefits after successful cultivation. LJT: The branches and leaves of honeysuckle cover a large area and have a good water and soil maintenance effect. Additionally, Lonicera japonica Thunb., as a multipurpose Chinese medicinal material, can increase farmers' economic income. It can be planted in the soil of stone gaps or holes due to its low requirements for the growth environment. ZBM: Zanthoxylum bungeanum Maxim. is a popular condiment. Its root system is well developed and drought tolerant. It can improve the environment while also providing economic benefits. ZBM has played an important role in local ecological restoration and alleviating poverty among farmers. AFR: Due to the harsh natural environment in rocky desertification areas, it is very difficult to restore vegetation. Replanting drought-tolerant and barren-tolerant shrubs and low trees will speed up the reconstruction and restoration of vegetation in rocky desertification areas. HA: Places with discontinuous land distribution, thin soil layers, and high organic matter content are suitable for the growth of a variety of trees and shrubs. It is possible to make full use of the advantages of both natural restoration to afforestation and to prohibit all human activities that are not conducive to the growth and reproduction of vegetation. CK: Zea mays L. is the main food crop in this area. With the implementation of the project of returning farmland to forest and grassland, especially the development of economic forest, the planting area of corn has gradually decreased, and Zea mays L. has been planted in the studied sample plot for 13 consecutive years.
Soil sample collection and experimental analysis. In July 2020, 6 sample plots were selected (5 vegetation restoration modes and 1 corn plot as the control group), and 3 sampling points were selected using the diagonal method in each plot. Litter, moss and other debris were removed before sampling from the surface of the sampling point. Topsoil (0~20 cm) was collected from each sample square (in sampling areas with soil layers thinner than 20 cm, samples were taken of the entire soil profile). Before samples were taken to determine the soil chemical properties, 0~20 cm undisturbed soil was obtained in 5 cm layers by using a cutting ring (5 cm high, 5 cm diameter); these samples were used to measure the soil water content. Three soil samples were collected from each sampling square, and roots, gravel, and other debris were removed. The samples were divided into 2 parts. One part was sieved through a 2 mm sieve and immediately placed into the incubator with an ice bag. After the samples were collected, they were stored in a refrigerator at 4°C in the laboratory for the determination of the soil microbial biomass carbon (MBC), nitrogen (MBN), and phosphorus (MBP) and the quantities of bacteria, fungi and actinomycetes. The other part of each soil sample was naturally air-dried, any debris was removed, and the soil was sieved through a 100-mesh sieve before the determination of the nutrient contents and pH.
The soil microbial biomass was determined using the chloroform fumigation-extraction method [37]. MBC and MBN were extracted by K2SO4, and the conversion coefficient for K was 0.45. MBP was extracted by NaHCO3, pH correction was performed, and the conversion coefficient for K was 0.4. The soil microbial quantity was calculated by the spread plate method, with dilutions of microbial cultures spread onto the following media: beef extract peptone agar medium for bacteria, Martin medium for fungi, and modified Gauze’s synthetic medium no. 1 for actinomycetes. More details on the above determination methods can be found in the literature [43]. The pH value of the soil was measured by an acidity meter. The SWC was measured by the drying method. The soil organic C (SOC) content was determined using the dichromate oxidation method [24], and the total N (TN) was analysed by the Kjeldahl method [3]. After the soil sample was digested by HCIO4-H2SO4, the total P (TP) was determined by the molybdenum blue method [33]. After the soil sample was melted and diluted with NaOH, the total potassium (TK) was determined by the flame photometer method [26]. After the soil was extracted with saturated CaSO42H2O solution, the nitrate nitrogen content was measured by phenoldisulfonic acid colorimetry [31], soil samples were extracted with 2 mol L–1 KCl solution, and the ammonium nitrogen content was analysed by indophenol blue colorimetry [7]. The total amounts of CaCO3 and MgCO3 in soil were determined by the air volume method [1], and the result was the calcium content (Ca). In addition, environmental factors other than soil chemistry properties were recorded, including altitude, slope, vegetation coverage, vegetation type, vegetation restoration years, soil depth, and soil water content.
Statistical analyses. Microsoft Excel 2010 software (Microsoft Corporation, Santa Rosa, Ca, USA) was utilized to preliminarily collate the data. The multcomp, agricolae, and ggplot2 packages in R software (R Development Core Team 2019) were used to perform one-way ANOVA on the soil pH, SWC, MBC, nutrient contents, microbial quantity, and biomass. The least significant difference (LSD) method was used to make multiple comparisons, and histograms were drawn. The pheatmap and corrplot2 software packages were used to analyse the correlation between environmental factors and soil microbial quantity and biomass using the Pearson correlation analysis method and to visualize the results. CANOCO 5.0 (Microcomputer Power, Ithaca, NY, USA) was used to analyse the relationship between soil microbial quantity and biomass and environmental factors in RDA and to extract the simple and conditional effects of each factor on the variation in soil microbial quantity and biomass. The data are expressed as the average ± standard deviation. The significance level was set at P = 0.05, and the extreme significance level was set at P = 0.01.
RESULTS
Characteristics of changes in soil physical and chemical properties under different vegetation restoration modes. As shown in Fig. 2, the range of soil pH under the different restoration modes was 7.32~7.58, and the pH decreased in the order LJT > HUF > CK > AFR > HA > ZBM (Fig. 2a). There were no significant differences between the different recovery modes. The SWC varied from 20.46 to 25.77% and decreased in the order AFR > LJT > ZBM > HA > CK > HUF (Fig. 2b). The SWC values of AFR and LJT were significantly higher than those of the other restoration modes (P < 0.05). In terms of soil nutrients, the contents of SOC, TN, TP, TK, Ca, nitrate nitrogen and ammonium nitrogen in HA were higher than those in the other vegetation restoration modes (Figs. 2c, 2d, 2e, 2g, 2i). Among them, SOC, TN, TP, Ca and ammonium nitrogen were significantly higher in HA than in the other vegetation restoration modes (P < 0.05). Furthermore, the SOC and TP of HUF were significantly lower than those of the other vegetation restoration methods (P < 0.05). The results showed that compared with the initial stage of vegetation restoration (Table 1), the SOC, TN, TP and TK contents all increased under the LJT, ZBM, AFR, and HA, indicating that these four modes were all conducive to the accumulation of soil nutrients, of which HA was more beneficial than other modes. The soil nutrient content of LJT decreased compared with that in the previous period of vegetation restoration.
Soil pH, soil moisture content and soil nutrients under the different vegetation restoration modes. Mean values (means ± SD, n = 3). Lowercase letters indicate significant differences in the same index among different vegetation restoration modes (n = 3, P < 0.05). HUF: Hylocereus undatus 'Foo-Lon’; LJT: Lonicera japonica Thunb.; ZBM: Zanthoxylum bungeanum Maxim.; AFR: Artificially accelerated forest regeneration; HA: Hillclosing afforestation; CK: Zea mays L.
Changes in the MBC, MBN, MBP and quantity of bacteria, fungi and actinomycetes under the different vegetation restoration modes. The MBC, MBN, MBP, and the quantity of bacteria, fungi and actinomycetes had no obvious regularity among the different vegetation restoration methods. The MBC of HUF and HA were significantly higher than in the other restoration modes (P < 0.05), 360.67, 363.00 mg/kg, respectively (Fig. 3a). The MBN of HA was significantly higher than that of the other vegetation restoration modes, at 30.43 mg/kg (Fig. 3b). The MBP of LJT was the highest, at 16.06 mg/kg, which was significantly different from those of HUF and AFR (Fig. 3c). The quantities of soil bacteria and fungi in ZBM and HA were significantly higher than those in the other vegetation restoration modes (P < 0.05); the bacterial counts were 2.80 × 106 and 2.60 × 106 cfu/g, respectively, and those of the fungi were 1.19 × 104 and 1.33 × 104 cfu/g, respectively (Figs. 3d, 3e). The quantity of actinomycetes in HA was significantly higher than that in the other vegetation restoration modes (P < 0.05), at 2.27 × 106 cfu/g (Fig. 3f). The MBC, MBN and MBP were the lowest under AFR. The quantities of bacteria, fungi and actinomycetes were relatively low in HUF and LJT. The results showed that there was no significant difference in soil microbial biomass, bacteria, fungi and actinomycetes in the plantation compared with those in CK, but the soil microbial biomass and number under natural restoration measures were higher than those in the plantation.
Correlation between environmental factors and soil microbial quantity and biomass. There were no significant correlations between MBC and environmental factors (Fig. 4). MBN was significantly positively correlated with TK. MBP was significantly positively correlated with the restoration years and significantly negatively correlated with nitrate nitrogen. Bacteria were significantly positively correlated with TP and vegetation type. Fungi were significantly positively correlated with TN, TP, and altitude; extremely significantly positively correlated with vegetation type; and significantly negatively correlated with pH. Actinomycetes were significantly positively correlated with vegetation type and altitude; extremely significantly positively correlated with SOC, TN, and TP; and significantly negatively correlated with TK. The results showed that the soil microbial quantity and biomass, especially those of fungi and actinomycetes, were significantly affected by environmental factors.
Correlation between soil microbial quantity and biomass and environmental factors. The circles in the figure indicate significance (P < 0.05). Red represents a negative correlation, blue represents a positive correlation, the size of the circle indicates the size of the correlation coefficient, and the color of the circle indicates the degree of correlation. Soil water content (%), soil depth (cm), slope (°), vegetation coverage (%), altitude (m).
Redundancy analyses of environmental factors and soil microbial quantity and biomass. RDA was carried out with soil bacteria, fungi, actinomycetes, MBC, MBN and MBP as response variables and soil physical and chemical properties and other environmental factors as environmental explanatory variables (Fig. 5, Table 3). The explanatory variables of the first axis and the second axis explained 81.37% and 11.51% of the variation, respectively, and the cumulative amount of explanation was 92.88%. This result indicates that the selected environmental factors can effectively explain the changes in soil microbial quantity and biomass. In terms of simple effects, environmental factors explained changes in the soil microbial quantity and biomass as follows, in %: TP (37.1) > TN (34.8) > vegetation type (32.6) > SOC (31.0)> altitude (22.4) > restoration years (16.5) > pH (13.1) > soil water content (12.7). Of these factors, TP, TN, vegetation type and SOC had significant effects on the soil microbial quantity and biomass. From the perspective of conditional effects, soil microbial quantity and biomass were affected mainly by TP and TN, and the results reached extreme significance. According to the RDA results, the effect of environmental factors on soil microbial biomass was lower than that on soil bacteria, fungi and actinomycetes; moreover, actinomycetes and fungi were more sensitive than bacteria to environmental changes, which was consistent with the results of the correlation analysis (Fig. 4). Among the environmental factors, SOC, TN, altitude, TP, vegetation type, restoration years and ammonia nitrogen had the strongest effects on the actinomycetes.
DISCUSSION
Comparative Analysis of Soil Nutrients and Microbial Quantity and Biomass under Different Vegetation Restoration Modes
Hillclosing afforestation is more conducive to soil nutrient accumulation than other modes. In this study, there were certain differences in soil nutrient levels under the different vegetation restoration modes. Among them, the SOC, TN, TP, Ca, and ammonium nitrogen contents in the closed-hill afforestation area were significantly higher than those in other vegetation restoration mode areas. As shown in Table 1, the SOC, TN, and TP of the closed-hill afforestation model are 2.72, 2.52, and 3.06 times higher than those of the initial restoration stage. The reason may be that natural restoration is more favourable to soil nutrient accumulation than other restoration methods [42]. After 15 years of hillclosing afforestation, the vegetation community at the HA site is mainly composed of low trees (1.0~2.3 m), shrubs, and fewer herbaceous plants and thicker litter than in the other restoration vegetation communities in this study. The HA site is in the tree-shrub stage of positive karst vegetation community succession and is nearly unaffected by human activities; thus, it provides good conditions for the accumulation of soil nutrients [21]. The average mass fractions of SOC, TN, TP, Ca and ammonium nitrogen at the HA site were 2.86~9.41, 2.67~7.17, 2.02~8.64, 4.73~9.51, and 1.79~4.49 times higher than those of the other restoration modes, respectively. Nutrients and carbon released from litter create an available nutrient pool for plants [9, 11]. Moreover, the surface litter in the HA area was thicker than that in the other restoration areas, which is more conducive to nutrient accumulation. These results indicated that except HUF, other vegetation restoration modes were conducive to the accumulation of soil nutrients, of which hillclosing afforestation had obvious advantages for the improvement of the soil environment.
Hillclosing afforestation resulted in higher soil nutrient levels, making it more conducive to the growth and reproduction of soil microorganisms. Different restoration modes and management strategies led to different structures and functions of the soil microbial community. The MBC of the five vegetation restoration modes ranged from 344.33~363.00 mg/kg, showing little variation; this range is lower than that of plateau karst [51]. The range of MBN was 24.37~30.43 mg/kg, showing little variation, which is lower than that of subtropical forest soil and karst peak cluster-depression formation [17]. The range of microbial biomass phosphorus was 27.93~160.57 mg/kg, which is quite wide and is higher than that in a karst peak cluster-depression formation [48]. Vegetation restoration and vegetation diversity can increase microbial biomass, which may be one of the reasons for the high MBC and MBN at the HA site. The quantities of bacteria, fungi and actinomycetes were also the highest at the HA site. The microbial quantities in the five vegetation restoration modes decreased in the order bacteria > actinomycetes > fungi; these results are consistent with the results reported by Q.J. Tan et al. [36]. Although the soil nutrient content of HA in the initial stage of vegetation restoration was not the highest among the five restoration modes (Table 1), its nutrient accumulation was faster than that in other restoration modes, which resulted in the soil at the HA having higher organic carbon and nitrogen levels than the other restoration modes and therefore provided more carbon and nitrogen sources for microorganisms. After a long period of natural recovery, the plant species diversity in natural restoration areas will continue to increase, producing more litter and root exudates that provide rich substrates for microorganism growth and thus accelerate microorganism reproduction and growth; this conclusion is consistent with the results of W. Gao et al. [12].
Analysis of the factors causing the soil nutrients and microbial quantity and biomass of the artificial economic forests to be lower than those resulting from hillclosing afforestation. Through the comparative analysis of the soil nutrients and microbial quantity and biomass under the different vegetation restoration modes, we determined that compared with restoration with artificial economic forests, natural restoration was more conducive to soil nutrient accumulation and improved the microbial community environment. The main reason behind this finding is that plantation species such as dragon fruit, honeysuckle and prickly ash not only perform vegetation restoration functions but also must provide economic benefits. Because they produce fruit, which affects the material and nutrient cycles of the “soil-vegetation-litter” system, artificial economic forest ecosystems are energy-consuming, unstable systems that need to absorb more nutrients from the soil than they return to the soil to ensure their own growth and maintain fruit production [30]. In addition, most of the artificial economic forests in this area are managed by “relying on nature for a living” approaches. Fertilization measures are unscientific or nonexistent, which may be the main reason for the low soil nutrient levels in these artificial economic forests. Due to the decrease in soil nutrients, the substrate conditions for microbial survival worsen, resulting in a decrease in the microbial turnover rate [18]. In the future, to provide scientific support for precision fertilization and field management, in-depth studies should be carried out on nutrient demand, the soil nutrient supply and nutrient cycling in artificial economic forests.
Effects of Environmental Factors on Soil Microbial Quantity and Biomass
Soil fungi and actinomycetes are more sensitive than bacteria to environmental changes. Figure 4 shows that there was no significant correlation between MBC and environmental factors, indicating that MBC varies greatly in the study area. The reason may be that there are differences in litter, root exudates and soil quality of different vegetation types, which have a certain impact on MBC [32]. Therefore, MBC cannot be used to characterize the soil microbial biomass in this study. Vegetation, pH, soil nutrients, organic matter, etc., have a greater impact on soil fungi and actinomycetes [15, 38]. The quantities of soil fungi and actinomycetes were positively correlated with environmental factors such as TN, TP, VT and AL. Moreover, Fig. 5 confirms that environmental factors had significant effects on soil fungi and actinomycetes. MBC, MBN, MBP and bacteria had no significant correlation with environmental factors. These results indicate that fungi and actinomycetes are more sensitive than bacteria to environmental changes and that their growth and reproduction are more easily affected by the external environment.
Analysis of the main factors limiting vegetation restoration in degraded karst ecosystems. In this study, TP explained 37.1% of the variation in soil microbial quantity and biomass, indicating that TP has an important impact on vegetation restoration in these rocky desertification areas. That is, P is the main limiting factor on vegetation growth in karst rocky desertification areas; this finding is consistent with the fact that terrestrial plants in China are generally P limited [16]. P affects microorganisms mainly indirectly by affecting the soil carbon cycle and chemical properties [22]. SOC controls energy and nutrient cycling in the soil and is a stable source of energy and nutrients for microbial communities, and nitrogen can increase the rate of organic matter decomposition by microorganisms [27]. SOC and TN were positively correlated with MBC, MBP, and the quantities of bacteria, fungi and actinomycetes, and they were able to explain 31.0 and 34.8%, respectively, of the simple effects; this finding was significant. N was second only to P in terms of explanatory power and limited vegetation restoration in this area. Vegetation type was positively correlated with the quantities of bacteria, fungi and actinomycetes (simple effect explanation, 32.6%); vegetation type affects the soil biota by affecting the soil water content, temperature, ventilation, pH value and organic carbon and nitrogen levels [52].
CONCLUSIONS
(1) Among the five vegetation restoration modes in the study area, the contents of soil SOC, TN, TP, Ca and ammonium nitrogen at the hillclosing afforestation site were significantly higher than those at the other vegetation restoration modes, indicating that hillclosing afforestation was more conducive to the accumulation of soil nutrients.
(2) Except for MBP, the soil microbial quantity and biomass indicators were the highest under hillclosing afforestation, which indicated that natural restoration vegetation was more beneficial to soil microbial reproduction and growth than the other restoration modes.
(3) Correlation analysis and RDA showed that the SOC, TN, TP and vegetation type were important factors affecting the soil microbial quantity and biomass. Soil fungi and actinomycetes were more sensitive than soil bacteria to changes in environmental factors. P is the principal factor limiting vegetation restoration in degraded karst ecosystems.
It is worth noting that the hillclosing afforestation is more conducive to the accumulation of soil nutrients and the reproduction and growth of microorganisms. In terms of improving soil quality, hillclosing afforestation is a more effective vegetation restoration measure than artificial economic forests, but artificial economic forests provide obvious social and economic benefits, and they form the core of the eco-economic industry in rocky desertification control areas. How to develop and improve effective fertilization measures and sustainable management strategies for artificial economic forests in this area needs to be further studied.
REFERENCES
S. D. Bao, H. Y. Qin, and J. S. Lao, Soil and Agricultural Chemistry Analysis (China Agricultural Press, Beijing, 1988), pp. 202–204.
A. Bobiec, “The mosaic diversity of field layer vegetation in the natural and exploited forests of Białowieża,” Plant Ecol. 136 (2), 175 (1998). https://doi.org/10.1023/A:1009736823553
J. M. Bremner, “Determination of nitrogen in soil by the Kjeldahl method,” J. Agric. Sci. 55 (1), 11–33 (1960). https://doi.org/10.1017/S0021859600021572
J. H. Cao, D. X. Yuan, L. Q. Tong, A. Mallik, H. Yang, and F. Huang, “An overview of karst ecosystem in Southwest China: current state and future management,” J. Resour. Ecol. 6 (4), 247–256 (2015). https://doi.org/10.5814/j.issn.1674-764x.2015.04.008
S. Cao, L. Chen, D. Shankman, C. Wang, X. Wang, and H. Zhang, “Excessive reliance on afforestation in China’s arid and semi-arid regions: lessons in ecological restoration,” Earth-Sci. Rev. 104 (4), 240–245 (2011). https://doi.org/10.1016/j.earscirev.2010.11.002
R. Condit, B. M. Engelbrecht, D. Pino, R. Perez, and B. L. Turner, “Species distributions in response to individual soil nutrients and seasonal drought across a community of tropical trees,” Proc. Natl. Acad. Sci. U.S.A. 110 (13), 5064–5068 (2013). https://doi.org/10.1073/pnas.1218042110
R. A. Dorich and D. W. Nelson, “Direct colorimetric measurement of ammonium in potassium chloride extracts of soils,” Soil Sci. Soc. Am. J. 47 (4), 833–836 (1983). https://doi.org/10.2136/sssaj1983.03615995004700040042x
R. Duan, C. Wang, X. A. Wang, Z. Zhu, and H. Guo, “Differences in plant species diversity between conifer (Pinus tabulaeformis) plantations and natural forests in middle of the Loess plateau,” Russ. J. Ecol. 40 (7), 501–509 (2009). https://doi.org/10.1134/S106741360907008X
H. Eijsackers and A. J. B. Zehnder, “Litter decomposition: a Russian matriochka doll,” Biogeochemistry 11 (3), 153–174 (1990). https://doi.org/10.1007/BF00004495
Forestry National, Grassland Administration China, Bulletin on rocky desertification in karst areas of China, 2018. http://www.forestry.gov.cn/main/138/20181214/ 161609114737455.html. Cited October 21, 2020.
J. Frouz, “Effects of soil macro- and mesofauna on litter decomposition and soil organic matter stabilization,” Geoderma 332, 161–172 (2018). https://doi.org/10.1016/j.geoderma.2017.08.039
W. Gao, M. M. Lin, Y. R. Huang, S. D. Huang, G. F. Ye, and Z. Q. Huang, “Effects of forest types and environmental factors on soil microbial biomass in a coastal sand dune of subtropical China,” J. Resour. Ecol. 11 (5), 454–465 (2020). https://doi.org/10.5814/j.issn.1674-764x.2020.05.003
D. Gillieson, P. Wallbrink, and A. Cochrane, “Vegetation change, erosion risk and land management on the Nullarbor Plain, Australia,” Environ. Geol. 28 (3), 145–153 (1996). https://doi.org/10.1007/s002540050087
L. Guo, Z. Sun, Z. Ouyang, D. Han, and F. Li, “A comparison of soil quality evaluation methods for Fluvisol along the lower Yellow River,” Catena 152, 135–43 (2017). https://doi.org/10.1016/j.catena.2017.01.015
H. Qin, Q. S. Ren, W. H. Yang, and C. X. Li, “Comparative studies on soil actinobacterial biodiversity after re-vegetation in the urban and rural hydro-fluctuation zone of the three gorges reservoir region,” Environ. Sci. 38 (5), 2065–2073 (2017).
W. Han, J. Fang, D. Guo, and Y. Zhang, “Leaf nitrogen and phosphorus stoichiometry across 753 terrestrial plant species in China,” New Phytol. 168 (2), 377–385 (2005). https://doi.org/10.1111/j.1469-8137.2005.01530.x
Y. He, C. T. Li, Y. C. Yu, H. P. He, and X. Tao, “Variation of subtropical forest soil microbial biomass and soil microbial community functional characteristics along the urban-rural gradient,” Chin. J. Appl. Ecol. 32 (1), 93–102 (2021). http://www.cjae.net/CN/10.13287/ j.1001-9332.202101.034
Z. L. He, “Soil microbial biomass and its significance in nutrient cycling and environmental quality assessment,” Soil 2, 61–69 (1997).
F. Hu, H. Du, F. P. Zeng, T. Q. Song, W. X. Peng, and F. Zhang, “Dynamics of soil nutrient content and microbial diversity following vegetation restoration in a typical karst peak-cluster depression landscape,” Acta Ecol. Sin. 38 (6), 2170–2179 (2018). https://doi.org/10.5846/stxb201703180458
P. Hu, J. Xiao, W. Zhang, L. Xiao, R. Yang, D. Xiao, J. Zhao, and K. L. Wang, “Response of soil microbial communities to natural and managed vegetation restoration in a subtropical karst region,” Catena 195, 104849 (2020). https://doi.org/10.1016/j.catena.2020.104849
Y. F. Huang, Y. G. Shu, S. Y. Xiao, and M. J. Chen, “Quantification of soil nutrient levels and enzyme activities in different grassland categories in karst mountains,” Acta Pratacult. Sin. 29 (6), 93–104 (2020). https://doi.org/10.11686/cyxb2019519
J. Huang, B. Hu, K. Qi, W. Chen, X. Pang, W. Bao, and G. L. Tian, “Effects of phosphorus addition on soil microbial biomass and community composition in a subalpine spruce plantation,” Eur. J. Soil Biol. 72, 35–41 (2016). https://doi.org/10.1016/j.ejsobi.2015.12.007
F. Hua, X. Wang, X. Zheng, B. Fisher, L. Wang, J. G. Zhu, Y. Tang, W. Y. Douglas, and S. W. David, “Opportunities for biodiversity gains under the world’s largest reforestation programme,” Nat. Commun. 7 (1), 12717 (2016). https://doi.org/10.1038/ncomms12717
K. R. Islam and R. R. Weil, “A rapid microwave digestion method for colorimetric measurement of soil organic carbon,” Commun. Soil Sci. Plant Anal. 29 (15–16), 2269–2284 (1998). https://doi.org/10.1080/00103629809370110
Z. C. Jiang, Y. Q. Lian, and X. Q. Qin, “Rocky desertification in Southwest China: impacts, causes, and restoration,” Earth-Sci. Rev. 132, 1–12 (2014). https://doi.org/10.1016/j.earscirev.2014.01.005
S. Jonasson and A. Michelsen, “Nutrient cycling in Subarctic and Arctic ecosystems, with special reference to the Abisko and Tornetrask region,” Ecol. Bull. 45, 45–52 (1996).
A. Y. Y. Kong, K. M. Scow, A. L. Córdova-Kreylos, W. E. Holmes, and J. Six, “Microbial community composition and carbon cycling within soil microenvironments of conventional, low-input, and organic cropping systems,” Soil Biol. Biochem. 43 (1), 20–30 (2011). https://doi.org/10.1016/j.soilbio.2010.09.005
E. E. Kuramae, E. Yergeau, L. C. Wong, A. S. Pijl, J. A. Veen, and G. A. Kowalchuk, “Soil characteristics more strongly influence soil bacterial communities than land-use type,” FEMS Microbiol. Ecol. 79 (1), 12–24 (2012). https://doi.org/10.1111/j.1574-6941.2011.01192.x
R. Li, L. J. Wang, M. Y. Sheng, and J. Guo, “Plant species diversity and its relationship with soil properties in karst rocky desertification succession,” Res. Soil Water Conserv. 23 (5), 111–119 (2016). https://doi.org/10.13869/j.cnki.rswc.2016.05.012
P. Meng, J. S. Zhang, and W. Fan, Agroforestry Ecosystem Research (Science Press, Beijing, 2004), pp. 183–185. https://dergipark.org.tr/en/pub/tbtkagriculture/ issue/11608/138344
D. J. Nicholas and A. Nason, “Determination of nitrate and nitrite,” in Methods in Enzymology (Academic, New York, 1957), Vol. 3, Ch. 144, pp. 981–984.
Ö. Kara and B. İlyas, “The effect of different land uses on soil microbial biomass carbon and nitrogen in Bartın province,” Turk. J. Agric. For. 32 (4), 281–288(2008).
S. R. Olsen and L. E. Sommers, “Total carbon, organic carbon, and organic matter,” in Methods of Soil Analysis, Part 2: Chemical and Microbiological Properties (American Society of Agronomy, Madison, WI, 1982), pp. 539–579.
J. Paz-Ferreiro and S. Fu, “Biological indices for soil quality evaluation: perspectives and limitations,” Land Degrad. Dev. 27 (1), 14–25 (2013). https://doi.org/10.1002/ldr.2262
E. J. Sayer, A. E. Oliver, J. D. Fridley, A. P. Askew, R. Mills, and J. P. Grime, “Links between soil microbial communities and plant traits in a species-rich grassland under long-term climate change,” Ecol. Evol. 7 (3), 855–862 (2017). https://doi.org/10.1002/ece3.2700
Q. J. Tan, T. Q. Song, W. X. Peng, F. P. Zeng, H. Du, H. Zhang, and F. J. Fan, “Characteristics of soil microbial populations and biomass under different ecosystems in a canyon karst region,” Acta Ecol. Sin. 34 (12), 3302–3310 (2014). https://doi.org/10.5846/stxb201310252573
D. Vance, P. C. Brookes, and D. S. Jenkinson, “An extraction method for measuring soil microbial biomass C,” Soil Biol. Biochem. 19 (6), 703–707 (1987). https://doi.org/10.1016/0038-0717(87)90052-6
E. Vukicevich, D. T. Lowery, J. A. Bennett, and M. Hart, “Influence of groundcover vegetation, soil physicochemical properties, and irrigation practices on soil fungi in semi-arid vineyards,” Front. Ecol. Evol. 7, 118 (2019). https://doi.org/10.3389/fevo.2019.00118
K. L. Wang, Y. M. Yue, H. S. Chen, X. B. Wu, J. Xiao, and X. K. Qi, “The comprehensive treatment of karst rocky desertification and its regional restoration effects,” Acta Ecol. Sin. 39 (20), 7432–7440 (2019). https://doi.org/10.5846/stxb201909051849
Y. Wei, L. F. Yu, J. C. Zhang, Y. C. Yu, and D. L. Deangelis, “Relationship between vegetation restoration and soil microbial characteristics in degraded karst regions: a case study,” Pedosphere 21 (1), 132–138 (2011). https://doi.org/10.1016/S1002-0160(10)60088-4
L. Wen, T. Q. Song, H. Du, K. L. Wang, W. X. Peng, F. P. Zeng, Z. X. Zeng, and T. G. He, “The succession characteristics and its driving mechanism of plant community in karst region, southwest China,” Acta Ecol. Sin. 35 (17), 5822–5833 (2015). https://doi.org/10.5846/stxb201310192524
J. F. Wu, N. Liu, P. L. Hu, K. L. Wang, W. Zhang, and D. S. Zou, “Soil carbon and nitrogen dynamics during vegetation restoration and their responses to extreme water-logging disasters in a typical karst depression,” Chin. J. Eco-Agric. 28 (3), 429–437 (2020). https://doi.org/10.13930/j.cnki.cjea.190711
J. S. Wu, Q. M. Lin, Q. Y. Huang, and H. A. Xiao, Soil Microbial Biomass-Methods and Application (China Meteorological Press, Beijing, 2006), pp. 54–83.
K. N. Xiong, D. Y. Zhu, T. Peng, L. F. Yu, J. H. Xue, and P. Li, “Study on Ecological industry technology and demonstration for karst rocky desertification control of the karst Plateau-Gorge,” Acta Ecol. Sin. 36 (22), 7109–7113 (2016). https://doi.org/10.5846/stxb201610172104
H. F. Xu, X. T. Liu, and J. H. Bai, “Dynamic change and environmental effects of soil microorganism in marsh soils from Carex meyeriana wetlands in Changbai Mountain,” J. Soil Water Conserv. 18 (3), 115–117 (2004). https://doi.org/10.3321/j.issn:1009-2242.2004.03.029
X. Yan and Y. L. Cai, “Multi-scale anthropogenic driving forces of karst rocky desertification in Southwest China,” Land Degrad. Dev. 26 (2), 193–200 (2015). https://doi.org/10.1002/ldr.2209
X. Yang, Z. Jia, and L. Ci, “Assessing effects of afforestation projects in China,” Nature 466 (7304), 315 (2010). https://doi.org/10.1038/466315c
Y. Y. Ye, S. J. Liu, W. Zhang, S. Y. Shu, S. Yang, and K. L. Wang, “Dynamics of soil microbial biomass and soil enzyme activity along a vegetation restoration gradient in a karst peak-cluster depression area,” Acta Ecol. Sin. 35 (21), 6974–6982 (2015). https://doi.org/10.5846/stxb201402150260
D. X. Yuan, “Rock desertification in the subtropical karst of south China,” Z. Geomorphol., Suppl. 108, 81–90 (1997).
W. Zech, N. Senesi, G. Guggenberger, K. Kaiser, J. Lehmann, T. M. Miano, A. Miltner, and G. Schroth, “Factors controlling humification and mineralization of soil organic matter in the tropics,” Geoderma 79 (1–4), 117–161 (1997). https://doi.org/10.1016/S0016-7061(97)00040-2
L. Q. Zhang, W. X. Peng, T. Q. Song, D. S. Zou, F. P. Zeng, M. Song, Z. Yu, and Y. Liu, “Spatial heterogeneity of soil microbial biomass carbon, nitrogen, and phosphorus in sloping farmland in a karst region on the Yunnan-Guizhou Plateau,” Acta Ecol. Sin. 32 (7), 2056–2065 (2012). https://doi.org/10.5846/stxb201108171204
T. Zhao, Y. L. Jiang, H. Yan, Y. M. Huang, and S. S. An, “Effects of different aspects on soil microbial biomass and dissolved organic carbon of the loess hilly area,” Environ. Sci. 34 (8), 3223–3230 (2013). https://doi.org/10.13227/j.hjkx.2013.08.014
Z. Zhu, S. Piao, R. B. Myneni, M. Huang, Z. Zeng, J. G. Canadell, P. Ciais, S. Sitch, et al., “Greening of the Earth and its drivers,” Nat. Clim. Change 6 (8), 791–795 (2016). https://doi.org/10.1038/nclimate3004
ACKNOWLEDGMENTS
We thank AJE for English language editing and Sci-Tech Innovation for experimental technical support.
Funding
This study was financially supported by the Key Project of Science and Technology Program of Guizhou Province: Poverty Alleviation Model and Technology Demonstration for Ecoindustries Derived from the karst desertification control (No. 5411 2017 Qiankehe Pingtai Rencai); the National Top Discipline Program of Guizhou Province: Geography in Guizhou Normal University (85 2017 Qianjiao Keyan Fa); and Youth Foundation of Guizhou Academy of Science (2020 [18] Qianke yuan).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Rights and permissions
About this article
Cite this article
Yang Cao, Xiong, Kn., Lv, Xx. et al. Effects of Different Vegetation Restoration Modes on Soil Microbial Quantity and Biomass in a Degraded Karst Ecosystem in South China. Eurasian Soil Sc. 54, 2009–2021 (2021). https://doi.org/10.1134/S1064229321130019
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1064229321130019