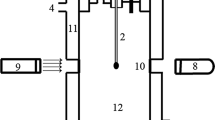
Abstract
The results from investigating the surface characteristics of liquid sodium, potassium, and four potassium–sodium alloys are considered. The Auger spectra and profiles of metal droplets with atomically pure surfaces in the solid and liquid state are studied under the same experimental conditions. It is shown that potassium is a surfactant with respect to sodium at the temperature of an experiment in the investigated range of volume concentrations. It is noted that the obtained values of surface tension are 15% higher than the reference values.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
O. G. Ashkhotov, M. V. Zdravomyslov, A. V. Sardlishvili, and R. V. Plyushchenko, Russ. J. Phys. Chem. A 71, 121 (1997).
O. G. Ashkhotov and M. V. Zdravomislov, Surf. Sci. 339, 279 (1995).
O. G. Ashkhotov, Poverkhnost, No. 2, 5 (1996).
F. D. Bashfort and J. E. Adams, An Attempt to Test Theories of Capillary Action by Comparing the Theoretical and Measured Forms of Drops of Fluid (Cambridge Univ. Press, Cambridge, 1883; Kessinger, Whitefish, Montana, 2007).
M. L. Knotek and P. Feibelman, Surf. Sci. 90, 78 (1979).
T. Sekine, Surf. Sci. 125, 565 (1983).
V. Kh. Sheriev, T. P. Osiko, and O. G. Ashkhotov, Teplofiz. Vys. Temp. 29, 85 (1991).
B. B. Alchagirov, L. Kh. Afaunova, Z. A. Kegadueva, and R. Kh. Arkhestov, Russ. J. Phys. Chem. A 85, 1097 (2011).
Z. Kh. Kalazhokov, K. V. Zikhova, Zaur Kh. Kalazhokov, and Kh. Kh. Kalazhokov, Rasplavy, No. 4, 12 (2015).
Wei-Heng Shih and D. Stroud, Phys. Rev. B 32, 804 (1985).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © O.G. Ashkhotov, I.B. Ashkhotova, M.A. Aleroev, T.T. Magkoev, A.P. Bliev, 2017, published in Zhurnal Fizicheskoi Khimii, 2017, Vol. 91, No. 7, pp. 1171–1173.
Rights and permissions
About this article
Cite this article
Ashkhotov, O.G., Ashkhotova, I.B., Aleroev, M.A. et al. Surface properties of sodium, potassium, and their binary alloys in the liquid state. Russ. J. Phys. Chem. 91, 1270–1272 (2017). https://doi.org/10.1134/S0036024417070032
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024417070032