Abstract
The composition of polar lipid classes and the composition of fatty acids of neutral and polar lipid fractions were studied in tissues of the brown alga Undaria pinnatifida infected with the endophyte Laminariocolax aecidioides. For the first time, the lipid glucuronosyldiacylglycerol was found in brown algae; while ceramide phosphoinositol, diacylglyceryltrimethylhomoserine, and diacylglycerylhydroxymethyltrimethyl-β-alanine were also found for the first time in significant amounts in algae of the order Laminariales. It has been established that presence of the endophyte leads to a decrease in the contents of phosphatidylinositol and ceramide phosphoinositol, to a significant increase in the concentration of saturated fatty acids, and to a decrease in the content of ω3 polyunsaturated acids in neutral lipids.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
INTRODUCTION
The annual brown alga Undaria pinnatifida (Harvey) Suringar, 1873 grows in the sublittoral zone at a depth of 1 to 15 m. Populations of U. pinnatifida have been found off the coasts of Japan, China, Korea, France, New Zealand, Tasmania, Argentina, England, and southern Australia, as well as in the Channel Islands [19, 24]. In Russian waters, U. pinnatifida has been found in the Peter the Great Bay of the Sea of Japan [21]. Because of its high contents of biologically active substances this alga is of great interest for biomedicine and pharmaceuticals [3, 7, 10, 20]. High concentrations of essential polyunsaturated fatty acids allow us to consider U. pinnatifida as a potentially important source of lipids [25].
Under natural conditions, U. pinnatifida sporophytes are often infected with the endophytic brown alga Laminariocolax aecidioides (Rosenvinge) A.F. Peters, 1998 (Ectocarpales: Phaeophyceae) [9, 22]. A few studies have shown that infection with this endophyte does not affect the reproductive potential and development of U. pinnatifida. However, perforations in tissues of the infected macrophytic alga can lead to secondary infection of macrophytes with bacterial or other infections, which can cause the death of the alga [2]. Epiphytic growth in cultivated algae is a serious problem leading to a decrease in the quantity and quality of production [26].
Comprehensive studies of endophytes of brown algae are extremely scarce. There is no information on the biochemical composition, in particular lipid composition, as well as on the mutual influence of the endophyte and the host. This study deals with investigation of the effect of the endophyte L. aecidioides on the lipid composition of the host macrophyte U. pinnatifida.
MATERIALS AND METHODS
Algal samples were collected in November 2020 in the Sobol Bay (Peter the Great Bay, Sea of Japan) at a water temperature of 5°C. Three infected and three uninfected specimens of Undaria pinnatifida were selected; tissue samples (weight of 0.2–0.5 g) were taken from each plant from the lower part of the plate, from intact areas of the upper part of the plate and from affected clearly visible pigmented areas in infected plants. The tissue pieces were quickly dried with filter paper, weighed, and homogenized in 5 mL of a mixture of chloroform : methanol (1 : 1 by volume); 1 mL of water was added to the homogenate, thoroughly mixed and centrifuged for 5 min at 5000 rpm. The lower layer was collected, evaporated to a constant weight, and redissolved in chloroform.
The lipid extract was separated into fractions of polar and neutral lipids on a silica gel column (100–160 μm), 1 cm in diameter and 3 cm high. 100 μL of the extract (concentration 10 mg/mL) was applied to the column; neutral lipids were eluted with pure chloroform, polar lipids were eluted with a mixture of methanol : water (40 : 1) (three times at 1 mL with each eluent). Fatty acid methyl esters were obtained by hydrolysis of lipid fractions in methanol with 2% sulfuric acid at a temperature of 90°C for 1 h. Analysis of fatty acid methyl esters was carried out on a Shimadzu GC-2010 gas chromatograph with a flame ionization detector on a Supelcowax-10 column 30 m × 0.25 mm (Supelco, United States) in isothermal mode at 200°C. Methyl esters of fatty acids were identified by calculating the equivalent chain length [12].
Polar lipid classes were analyzed by high performance liquid chromatography on an Ascentis Si 25 cm × 2.1 mm column (Supelco, United States) in the hydrophilic chromatography mode (Fig. 1). The following compositions were used as eluents: A, acetonitrile + 50 mM formic acid and B, acetonitrile : water (1 : 1 by volume) + 100 mM formic acid + 40 mM ammonia. Gradient elution was carried out according to the following program: start (2% B, total flow 0.2 ml/min), 2.5 min (24% B), 3 min (flow decrease from 0.2 to 0.15 mL/min), 5 min (26% B), 10 min (40% B), 15 min (100% B), 19 min (flow increase from 0.15 to 0.3 mL/min), 21 min (concentration decrease from 100 to 2% B), 24 min (flow decrease from 0.3 to 0.2 mL/min) and 26 min (stop).
Separation of classes of polar lipids by HPLC-MS/MS in the lipid extract of the upper part of the plate of an infected sample of Undaria pinnatifida. An overlay of chromatograms of the specific fragmentation reactions for each class is presented. To equalize the intensity, the chromatograms are shown at different scales.
Polar lipid classes were detected using a Shimadzu LCMS-8060 triple quadrupole mass spectrometer with electrostatic sputter ionization. The ionizer parameters were: evaporator gas flow (nitrogen), 3 L/min; drying gas (nitrogen), 10 L/min; auxiliary heating gas (air), 10 L/min; evaporation capillary temperature, 300°C; desolvation line temperature, 250°C; and heating block temperature, 400°C. Each lipid class was detected by its specific fragmentation reaction, which presents a loss of a certain neutral fragment or formation of a daughter ion (Table 1). In this case, the scanning rate of the first quadrupole was 3000 Da/s and the time for the complete cycle of fragmentation of all lipid classes was 2 s. Appropriate lipid standards from Avanti Polar Lipids (United States) were used for identification and optimization of fragmentation parameters and for construction of calibration plots. Because of the lack of standards for glucuronosyldiacylglycerol (GlcADG), diacylglycerylhydroxymethyltrimethyl-β-alanine (DGTA), and phosphatidylhydroxyethylglycine (PHEG), the calibration data for sulfoquinovosyldiacylglycerol (SQDG), diacylglyceryltrimethylhomoserine (DGTS), and phosphatidylserine, were used, respectively, to calculate the lipid contents of these classes. GlcADG [18], DGTA [16], PHEG and ceramide phosphoinositol (CPI) [27] were identified basing on previously published data on the mechanisms of fragmentation. The mass spectra of decomposition and the mechanism of fragmentation of GlcADG, a lipid discovered in macrophytic algae for the first time, are given in Fig. 2.
Statistical processing was carried out in the Microsoft Excel program and the Student’s t-test was used to assess the significance of the changes. Changes with a significance level of p < 0.05 were considered significant.
RESULTS
The Compositions of Polar Lipids
The analysis of the tissues of Undaria pinnatifida made it possible to detect glycolipids: monogalactosyldiacylglycerol (MGDG), digalactosyldiacylglycerol (DGDG), SQDG, and GlcADG); phospholipids: phosphatidylcholine (PC), phosphatidylethanolamine (PE), phosphatidylinositol (PI), ceramide phosphoinositol (CPI), phosphatidylglycerol (PG), and PHEG; and betaine lipids: DGTS and DGTA (Table 2). The contents of MGDG (25.7–28.7%) and SQDG (9.7–11.1%) remained at the same level in all studied tissues. The content of DGDG in the lower part of the plate was noticeably lower than in the upper part (14.7−16.8% vs. 20.9−23.3%).
The contents of the majority of U. pinnatifida phospholipids also did not practically change and included 10.4–14.2% PG, 9.1–10.9% PE, 7.5–10.3% PC, and 2.5–3.5% PHEG. An increased amount of PG was detected in the lower parts of the plates of infected plants (14.2% vs. 10.3–11.1% in other parts of the plates), an increased level of PC was found in the lower parts of uninfectedplants (10.3% vs. 7.5% in infected plants).
The contents of phospholipids with inositol in the composition of the polar group (PI and CPI) in the upper part of the plate of infected algae were lower than in uninfected plants, both in the tissue with clear signs of the presence of the endophyte (Ue) and in the adjacent tissue (Ui). For example, the content of PI decreased from 3.7 to 2.5–2.6%, while the content of CPI decreased from 2.1 to 1.0%. In the lower part of plates of infected plants, the content of PI was also lower: 3.5% vs. 4.2% in uninfectedsamples.
The content of betaine lipids DGTS and DGTA (1.5–3.2%) and the content of glycolipid GlcADG (0.5–1.3%) did not correlate with the presence of the endophyte.
We should note that no significant amount of lysolipids was detected, except for traces of lyso-MGDG in some samples.
The Composition of Fatty Acids
The main fatty acids of U. pinnatifida were 16:0, 18:0, 18:2n-6, 18:1n-9, 18:3n-3, 18:4n-3, 20:4n-6 and 20:5n-3, while the content of polyunsaturated fatty acids was significantly higher in the fraction of polar lipids (Table 3). In uninfected individuals the upper parts of the plates contained a higher level of eicosapentaenoic acid.
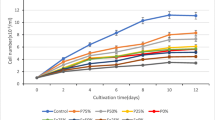
The algal tissue with an evident presence of endophyte (Ue) showed a significantly changed composition of fatty acids of polar and neutral lipids. This change was presented by an increase in the concentration of saturated FAs (16:0 and 18:0) and in a decrease in the level of polyunsaturated fatty acids of the ω3 series (18:3n-3, 18:4n-3, and 20:5n-3) (Fig. 3). Moreover, such changes in fatty acids of polar lipids were detected only in the part of the plate where the presence of an endophyte was visually evident, while this effect in neutral lipids was displayed in all parts of the plate.
The influence of an endophyte on the content of fatty acids in polar and neutral lipids from different parts of the plate of Undaria pinnatifida (% of the total of all FAs). L, lower and U, upper parts of the plate of healthy samples; Li, lower and Ui, upper parts of the plate of infected samples; Ue, the upper part of the plate of infected samples with visually evident presence of an endophyte on the surface of the algal plate.
DISCUSSION
Glycolipids, along with PG, form the basis of chloroplast membranes and are the main polar lipids of algae; therefore, intensely photosynthesizing tissues are characterized by a high content of plastid polar lipids. An earlier study on the brown alga Saccharina japonica from the order Laminariales revealed an increased content of DGDG and SQDG in old parts of the thallus; this can be explained by more intense photosynthesis in older parts than in young tissues [14]. The U. pinnatifida plate grows due to the intercalary meristem located at its base; therefore, the upper part of the plate is represented by an older tissue with active photosynthesis and, accordingly, with a higher level of DGDG. The increased content of eicosapentaenoic acid in the upper part of the plate can also be explained by more intense photosynthesis and de novo FA synthesis. The study on Laminaria japonica revealed a more general pattern, that is, an increased ω3/ω6 ratio of FAs in the upper parts of the thallus [14].
GlcADG was not found in macrophyte algae previously. The presence of GlcADG was shown for higher plants [18], unicellular algae [4], sea grasses [15], and in some bacteria and fungi [11]. The role of this lipid has not been yet elucidated, but it is known that GlcADG content increases during phosphorus starvation [18].
Only one study [27] found CPI in brown algae, this lipid is common for red algae, fungi and protozoa [13, 23]. It cannot be excluded that the presence of CPI indicates also the presence of fungi, and epi- or endophytic red algae, in the studied tissues of U. pinnatifida. However, a high degree of infection is usually characteristic of older tissues, but the data we obtained indicate the opposite: the level of CPI was higher in the lower and younger part of the plate, which indicates the endogenous origin of this lipid.
DGTA was found in many brown algae, but is not characteristic of the order Laminariales, while DGTS is generally rare in brown algae [1]. In algae of the order Laminariales, these lipids were found earlier only in trace amounts [5].
The most noticeable response to endophytic infection is an increase in the content of saturated fatty acids and a decrease in the level of polyunsaturated ω3 fatty acids in the fraction of neutral lipids. Previously, it was found that unicellular marine algae infected with a virus showed increased synthesis of triglycerides with saturated fatty acids, which accumulated in the form of fat droplets [17]. A decrease in the content of polyunsaturated fatty acids might be caused by destruction of FAs with reactive oxygen species formed in response to infection [6]. However, we found such changes in the FA composition of neutral lipids either at the site of direct infection or in the remote lower parts of the plate. It is known that the presence of an endophyte disrupts the integrity of the outer integuments; this opens access for secondary infections [8]. Thus, we suppose that the secondary viral or bacterial infection spread through the affected areas of the upper part of the plate throughout the entire algal plate and provoked changes in the FA composition of the tissues adjacent to the site of the direct exposure to the endophyte.
REFERENCES
Khotimchenko, S.V., Lipidy morskikh vodorosley-makrofitov i trav (Lipids of Marine Macrophytic Algae and Grasses), Vladivostok: Dal’nauka, 2003.
Campo, E. del, Garcia-Reina, G., and Correa, J.A., Degradative disease in Ulva rigida (Chlorophyceae) associated with Acrochaete geniculata (Chlorophyceae), J. Phycology, 1998, vol. 34, no. 1, pp. 160–166.
Cho, J.-Y., Kang, J.-Y., Khan, M.N.A., et al., Anti-inflammatory activities of Undaria pinnatifida and Laminaria japonica (Phaeophyta), J. Fish. Sci. Technol., 2007, vol. 10, no. 3, pp. 127–132.
Eichenberger, W., Araki, S., and Müller, D.G., Betaine lipids and phospholipids in brown algae, Phytochemistry, 1991, vol. 34, no. 5, pp. 1323–1333.
Eichenberger, W. and Gribi, C., Diacylglyceryl-α-D-glucuronide from Ochromonas danica (Chrysophyceae), J. Plant Physiol., 1994, vol. 144, no. 3, pp. 272–276.
Evans, C., Pond, D.W., and Wilson, W.H., Changes in Emiliania huxleyi fatty acid profiles during infection with E. huxleyi virus 86: Physiological and ecological implications, Aquat. Microb. Ecol., 2009, vol. 55, no. 3, pp. 219–228.
Faggio, C., Morabito, M., Minicante, S.A., et al., Potential use of polysaccharides from the brown alga Undaria pinnatifida as anticoagulants, Braz. Arch. Biol. Technol., 2015, vol. 58, no. 5, pp. 798–804. https://doi.org/10.1590/S1516-8913201500400
Gao, X., Ogandaga, C.A.M., Park, S.K, et al., Algal endophytes of commercial Chondrus ocellatus (Gigartinaceae, Rhodophyta) from different wild populations in Korea, J. Appl. Phycol., 2020, vol. 32, no. 1, pp. 697–703.
Gauna, M.C., Parodi, E.R., and Caceres, E.J., Epi-endophytic symbiosis between Laminariocolax aecidioides (Ectocarpales, Phaeophyceae) and Undaria pinnatifida (Laminariales, Phaeophyceae) growing on Argentinian coasts, J. Appl. Phycol., 2009, vol. 21, no. 1, pp. 11–18.
Hayashi, K., Nakano, T., Hashimoto, M., et al., Defensive effects of a fucoidan from brown alga Undaria pinnatifida against herpes simplex virus infection, Int. Immunopharmacol., 2008, vol. 8, no. 1, pp. 109–116.
Hölzl, G. and Dörmann, P., Structure and function of glycoglycerolipids in plants and bacteria, Prog. Lipid Res., 2007, vol. 46, no. 5, pp. 225–243.
Jamieson, G.R., GLC identification techniques for long-chain unsaturated fatty acids, J. Chromatogr. Sci., 1975, vol. 13, no. 10, pp. 491–497.
Kaneshiro, E.S., Jayasimhulu, K., and Lester, R.L., Characterization of inositol lipids from Leishmania donovani promastigotes: Identification of an inositol sphingophospholipid, J. Lipid Res., 1986, vol. 27, no. 12, pp. 1294–1303.
Khotimchenko, S.V. and Kulikova, I.V., Lipids of different parts of the lamina of Laminaria japonica Aresch., Bot. Mar., 2000, vol. 43, no. 1, pp. 87–91.
Koelmel, J.P., Campbell, J.E., Guingab-Cagmat, J., et al., Re-modeling of foliar membrane lipids in a seagrass allows for growth in phosphorus-deplete conditions, PLoS One, 2019, vol. 14, no. 11, e0218690.
Li, Y., Lou, Y., Mu, T., et al., Simultaneous structural identification of diacylglyceryl-N-trimethylhomoserine (DGTS) and diacylglycerylhydroxymethyl-N,N,N-trimethyl-β-alanine (DGTA) in microalgae using dual Li+/H+ adduct ion mode by ultra-performance liquid chromatography/quadrupole time-of-flight mass spectrometry, Rapid Commun. Mass Spectrom., 2017, vol. 31, no. 5, pp. 457–468. https://doi.org/10.1002/rcm.7818
Malitsky, S., Ziv, C., Rosenwasser S., et al., Viral infection of the marine alga Emiliania huxleyi triggers lipidome remodeling and induces the production of highly saturated triacylglycerol, New Phytol., 2016, vol. 210, no. 1, pp. 88–96.
Okazaki, Y., Otsuki, H., and Nishizawa, T., A new class of plant lipid is essential for protection against phosphorus depletion, Nat. Commun., 2013, vol. 4, no. 1, 1510.
Pereira, R. and Yarish, C., Mass production of marine macroalgae, in Encyclopedia of Ecology, Jorgensen, S.E. and Fath, B.D., Eds., Oxford: Elsevier, 2008, pp. 2236–2247.
Shibata, T., Ishimaru, K., Kawaguchi, S., et al., Antioxidant activities of phlorotannins isolated from Japanese Laminariaceae, J. Appl. Phycol., 2008, vol. 20, no. 5, 705.
Skriptsova, A., Khomenko, V., and Isakov, V.I., Seasonal changes in growth rate, morphology and alginate content in Undaria pinnatifida at the northern limit in the Sea of Japan (Russia), J. Appl. Phycol., 2004, vol. 16, no. 1, 17.
Skriptsova, A. V. and Kalita, T.L., The first record of the brown endophytic alga Laminariocolax aecidioides (Rosenvinge) A.F. Peters, 1998 in the Russian Far-Eastern Seas, Russ. J. Mar. Biol., 2020, vol. 46, no. 1, pp. 42–48.
Smith, S.W. and Lester, R.L., Inositol phosphorylceramide, a novel substance and the chief member of a major group of yeast sphingolipids containing a single inositol phosphate, J. Biol. Chem., 1974, vol. 249, no. 11, pp. 3395–3405.
Stuart, M.D., Hurd, C.L., and Brown, M.T., Effects of seasonal growth rate on morphological variation of Undaria pinnatifida (Alariaceae, Phaeophyceae), Hydrobiologia, 1999, vol. 398, no. 1911, pp. 191–199.
Tabakaeva, O. V. and Tabakaev, A. V., Compositions of lipids and fatty acids from various parts of the brown alga Undaria pinnatifida, Chem. Nat. Compd., 2017, vol. 53, no. 5, pp. 843–848.
Titlyanov, E.A. and Titlyanova, T. V., Seaweed cultivation: Methods and problems, Russ. J. Mar. Biol., 2010, vol. 36, no. 4, pp. 227–242.
Vyssotski, M., Lagutin, K., MacKenzie, A., et al., Phospholipids of New Zealand edible brown algae, Lipids, 2017, vol. 52, no. 7, pp. 629–639.
ACKNOWLEDGMENTS
The authors are grateful to Dr. Anna V. Skriptsova (Laboratory of Physiology of Autotrophic Organisms of the National Scientific Center of Marine Biology, Far Eastern Branch, Russian Academy of Sciences) for her help in identification of algae.
Funding
The work was carried out with the financial support of the Russian Foundation for Basic Research (project no. 20-34-90112).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest. The authors declare that they have no conflicts of interest.
Statement on the welfare of animals. This article does not describe any research using humans and animals as objects.
Additional information
Translated by I. Barsegova
Rights and permissions
About this article
Cite this article
Chadova, O.A., Velansky, P.V. The Influence of Endophyte Laminariocolax aecidioides (Rosenvinge) A.F. Peters, 1998 (Phaeophyceae, Ectocarpales) on the Lipid Composition of the Brown Alga Undaria pinnatifida (Harvey) Suringar, 1873 (Phaeophyceae: Laminariales). Russ J Mar Biol 48, 373–379 (2022). https://doi.org/10.1134/S1063074022040034
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1063074022040034