Abstract
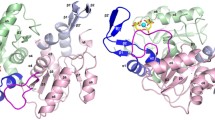
Uridine phosphorylases play an essential role in the cellular metabolism of some antibacterial agents. Acute infectious diseases (bubonic plague, yersiniosis, pseudotuberculosis, etc., caused by bacteria of the genus Yersinia) are treated using both sulfanilamide medicines and antibiotics, including trimethoprim. The action of an antibiotic on a bacterial cell is determined primarily by the character of its interactions with cellular components, including those which are not targets (for example, with pyrimidine phosphorylases). This type of interaction should be taken into account in designing drugs. The three-dimensional structure of uridine phosphorylase from the bacterium Yersinia pseudotuberculosis (YptUPh) with the free active site was determined for the first time by X-ray crystallography and refined at 1.40 Å resolution (DPI = 0.062 Å; ID PDB: 4OF4). The structure of the complex of YptUPh with the bacteriostatic drug trimethoprim was studied by molecular docking and molecular dynamics methods. The trimethoprim molecule was shown to be buffered by the enzyme YptUPh, resulting in a decrease in the efficiency of the treatment of infectious diseases caused by bacteria of the genus Yersinia with trimethoprim.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
C. Carnoy, N. Lemaitre, and M. Simonet, The Comprehensive Sourcebook of Bacterial Protein Toxins (Elsevier, Burlington, 2006).
R. Robins-Browne and E. Hartland, Yersinia Species. InternationalHandbook of Foodborne Pathogens (Marcel Dekker, New York, 2003).
J. F. MacFaddin, Biochemical Tests for Identification of Medical Bacteria (Williams & Wilkins, 1980).
M. B. Prentice, Plague and Other Yersinia Infections. Harrison’s Principles of Internal Medicine (1950).
A. N. Kravchenko and B. N. Mishan’kin, Antibiot Khimioter. 37 (1), 17 (1992).
B. N. Mishan’kan, A. N. Kravchenko, and V. G. Maiskii, Z. Mikrobiol. Epidemiol. Immunobiol., No. 2, 24 (1989).
O. K. Molchan, N. A. Dmitrieva, D. V. Romanova, et al., Biochemistry (Mosc). 63 (2), 195 (1998).
M. Achtman, K. Zurth, G. Morelli, et al., Proc. Natl. Acad. Sci. USA 96 (24), 14043 (1999).
M. Zolotukhina, I. Ovcharova, S. Eremina, et al., Res. Microbiol. 154 (7), 510 (2003).
W. Kabsch, International Tables for Crystallography (Kluwer, Dordrecht, 2001).
P. R. Evans, Acta Crystallogr. D 67 (4), 282 (2011).
M. D. Winn, C. C. Ballard, K. D. Cowtan, et al., Acta Crystallogr. D 67 (4), 235 (2011).
A. J. McCoy, Acta Crystallogr. D 63 (1), 32 (2007).
P. D. Adams, P. V. Afonine, G. Bunkoczi, et al., Acta Crystallogr. D 66 (2), 213 (2010).
G. N. Murshudov, A. A. Vagin, and E. J. Dodson, Acta Crystallogr. D 53 (3), 240 (1997).
P. Emsley and K. Cowtan, Acta Crystallogr. D 60 (12), 2126 (2004).
P. Emsley, B. Lohkamp, W. G. Scott, et al., Acta Crystallogr. D 66 (4), 486 (2010).
I. W. Davis, A. Leaver-Fay, V. B. Chen, et al., Nucl. Acids Res. 35, 375 (2007).
R. A. Friesner, J. L. Banks, R. B. Murphy, et al., J. Med. Chem. 47 (7), 1739 (2004).
T. A. Halgren, R. B. Murphy, R. A. Friesner, et al., J. Med. Chem. 47 (7), 1750 (2004).
D. Van Der Spoel, E. Lindahl, B. Hess, et al., J. Comput. Chem. 26 (16), 1701 (2005).
T. Schlesier and G. Diezemann, J. Phys. Chem. B 117 (6), 1862 (2013).
A. A. Lashkov, N. E. Zhukhlistova, A. G. Gabdulkhakov, et al., Crystallogr. Rep. 54 (2), 267 (2009).
M. V. Dontsova, A. G. Gabdoulkhakov, O. K. Molchan, et al., Acta Crystallogr. F 61 (4), 337 (2005).
T. T. Caradoc-Davies, S. M. Cutfield, I. L. Lamont, et al., J. Mol. Biol. 337 (2), 337 (2004).
S. T. Rao and M. G. Rossmann, J. Mol. Biol. 76 (2), 241 (1973).
A. A. Lashkov, N. E. Zhukhlistova, A. H. Gabdoulkhakov, et al., Acta Crystallogr. D 66 (1), 51 (2010).
W. L. DeLano, The PyMOL Molecular Graphics System (DeLano Scientific, San Carlos, CA, 2002).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V.V. Balaev, A.A. Lashkov, A.G. Gabdulkhakov, M.V. Dontsova, A.S. Mironov, C. Betzel, A.M. Mikhailov, 2015, published in Kristallografiya, 2015, Vol. 60, No. 4, pp. 579–585.
Rights and permissions
About this article
Cite this article
Balaev, V.V., Lashkov, A.A., Gabdulkhakov, A.G. et al. Three-dimensional structures of unligated uridine phosphorylase from Yersinia pseudotuberculosis at 1.4 Å resolution and its complex with an antibacterial drug. Crystallogr. Rep. 60, 525–531 (2015). https://doi.org/10.1134/S1063774515040069
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1063774515040069