Abstract
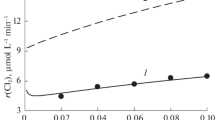
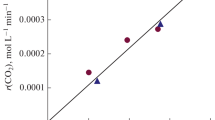
Reactions of the oxidation of bivalent cobalt ions by ozone, of the spontaneous decomposition of trivalent cobalt, and of interactions between Co(III) and chloride ions in solutions of sulfuric acid are studied. The order and rate constant of the process of decomposition of Co(III) are determined. Information on the kinetics of the interaction between Co(III) and Cl– is obtained. Kinetic patterns of the accumulation of Co(III) during the ozonation of solutions of CoSO4 in sulfuric acid are explained. Molar absorption coefficients of Co(III) and Co2+ ions in the visible range of wavelengths are determined.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
D. S. Pines and D. A. Reckho., Environ. Sci. Technol. 36, 4046 (2002).
F. J. Beltrán, F. J. Rivas, and R. Montero-De-Espinos., Ind. Eng. Chem. Res. 42, 3218 (2003).
F. J. Beltrán, F. J. Rivas, and R. Montero-De-Espinos., Ind. Eng. Chem. Res. 42, 3210 (2003).
L. R. B. Yeatts and H. Taub., J. Am. Chem. Soc. 71, 4100 (1949).
A. V. Levanov, I. V. Kuskov, K. B. Koiaidarova, A. V. Zosimov, E. E. Antipenko, and V. V. Luni., Kinet. Catal. 46, 138 (2005).
G. R. Hil., J. Am. Chem. Soc. 70, 1306 (1948).
G. R. Hil., J. Am. Chem. Soc. 71, 2434 (1949).
A. V. Levanov, O. Ya. Isaikina, A. N. Tyutyunnik, E. E. Antipenko, and V. V. Luni., J. Anal. Chem. 71, 549 (2016).
V. N. Alekseev, Quantitative Analysis (Goskhimizdat, Moscow, 1958, p. 370; Mir, Moscow, 1969).
R. K. Murmann, J. C. Sullivan, and R. C. Thompso., Inorg. Chem. 7, 1876 (1968).
G. Wangila and R. B. Jorda., Inorg. Chim. Acta 343, 347 (2003).
J. C. Sullivan and R. C. Thompso., Inorg. Chem. 6, 1795 (1967).
G. Davies and K. O. Watkin., J. Phys. Chem. 74, 3388 (1970).
C. E. Bricker and L. J. Loeffle., Anal. Chem. 27, 1419 (1955).
The Chemist’s Reference Book, Ed. by B. P. Nikol’skii (Khimiya, Leningrad, 1967), Vol. 4, p. 747 [in Russian].
G. W. Wangila and R. B. Jorda., Inorg. Chim. Acta 359, 1023 (2006).
J. H. Baxendale and C. F. Well., Trans. Faraday Soc. 53, 800 (1957).
C. E. H. Bawn and A. G. Whit., J. Chem. Soc., 331 (1951).
G. Davies and B. Warnqvis., Coord. Chem. Rev. 5, 349 (1970).
H. Taub., J. Gen. Physiol. 49, 29 (1965).
A. V. Levanov, I. V. Kuskov, E. E. Antipenko, and V. V. Luni., Russ. J. Phys. Chem. A 82, 1126 (2008).
A. V. Levanov, O. Y. Isaykina, N. K. Amirova, E. E. Antipenko, and V. V. Luni., Environ. Sci. Pollut. Res. 22, 16554 (2015).
T. J. Conocchioli, G. H. Nancollas, and N. Suti., Inorg. Chem. 5, 1 (1966).
A. Mcauley, M. N. Malik, and J. Hil., J. Chem. Soc. A, 2461 (1970).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.V. Levanov, O.Ya. Isaikina, V.V. Lunin, 2016, published in Zhurnal Fizicheskoi Khimii, 2016, Vol. 90, No. 12, pp. 1791–1796.
Rights and permissions
About this article
Cite this article
Levanov, A.V., Isaikina, O.Y. & Lunin, V.V. Oxidation of Co(II) by ozone and reactions of Co(III) in solutions of sulfuric acid. Russ. J. Phys. Chem. 90, 2358–2363 (2016). https://doi.org/10.1134/S0036024416120190
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024416120190