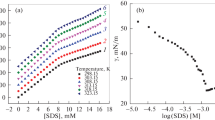
Abstract
The effect of anionic surfactant sodium dodecyl sulfate (SDS) on the hydrolysis of a substrate (mono-4-methyl-2-nitroaniline phosphate) by HCl was studied at 303 K. The reaction followed the first-order kinetics with respect to both HCl and the substrate. SDS effectively catalyzes this reaction, which rate increases with the concentration of SDS due to an increase of dielectric constant of the medium. The kinetic data were fitted to Menger-Portnoy, Piszkiewicz and Berezin kinetic models to explain the observed micellar effects. The various activation parameters both in the presence and absence of SDS were evaluated; a reasonable mechanism was proposed. The rate constant in micellar phase, binding constant and index of cooperativity were calculated accordingly.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Vincent, J.B., Crowder, M.W., and Averill, B.A., Trends Biochem. Sci., 1992, vol.17, p. 105.
Baskin, S.I., Principles of Cardiac Toxicology, Boca Raton: CRC, 1991.
Cox, J.R. and Ramsay, O.B., Chem. Rev., 1964, vol.64, p. 317.
Moss, R.A. and Gong, P.K., Langmuir, 2000, vol. 16, p. 8551.
Pope, C., Karanth, S., and Liu, J., Environ. Toxicol. Pharmacol., 2005, vol.19, p. 433.
Bajgar, J., Acta Med. (Hradec Kralove), 2005, vol. 48, p. 3.
Rahman, M. and Brazel, C.S., Prog. Polym. Sci., 2004, vol.29, p. 1223.
García-Río, L., Hervés, P., Mejuto, J.C., Pérez-Juste, J., and Rodríguez-Dafonte, P., New. J. Chem., 2003, vol.27, p. 372.
García-Río, L., Leis, J.R., and Mejuto, J.C., J. Phys. Chem., 1996, vol.100, p. 10981.
Fendler, J.H., Pure Appl. Chem., 1982, vol.54, p. 1809.
Muller, N., Acc. Chem. Res., 1990, vol.23, p. 23.
Tanford, C. The Hydrophobic Effect: Formation of Micelles and Biological Membranes, 2nd Ed., New York: Wiley, 1980.
Bunton, C.A. and Savelli, G., Adv. Phys. Org. Chem., 1986, vol. 22, p. 213.
Menger, F.M. and Portnoy, C.E., J. Am. Chem. Soc., 1967, vol. 89, p. 4698.
Bunton, C.A. and Moffatt, J.R., J. Phys. Chem., 1985, vol.89, p. 4166.
Duynstee, E.F.J. and Grunwald, E., J. Am. Chem. Soc., 1959, vol.81, p. 4540.
Buurma, N.J., Serena, P., Blandamer, M.J., and Engberts, J.B.F.N., J. Org. Chem., 2004, vol.69, p. 3899.
Bunton, C.A., Foroudian, H.J., Gillitt, N.D., and Whiddon, C.R., J. Colloid Interface Sci., 1999, vol.215, p. 64.
Baldwin, D.S., Beattie, J.K., Coleman, L.M., and Jones, D.R., Environ. Sci. Technol., 2001, vol.35, p. 713.
Desloges, W., Neverov, A.A., and Brown, R.S., Inorg. Chem., 2004, vol.43, p. 6752.
Kim, Y.H., Ahn, J.Y., Moon, S.H., and Lee, J., Chemosphere, 2005, vol. 60, p. 1349.
Simanenko, Y.S., Prokop’eva, T.M., Popov, A.F., Bunton, C.A., Karpichev, E.A., Savelova, V.A. and Ghosh, K.K., Russ. J. Org. Chem., 2004, vol.40, p. 1337.
Tsang, J.S.W., Neverov, A.A., and Brown, R.S., J. Am. Chem. Soc., 2003, vol.125, p. 7602.
Simanenko, Y.S., Popov, A.F., Prokop’eva, T.M., Karpichev, E.A., Savelova, V.A., Suprun, I.P., and Bunton, C.A., Russ. J. Org. Chem., 2002, vol.38, p. 1286.
Hammond, P.S., Forster, J.S., Lieske, C.N., and Durst, H.D., J. Am. Chem. Soc., 1989, vol.111, p. 7860.
Ghosh, K.K., Satnami, M.L., and Sinha, D., Tetrahedron Lett., 2004, vol.45, p. 9103.
Campbell, M.K. and Farrell, S.O., Biochemistry, 5th Ed., Belmont: Thomson Brooks/Cole, 2006, p. 5.
Cavalier, J., Bull. Soc. Chim. Fr., 1895, vol.13, p. 885.
Laidler, K.J., Chemical Kinetics, 3th Ed., New York: Harper and Row, 1987.
Chanley, J.D. and Feageson, E., J. Am. Chem. Soc., 1958, vol.80, p. 2686.
Imae, T. and Ikeda, S., Colloid Polym. Sci., 1987, vol.265, p. 1090.
Piszkiewicz, D., J. Am. Chem. Soc., 1977, vol.99, p. 1550.
Berezin, I.V., Martinek, K., and Yatsimirskii, A.K., Russ. Chem. Rev., 1973, vol.42, p. 787.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Bairagi, B., Bhoite, S.A. & Singh, A.K. Micellar effect on hydrolysis of 4-methyl-2-nitroaniline phosphate. Colloid J 76, 765–773 (2014). https://doi.org/10.1134/S1061933X14060027
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1061933X14060027