Abstract
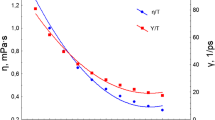
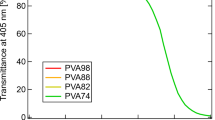
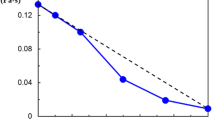
The particular permeability of hydrated poly(vinyl alcohol) (PVA) was investigated from the point of polymeric relaxation behavior, and the more essential effect of hydrogen bonds in supramolecular structure was further analyzed as well. The permeability measurement showed that various hydrated PVA samples all had lower permeability to non-polar solvents, and the permeability was affected by relaxation state, which can be controlled by water content. Dynamic mechanical analysis measurements indicated that, in PVA with lower water content the relaxation motion benefiting permeation almost became frozen in glassy amorphous region, due to high activation energy of conformation transition, which then induced the slower permeation to occur. Solid-state 1H and 13C NMR spectra of hydrated PVA samples allowed to analyze the effect of hydrogen bonds. It was found that in PVA with lower water content conformation transition in amorphous region was restricted by more intermolecular and intramolecular hydrogen bonds, especially by the former. Also relatively added crystalline regions, which can affect permeability as physical cross-linking points, were mainly constructed by chain segments with no intramolecular hydrogen bonds. Above results suggest that intermolecular and intramolecular hydrogen bonds via conformation transition control relaxation behavior. Consequently, the relaxation behavior affects the permeability of hydrated PVA, in which water play an important role.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
G. E. Karlsson, U. W. Gedde, and M. S. Hedenqvist, Polymer 45, 3893 (2004).
I. Cozmuta, M. Blanco, and W. A. Goddard, J. Phys. Chem. B 111, 3151 (2007).
J. Jang and D. K. Lee, Polymer 45, 1599 (2003).
F. Peng, L. Lu, H. Sun, Y. Wang, H. Wu, and Z. Jiang, J. Membrane Sci. 275, 97 (2006).
H. Matsuyama, M. Teramoto, and H. Urano, J. Membrane Sci. 126, 151 (1997).
J. T. Yeh, L. H. Qang, K. N. Chen, and W. S. Jou, J. Mater. Sci. 36, 1891 (2001).
C. Gagnard, Y. Germain, P. Keraudren, and B. Barriére, J. Appl. Polym. Sci. 92, 676 (2004).
Polymer Permeability, Ed. by J. Comyn (Elsevier, New York, 1985})
R. M. Hodge, T. J. Bastow, G. H. Edward, G. P. Simon, and A. J. Hill, Macromolecules 29, 8137 (1996).
T. J. Bastow, R. M. Hodge, and A. J. Hill, J. Membrane Sci. 131, 207 (1997).
T. Terao, S. Maeda, and A. Saika, Macromolecules 16, 1535 (1983).
S. Lai, M. Casu, G. Saba, A. Lai, I. Husu, G. Masci, V. Crescenzi, G. Masci, and V. Crescenzi, Solid State Nucl. Magn. Reson. 21, 187 (2002).
M. Kobayashi, M. Kanekiyo, I. Ando, and S. Amiya, Polymer Gels and Networks 6, 425 (1998).
M. Kobayashi, I. Ando, T. Ishii, and S. Amiya, Macromolecules 28, 6677 (1995).
M. Kobayashi, I. Ando, T. Ishii, and S. Amiya, J. Mol. Struct. 440, 155 (1998).
R. Ricciardi, C. Gaillet, G. Ducouret, F. Lafuma, and F. Lauprétre, Polymer 44, 3375 (2003).
F. Horii, S. Hu, K. Deguchi, H. Sugisawa, H. Ohgi, and T. Sato, Macromolecules 29, 3330 (1996).
K. Masuda and F. Horri, Macromolecules 31, 5810 (1998).
K. Masuda, H. Kaji, and F. Horri, Polym. J. 31, 105 (1999).
K. Masuda, H. Kaji, and F. Horri, J. Polym. Sci. Pol. Phys. 38, 1 (2000).
K. Masuda, H. Kaji, and F. Horri, Polym. J. 33, 190 (2001).
K. Masuda, H. Kaji, and F. Horri, Polym. J. 33, 356 (2001).
H. Ohgi, H. Yang, T. Sato, and F. Horii, Polymer 48, 3850 (2007).
N. Chen, L. Li, and Q. Wang, Plast. Rubber Compos. 36, 284 (2007).
Lange’s Handbook of Chemistry, Ed. by J. A. Dean, 15th ed. (McGraw-Hill, New York, 1999).
Recommended Reference Materials for the Realization of Physicochemical Properties, Ed. by K. N. Marsh (Blackwell Scientific, Oxford, 1987})
W. B. Li, F. Xue, and R. S. Cheng, Polymer 46, 12026 (2005).
S. S. Wong, S. A. Altinkaya, and S. K. Mallapragada, Polymer 45, 5151 (2004).
Hansen Solubility Parameters: a User’s Handbook, Ed. by C. M. Hansen, 2nd ed. Taylor & Francis, New York, 2007})
Plastic Packaging Materials for Food, Eds. by O. G. Piringer and A. L. Baner (Wiley-Vch Verlag Gmbh, Malden, 2000})
V. J. McBrierty, F. X. Quinn, C. Keely, A. C. Wilson, and G. D. Friends, Macromolecules 25, 4281 (1992).
D. Capitani, G. Mensitieri, F. Porro, N. Proietti, and A. L. Segre, Polymer 44, 6589 (2003).
J. M. Lagaron, A. K. Powell, and G. Bonner, Polym. Test 20, 569 (2001).
A. T. Dibenedetto, J. Polym. Sci. A 1, 3459 (1963).
A. T. Dibenedetto, J. Polym. Sci. A 1, 3477 (1963).
J. S. Vrentas and J. L. Duta, J. Polym. Sci. Pol. Phys. 15, 403 (1977).
L. Fritz and D. Hofmann, Polymer 38, 1035 (1997).
Diffusion in Polymer, Ed. by P. Neogi (Marcel Dekker, New York, 1996})
A. D. L. Rosa, L. Heux, and J. Y. Cavaillé, Polymer 41, 7547 (2000).
Q. Yu, M. Zhou, Y. Ding, B. Jiang, and S. Zhu, Polymer 48, 7058 (2007).
F. Horii, S. Hu, T. Ito, H. Odani, S. Matsuzawa, and K. Yamaura, Polymer 33, 2299 (1992).
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Huang, L., Wang, J.H. Permeability of hydrated poly(vinyl alcohol): Effect of relaxation behaviors and hydrogen bonds in supramolecular structure. Polym. Sci. Ser. A 57, 656–666 (2015). https://doi.org/10.1134/S0965545X15050107
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0965545X15050107