Abstract
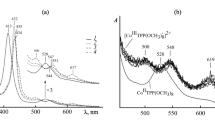
Kinetic and thermodynamic parameters of the donor–acceptor self-organization in the system (5,10,15,20-tetraphenyl-21H,23H-porphyrinato)cobalt(II) (CoTPP)–1-methyl-2-(pyridin-4′-yl)-3,4-fullero[60]pyrrolidine (PyC60)–toluene yielding the 1: 2 complex (PyC60)2CoTPP are presented. The chemical structure of the supramolecular triad was elucidated by UV-vis, IR, and (1H, 13C) NMR spectroscopy. The photoelectrochemical characteristics of the Ti|TiO2 electrode modified by the triad and its precursors were determined and the redox behavior of the latter was studied by cyclic voltammetry in dichloromethane.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
M. Li, Y.-J. Liu, J.-X. Zhao, et al., Appl. Surf. Sci. 355, 1145 (2015). https://doi.org/10.1016/j.apsusc.2015.03.144
M. Moradi, Z. Bagheri, and A. Bodaghi, Physica E 89, 148 (2017). https://doi.org/10.1016/j.physe.2017.02.018
L. Qiao, X. Sun, Z. Yang, et al., Carbon 54, 29 (2013). https://doi.org/10.1016/j.carbon.2012.10.066
K. Tada, Sol. Energy Mater. Sol. Cells 130, 331 (2014). https://doi.org/10.1016/j.solmat.2014.07.032
K. Tada, Appl. Phys. Express 7, 051601 (2014). https://doi.org/10.7567/APEX.7.051601
N. V. Kalacheva, G. R. Tarasova, G. M. Fazleeva, etal., Bioorg. Med. Chem. Lett. 28, 1097 (2018). https://doi.org/10.1016/j.bmcl.2018.02.009
Z. Chen, L. Ma, Y. Liu, et al., Theranostics 2, 238 (2012). https://doi.org/10.7150/thno.3509
S. Goodarzi, T. Da Ros, J. Conde, et al., Mater. Today 20, 460 (2017). https://doi.org/10.1016/j.mattod.2017.03.017
N. Yu. Borovkov and S. V. Blokhina, Colloids Surf., A: Physicochem. Eng. Aspects 377, 393 (2011). https://doi.org/10.1016/j.colsurfa.2011.01.044
M. Baibarac, I. Baltog, M. Daescu, et al., J. Mol. Struct. 1125, 340 (2016). https://doi.org/10.1016/j.molstruc.2016.07.001
B. Chandra and F. D’Souza, Coord. Chem. Rev. 322, 104 (2016). https://doi.org/10.1016/j.ccr.2016.05.012
C. Solis, M. B. Ballatore, M. B. Suarez, et al., Electrochim. Acta 238, 81 (2017). https://doi.org/10.1016/j.electacta.2017.04.015
L.-L. Li and E. W.-G. Diau, Chem. Soc. Rev. 42, 291 (2013). https://doi.org/10.1039/C2CS35257E
T. Higashino and H. Imahori, J. Chem. Soc., Dalton Trans. 44, 448 (2015). https://doi.org/10.1039/C4DT02756F
M. Urbani, M. Gratzel, M. K. Nazeeruddin, et al., Chem. Rev. 114, 12330 (2014). https://doi.org/10.1021/cr5001964
H. Imahori, T. Umeyama, K. Kurotobi, et al., Chem. Commun. 48, 4032 (2012). https://doi.org/10.1039/C2CC30621B
T. N. Lomova, E. V. Motorina, and M. V. Klyuev, CRC Concise Encyclopedia of Nanotechnology, Ed. by B. I. Kharisov, O. V. Kharissova, and U. Ortiz-Mendez (CRC Press, London, 2016).
E. V. Motorina, T. N. Lomova, and M. V. Klyuev, Mend. Commun. 28, 426 (2018). https://doi.org/10.1016/j.mencom.2018.07.029
E. N. Ovchenkova, N. G. Bichan, N. O. Kudryakova, et al., Dyes Pigm. 153, 225 (2018). https://doi.org/10.1016/j.dyepig.2018.02.023
J. M. Gottfried, Surf. Sci. Rep. 70, 259 (2018). https://doi.org/10.1016/j.surfrep.2015.04.001
D. M. Lyons, J. Kesters, W. Maes, et al., Synth. Met. 178, 56 (2013). https://doi.org/10.1016/j.synthmet.2013.06.013
S. Ciurli, S. Gambarotta, C. Floriani, et al., Angew. Chem., Int. Ed. Engl. 98, 553 (1986).
S. Fukuzumi and J. Maruta, Inorg. Chim. Acta 226, 145 (1994). https://doi.org/10.1016/0020-1693(94)04080-X
W. Choi, P. G. Ingole, H. Li, et al., J. Cleaner Prod 133, 1008 (2016). https://doi.org/10.1016/j.jclepro.2016.06.031
L. P. H. Saravia, S. Anandhakumar, A. L. A. Parussulo, et al., J. Electroanal. Chem. 775, 72 (2016). https://doi.org/10.1016/j.jelechem.2016.05.026
H. Li, W. Choi, P. G. Ingole, et al., Fuel 185, 133 (2016). https://doi.org/10.1016/j.fuel.2016.07.097
D. V. Konarev, S. S. Khasanov, A. Otsuka, et al., J. Chem. Soc., Dalton Trans., No. 32, 6416 (2009). https://doi.org/10.1039/B904293H
D. V. Konarev, S. S. Khasanov, A. Otsuka, et al., Chem.-Eur. J. 9, 3837 (2003). https://doi.org/10.1002/chem.200204470
D. V. Konarev, S. S. Khasanov, G. Saito, et al., Proceedings of the International Young Scientist Workshop “Chemistry of Porphyrins and Related Compounds,” Ivanovo, 2012 (Ivanovo, 2012), p. 59 [in Russian].
J. Kim, S. H. Lim, Y. Yoon, et al., Tetrahedron Lett. 52, 2645 (2011). https://doi.org/10.1016/j.tetlet.2011.03.048
L. Zheng, Y. Dan, L. Xiong, et al., Anal. Chim. Acta 768, 69 (2013). https://doi.org/10.1016/j.aca.2013.01.019
K. Deng, J. Zhou, and X. Li, Electrochim. Acta 114, 341 (2013). https://doi.org/10.1016/j.electacta.2013.09.164
M. Prato, M. Maggini, C. Giacometti, et al., Tetrahedron 52, 5221 (1996). https://doi.org/10.1016/0040-4020(96)00126-3
N. G. Bichan, E. N. Ovchenkova, M. S. Gruzdev, et al., Russ. J. Struct. Chem. 59, 711 (2018). https://doi.org/10.1134/S0022476618030320
E. V. Motorina, T. N. Lomova, and M. V. Klyuev, Makrogeterotsikly 6, 327 (2013). https://doi.org/10.6060/mhc1306441
N. G. Bichan, E. N. Ovchenkova, N. O. Kudryakova, et al., J. Coord. Chem. 70, 2371 (2017). https://doi.org/10.1080/00958972.2017.1335867
A. Mansour, M. Zaied, I. Ali, et al., Polyhedron 127, 496 (2017). https://doi.org/10.1016/j.poly.2016.10.031
J. Macka, M. J. Stillman, and N. Kobayashi, Coord. Chem. Rev. 251, 429 (2007).
Y. Terazono, B. O. Patrick, and D. H. Dolphin, Inorg. Chim. Acta 346, 265 (2003).
M. C. Martin, X. Du, and L. Mihaly, Phys. Rev. B 50, 173 (1994).
E. N. Ovchenkova, N. G. Bichan, and T. N. Lomova, Russ. J. Org. Chem. 52, 1503 (2016). https://doi.org/10.1134/S1070428016100213
N. G. Bichan, E. N. Ovchenkova, N. O. Kudryakova, et al., New J. Chem., No. 42, 12449 (2018). https://doi.org/10.1039/C8NJ00887F
F. D’ Souza, G. R. Deviprasad, M. E. Zandler, et al., J. Phys. Chem. A 106, 3243 (2002). https://doi.org/10.1021/jp013165i
F. D’ Souza, G. R. Deviprasad, M. E. Zandler, et al., J. Phys. Chem. A 107, 4801 (2003). https://doi.org/10.1021/jp030363w
P. A. Troshin, A. S. Peregudov, D. Muhlbacher, et al., Eur. J. Org. Chem., No. 14, 3064 (2005). https://doi.org/10.1002/ejoc.200500048
K. M. Kadish, X. Q. Lin, and B. C. Han, Inorg. Chem. 26, 4161 (1987). https://doi.org/10.1021/ic00272a006
Author information
Authors and Affiliations
Corresponding author
Additional information
Russian Text © The Author(s), 2019, published in Zhurnal Neorganicheskoi Khimii, 2019, Vol. 64, No. 5, pp. 490–499.
Rights and permissions
About this article
Cite this article
Bichan, N.G., Ovchenkova, E.N., Mozgova, V.A. et al. Formation Reaction, Spectroscopy, and Photoelectrochemistry of the Donor–Acceptor Complex (5,10,15,20-Tetraphenyl-21,23H-porphinato)cobalt(II) with Pyridyl-Substituted Fullero[60]pyrrolidine. Russ. J. Inorg. Chem. 64, 605–614 (2019). https://doi.org/10.1134/S0036023619050024
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023619050024