Abstract—The results of a study of delayed changes in the frequency of unstable chromosomal aberrations and the CNA- and LOH-genetic landscape of blood leukocytes in healthy employees at the main facilities of the Siberian Group of Chemical Enterprises exposed to external irradiation are presented. In 2014, 11 persons with de novo induced mosaic deletions and amplifications associated with dicentric and/or ring chromosomes (markers for individual radiosensitivity) were detected. Three years later, the frequency levels of chromosomal aberrations were compared. In 2017 the frequency of aberrant cells had increased by 1.7 times compared to 2014, the frequency of the chromosomal type aberrations had doubled, the frequency of pair fragments had a more than fourfold increase, and the frequency of dicentrics had almost doubled. The mosaic deletion in employee no. 1490 (3q12.3–13.11) identified in 2014 persisted until 2017. Mosaic amplifications identified in 2014 were preserved in five out of six employees (nos. 178, 203, 450, 1620, and 1792). Moreover, we identified that the CNstate in employee no. 450 had increased 1.5 times by 2017. The mosaic amplification of the short arm of the third chromosome in employee no. 278 had been eliminated by 2017. Other changes in the CNA and LOH-genetic landscape were not detected. Thus, these data indicate the formation of self-sustained bone marrow clones, whose descendants produce a population of aberrant cells in the blood of irradiated individuals. Further research into the preservation of chromosomal aberrations and CNA is needed, as well as studies aimed at detecting the clones of aberrant cells in the bone marrow.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
INTRODUCTION
In 2014, at Seversk Biophysical Research Centre of the Russian Federal Medical and Biological Agency (SBR Center), using the CytoScan™ HD Array microarray a study was conducted on the blood lymphocyte DNA of 46 employees of the Siberian Chemical Combine (SCC) who had been exposed to external irradiation during professional activities of. In the recent past, SCC was one of the world’s largest complexes of nuclear industry enterprises. Mosaic amplifications were identified in nine employees and mosaic deletions in two employees of the total number of subjects. It was shown that mosaic deletions and amplifications of chromosomal loci in individual employees can be detected even with external doses of just over 100 mSv [1]. Deletions and amplifications are CNA (copy number aberrations, or induced de novo CNV—copy number variations), resulting from repair errors [2] and can affect the expression of genes localized in regions with CNA [3, 4]. Ionizing radiation causes single-stranded and double-stranded breaks; if the replicative fork comes into contact with a single-stranded break, this can lead to replication collapse, new replicative forks and the formation of CNA, including the FoSTes (fork stalling and template switching) mechanism [5].
A 2014 study suggested that mosaic CNA could be formed in mitotically active cells in a human body exposed to radiation (e.g., in bone marrow cells). As a result, CNA clones formed in the bone marrow and their descendant leukocytes accumulated in the blood of irradiated individuals. If this is the case, then the representation of the clone with the mutation can change over time, both in the direction of decrease, and increase. The same applies to unstable chromosomal aberrations (CA), determined by routine methods. In this study, we studied the change over the past 3 years in the frequency of unstable CA and the genetic landscape (the appearance of new CNA and loss of heterozygosity sites (LOH), the preservation of mosaic deletions and amplifications, and the presence of mutant clones) in the blood leukocytes of employees exposed to prolonged radiation exposure.
MATERIALS AND METHODS
The study included seven of 11 healthy SCC employees who, during their professional activities, were exposed to gamma radiation. In these employees, according to the results of the 2014 studies, de novo induced mosaic deletions and amplifications were detected. The study was carried out in accordance with the 1964 Helsinki Declaration (as amended 1975–2013) and with the permission of the local ethics committee of the SBR Center. Informed consent was obtained from all SCC employees participating in this study.
Three employees refused the reinvestigation, and one worker died before the 2017 research phase. The average dose of seven of the examined employees in 2014 was 361.63 mSv, in 3 years this dose had not changed since they had all retired. The individual radiation doses of each employee in 2014 and 2017 are in Table 1.
Blood was used for standard cytogenetic analysis and DNA isolation for subsequent microarray analysis. For all examined individuals, a standard cytogenetic analysis of peripheral blood lymphocytes was performed as described in detail in the 2014 study [6]. Routine staining of chromosomes was performed with Giemsa dye prepared in a phosphate buffer. Chromosomal analysis was performed using a Leica DM2500 microscope (Germany). At least 300 metaphases were studied for each individual. All types of chromosome aberrations recognized without karyotyping were analyzed. The frequency of aberrant cells and all types of CA was calculated per 100 metaphases.
DNA was isolated from blood using a QIAamp DNA Blood mini Kit (Qiagen, Germany). The concentration and purity of DNA isolation were evaluated on a NanoDrop-2000 spectrophotometer (Thermo Scientific, United States). The concentration was from 50 to 150 ng/μL, А260/А280 = 2.10–2.35, А260/А230 = 2.15–2.40. DNA integrity was assessed by capillary electrophoresis on a TapeStation instrument (Agilent Technologies, United States) using an Agilent Genomic DNA ScreenTape System Quick Guide kit (Agilent Technologies, United States). Most DNA fragments had a weight of more than 48 kb. The isolated DNA was stored at –80°С in a low-temperature Sanyo freezer (Japan) and was used for microarray studies.
Microarray analysis was performed on high-density microarrays (DNA chips) of an Affymetrix (United States) CytoScan™ HD Array (http://www.affymetrix.com/ esearch/search.jsp?pd=prod520004&N=4294967292). Sample preparation, hybridization, and scanning procedures were performed in accordance with the manufacturer’s protocol using the Affymetrix GeneChip® SCNAner 3000 7G system (Affymetrix, United States).
The microchip results were processed using the Chromosome Analysis Suite 3.0 program (Affymetrix, United States), which was developed specifically for analyzing the results of studies using the CytoScan™ HD Array matrix. In all cases, the snpQC was higher than 15. Using the program, a CNA loss and gain of at least 100 kb and loss mosaic and gain mosaic that formed the CNA genetic landscape of blood leukocytes were determined in the chromosomes. For analysis of the representation of mosaic CNA, the CNstate index determined in the software was used. In addition, the sites with LOH forming the LOH landscape were determined. Changes in the LOH landscape can serve as a marker of emerging genomic cell instability [7]. All these data were compared with the results of microarray scanning in 2014.
Statistical analysis was performed using the STATISTICA 8.0 software package (StatSoft Inc., United States); for comparison of groups, the nonparametric Mann–Whitney U test was used.
RESULTS
At the first stage of the study, the frequency of CA was determined in employees with mosaic CNA identified in 2017 and compared with that recorded in 2014, both at the individual level (Table 2) and for the group on average (Table 3). In general, there was a statistically significant increase in the average frequencies of aberrant cells, chromosome-type aberrations (2‑fold increase), paired fragments, and dicentric chromosomes, but not ring chromosomes (Table 3).
Despite the fact that the total radiation dose of the employees examined in 2017 did not change compared to that of 2014 (Table 1), in four employees (nos. 178, 278, 1490, 1620) the frequency of aberrant cells increased by 2.4–3.4 times, while in employees nos. 203, 450, 1792, the frequency of such cells remained virtually unchanged (Table 2). In employee no. 203 the ring chromosomes were eliminated, but the frequency of dicentric chromosomes increased; in employees nos. 178, 278, 1490 the level of both markers of radiation exposure (ring and dicentric chromosomes) increased; in employees nos. 450 and 1620, the frequency of ring chromosomes increased. Only in employee no. 1792, the level of ring chromosomes decreased over 3 years (Table 2).
Thus, in the examined SCC employees, significant individual changes in the frequency of CA over time were observed, which might testify to the assumption about the formation of aberrant bone marrow clones.
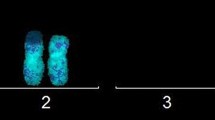
Using the full-genome CytoScan™ HD Array microarray, we studied the CNA and LOH genetic landscapes of employee white blood cells. When comparing the results of the full-genome CNA scans obtained in 2014 and 2017, we can conclude that in the examined employees any new CNA were not revealed, except for the previously identified mosaic CNA (see below). There were no cases of chromothripsis and kataegis on any of the chromosomes. Changes in the LOH sites were not detected, the LOH landscape completely coincided in 2014 and 2017. The LOH landscape of 2014 and 2017 is shown for employee no. 278 as an example (Fig. 1).
In the 2014 study, mosaic deletion (mosaic loss 3q12.3–13.11) was found in employee no. 1490 [1]. A second study showed that the mosaic deletion was preserved and the representation of the mutant CNstate clone remained virtually unchanged (29 and 28%) (Fig. 2).
Earlier in employees no. 178 (22q11.23–12.1), no. 203 (11p11.12), no. 278 (3p14.2–14.1), no. 1620 (22q11.23–12.1), no. 1792 (15q11.2) and no. 450 (22q13.31) mosaic amplifications were detected. In repeat study, it was found that the mosaic amplifications were preserved in five employees (Figs. 3–7), and their CNstate representation did not change. In employee no. 278, the mosaic amplification of the short arm of chromosome 3 was eliminated (Fig. 1). It should be noted that the CA frequency, determined by routine methods, in this employee, increased by 3.3 times (Table 2) and changes in the LOH landscape as a marker of genomic instability were not detected (Fig. 1).
Interesting data were obtained for employee no. 450. According to the 2014 study, mosaic amplification of the 22nd chromosome (2044 ± 514 kb) was established. In 2017, it was found that CNState increased by 1.5 times (from 26 to 40%) (Fig. 7).
Thus, in six of the seven examined employees, mosaic deletion and mosaic amplifications were preserved; their representation in 5/6 cases did not change, and in only one case increased by 1.5 times. One case of elimination of mosaic amplification over time was also noted.
DISCUSSION
It is believed that unstable CA determined by routine methods (in particular, dicentrics) is eliminated during the division process. As was found, in some of the examined employees, the frequency of CA did not significantly change over time, or certain CA types were eliminated. This is a natural phenomenon, and it can be explained by the restoration of the body after radiation exposure. It is much more difficult to explain the observed significant increase in CA frequency after retirement from work with radiation exposure (Table 3). The vast majority of studies indicate a decrease in the CA level in employees after the cessation of radiation exposure, but among the liquidators of the Chernobyl accident, professionals working with sources of ionizing radiation, and in patients with chronic and acute radiation sickness, they can persist for a long time [8, 10–12]. In other words, despite the fact that unstable CA must be eliminated, the population of aberrant cells in the blood is maintained for a long time. The increase in the CA frequency cannot be explained by age-related changes, since only 3 years had passed. However, the possibility should not be excluded that during this time the examined employees may have received additional radiation exposure due to medical exposure, data on which were not available, and therefore it is not possible to quantify and individually assess. It should also be noted that a significant increase in radiation load due to medical exposure occurs in people during the diagnosis and treatment of cancer or cardiovascular disease. According to the results of medical examinations, the employees were healthy (did not have oncological and/or cardiovascular diseases). In the examined employees, aberrant cells not only persist for a long time (at least for 3 years), but their frequency increases, e.g. they appear de novo. It can be assumed that prolonged radiation exposure led to the formation of genomic instability in bone marrow cells, as a result of which there is a constant induction of new unstable CA. If this assumption is true, then, since genomic instability is nonspecific, in addition to unstable CA determined by routine methods, other types of aberrations should also be observed, such as copy number aberrations, the appearance of new LOH sites, chromothripsis, and kataegis.
Chromothripsis is a mutagenic process in which one or more chromosome region undergoes catastrophic destruction, followed by accidental repair of DNA fragments. This process leads to genomes in which tens or hundreds of genomic permutations affect one or more segments of chromosomes. Recently, it has been shown that chromothripsis is one of the results of a telomere crisis [7]. Kataegis is a recently discovered phenomenon describing regions of localized hypermutations in which multiple substitutions of C : G pairs spaced several hundred nucleotides apart are grouped by kilobase-sized regions, often associated with DNA rearrangement sites. It is believed that kataegis is the result of the activity of the apolipoprotein B catalytic subunit (APOBEC) enzyme family, which can deaminate cytosine residues for the formation of uracil [7, 9].
Chromothripsis and kataegis cannot be detected using routine methods, but they are easily detected using a polygenic SNP array study (in the form of multiple CNA on one or several chromosomes for chromothripsis, and an increase in the length of the copies of neutral LOH during kataegis). Our study using a polygenic microarray showed no changes in the CNA and LOH landscape of blood leukocytes over 3 years. There was not a single case of chromothripsis and kataegis. This placed in doubt the assumption about the formation of genomic instability in bone marrow cells, which is responsible for increasing the CA frequency.
The second assumption about the nature of the increase in the CA frequency in employees not exposed to additional radiation exposure is that, the elimination of unstable CA during division does not always occur. The increase in the CA frequency is associated with the formation of clones of aberrant cells in the bone marrow, in which the unstable CA are not eliminated (identification of the mechanisms involved awaits further study) in the process of division, and they are preserved in the blood leukocyte descendants of this clone. In this case, there will be no signs of genomic instability, the CA frequency will increase in some cases, and the representation of the detected mosaic CNA may also decrease or increase. As our studies have shown, after 3 years, employees had no signs of genomic instability, and CA frequency changes. Most importantly, it was found that the representation of mosaic amplification can increase or decrease to zero. These data support the assumption about the formation of mutant clones in the bone marrow of long-exposed individuals, which are the source of aberrant cells in the blood. The mutant clone of employee no. 450 has not only existed for a long time, but multiplies and its representation in the blood increases, which may indicate a high risk of developing a disease of the hemoblastoses group in this employee.
In general, almost all regions whose mosaic CNA are preserved in the blood leukocytes of irradiated employees are somehow related to the development of hemoblastoses, as evidenced by the literature. In a study by T. Pierini et al. (2016) it was shown that the basis of leukemic cell transformation is a complex rearrangement of chromosomes, including chromosome 22 [13]. In 2007, 35 cases of mantle cell lymphoma characterized by extensive anomalies distributed throughout the genome, including deletions of 22q11.23–13q.33 were described [14]. Deletions and amplifications of the long arm of the 22nd chromosome have also been described in several studies of T‑cell prolymphocytic leukemia [15, 16]. In a study of mantle cell lymphoma, published in 1998, it was found that two of the most common aberrations in this disease are amplifications of 3q and 15q. The same study showed that in five cases of lymphomas highly amplified regions, including the region of interest 3p14–22 were present [17]. In the case of angioimmunoblastic T-cell lymphoma, the most frequent amplifications were localized in regions 22q and 11p11–q14. Amplifications of chromosomes 3 and 15, as well as the region 11p11.2–11q13.5, were less common. In two cases, a deletion was recorded on chromosome 3, including 3q13.3 [18].
There is also evidence showing that about a quarter of patients diagnosed with diffuse large B-cell lymphoma have abnormalities of chromosome 3 [19–21]. In a study of E.M. Hartmann et al. (2010) regions of chromosomes with CNA affecting gene expression in mantle cell lymphoma were described. The imaginary amplification of the region of interest 15q11.2 was also included in the presented list of loci [22].
In a review published in 2012, a comparative analysis of the results of studies on the detection of CNA using the aCGH hybridization method was performed. Among others the following CA characteristic were distinguished: for patients with diffuse large B cell bone lymphoma, amplification of the region 15q11–26 is typical, in cases of diffuse large B-cell lymphoma, amplification of 15q12 – q21.1 is characteristic, in cases of primary cutaneous B-cell lymphoma, amplification of 22q13 was often detected, and plasmablastic lymphoma was characterized by amplification of 22q12.2–22q13.3 region [23].
CONCLUSIONS
Thus, our study of changes in the CA and CNA frequency of the genetic landscape of blood leukocytes of employees exposed to prolonged gamma irradiation indicates the formation of self-sustaining bone marrow clones in the exposed individuals, the descendants of which produce a population of aberrant cells in the blood. Further research on the mechanisms of preservation of CA, as well as the identification of clones of aberrant cells in the bone marrow is needed. We believe that the dynamic observation and assessment of changes in the representation of a mutant clone may serve as a potential marker for the subclinical stages of hemoblastoses.
REFERENCES
Litvyakov, N.V., Khalyuzova, M.V., Takhauov, R.M., et al., Aberrations in the number of DNA copies in blood lymphocytes of occupationally exposed persons as a potential marker of high radiosensitivity, Vestn. Tomsk. Gos. Univ., Ser. Biol., 2015, no. 2 (30), pp. 113–133.
Zhang, F., Gu, W., Hurles, M.E., and Lupski, J.R., Copy number variation in human health, disease, and evolution, Annu. Rev. Genomics Hum. Genet., 2009, no. 10, pp. 451–481.
Huang, N., Shah, P.K., and Li, C., Lessons from a decade of integrating cancer copy number alterations with gene expression profiles, Brief. Bioinform., 2012, vol. 13, no. 3, pp. 305–316.
Xu, Y., DuanMu, H., Chang, Z., et al., The application of gene co-expression network reconstruction based on CNA and gene expression microarray data in breast cancer, Mol. Biol. Rep., 2012, vol. 39, no. 2, pp. 1627–1637.
Arlt, M.F., Rajendran, S., Birkeland, S.R., et al., Copy number variants are produced in response to low-dose ionizing radiation in cultured cells, Environ. Mol. Mutagen., 2014, vol. 55, no. 2, pp. 103–113.
Litvyakov, N.V., Freidin, M.B., Khalyuzova, M.V., et al., The frequency and spectrum of cytogenetic abnormalities in workers of the Siberian Chemical Combine, Radiats. Biol. Radioecol., 2014, vol. 54, no. 3, pp. 283–296.
Maciejowski, J. and de Lange, T., Telomeres in cancer: tumour suppression and genome instability, Nat. Rev. Mol. Cell Biol., 2017, vol. 18, no. 3, pp. 175–186.
Nik-Zainal, S., Alexandrov, L.B., Wedge, D.C., et al., Mutational processes molding the genomes of 21 breast cancers, Cell, 2012, vol. 149, no. 5, pp. 979–993.
Awa, A.A., Sofuni, T., and Honda, T., Relationship between the radiation dose and chromosome aberrations in atomic bomb survivors of Nagasaki and Hiroshima, J. Radiat. Res., 1978, vol. 19, no. 2, pp. 126–140.
Okladnikova, N.D. and Pesternikova, B.C., Chromosomal aberrations in peripheral blood lymphocytes of people 43–46 years after the acute radiation sickness, Radiats. Biol. Radioecol., 2005, vol. 45, no. 3, pp. 287–290.
Vorobtsova, I.E. and Semenov, A.V., Integrated cytogenetic characteristic of persons affected by the Chernobyl accident, Radiats. Biologiya. Radioecol., 2006, vol. 46, no. 2, pp. 140–151.
Snigireva, G.P., Bogomazova, A.N., Novitskaya, N.N., et al., Experience in the use of cytogenetic methods in radiation research, Vestn. Ros. Voen.-Med. Akad., Pril. I, 2008, no. 3 (23), pp. 184–185.
Pierini, T., Di Giacomo, D., Pierini, V., et al., MYB deregulation from a EWSR1–MYB fusion at leukemic evolution of a JAK2 (V617F) positive primary myelofibrosis, Mol. Cytogenet., 2016, vol. 9, no. 1, p. 68.
Flordal Thelander, E., Ichimura, K., Collins, V.P., et al., Detailed assessment of copy number alterations revealing homozygous deletions in 1p and 13q in mantle cell lymphoma, Leuk. Res., 2007, vol. 31, no. 9, pp. 1219–1230.
Thorat, K.B., Gujral, S., Kumar, A., and Nair, C.N., Small cell variant of T-cell prolymphocytic leukemia exhibiting suppressor phenotype, Leuk. Lymphoma, 2006, vol. 47, no. 8, pp. 1711–1713.
Stengel, A., Kern, W., Zenger, M., et al., Genetic characterization of T-PLL reveals two major biologic subgroups and JAK3 mutations as prognostic marker, Genes Chromosomes Cancer, 2016, vol. 55, no. 1, pp. 82–94.
Monni, O., Oinonen, R., Elonen, E., et al., Gain of 3q and deletion of 11q22 are frequent aberrations in mantle cell lymphoma, Genes Chromosomes Cancer, 1998, vol. 21, no. 4, pp. 298–307.
Thorns, C., Bastian, B., Pinkel, D., et al., Chromosomal aberrations in angioimmunoblastic T-cell lymphoma and peripheral T-cell lymphoma unspecified: a matrix-based CGH approach, Genes Chromosomes Cancer, 2007, vol. 46, no. 1, pp. 37–44.
Lenz, G., Wright, G.W., Emre, N.C., et al., Molecular subtypes of diffuse large B-cell lymphoma arise by distinct genetic pathways, Proc. Natl. Acad. Sci. U. S. A., 2008, vol. 105, no. 36, pp. 13520–13525.
Bea, S., Zettl, A., Wright, G., et al., Diffuse large B-cell lymphoma subgroups have distinct genetic profiles that influence tumor biology and improve gene-expression-based survival prediction, Blood, 2005, vol. 106, pp. 3183–3190.
Tagawa, H., Suguro, M., Tsuzuki, S., et al., Comparison of genome profiles for identification of distinct subgroups of diffuse large B-cell lymphoma, Blood, 2005, vol. 106, pp. 1770–1777.
Hartmann, E.M., Campo, E., Wright, G., et al., Pathway discovery in mantle cell lymphoma by integrated analysis of high-resolution gene expression and copy number profiling, Blood, 2010, vol. 116, no. 6, pp. 953–961.
Tirado, C.A., Chen, W., García, R., et al., Genomic profiling using array comparative genomic hybridization define distinct subtypes of diffuse large B-cell lymphoma: a review of the literature, J. Hematol. Oncol., 2012, vol. 5, no. 54. https://doi.org/10.1186/1756-8722-5-54
Funding
The study was performed with the financial support of the Russian Foundation for Basic Research (project no. 16-34-00876 mol_a).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests. The authors declare that they have no conflict of interest.
Statement of compliance with standards of research involving humans as subjects. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants involved in the study.
Additional information
Translated by V. Mittova
Rights and permissions
About this article
Cite this article
Khalyuzova, M.V., Litviakov, N.V., Takhauov, R.M. et al. Delayed Changes in the Frequency of Unstable Chromosomal Aberrations and the CNA-Genetic Landscape of Blood Leukocytes in People Exposed to Long-Term Occupational Irradiation. Biol Bull Russ Acad Sci 46, 1503–1511 (2019). https://doi.org/10.1134/S1062359019110049
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1062359019110049