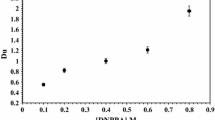
Abstract
A possibility to use acid-type phosphoryl-containing podands with diethylene glycol polyether chain, which differ in substituent at the phosphoryl group, as extractants for the recovery of U(VI), Th(IV), and rare earth elements(III) from nitric acid solutions has been studied. Features of the effect of HNO3 concentration on U(VI) and Th(IV) extraction with solutions of the phosphoryl-containing podands in dichloroethane has been revealed. Uranium(VI) is extracted as a normal intracomplex salt of dibasic acid UO2L3 with chelate coordination of both POO− groups to one cation. Thorium(IV) produces a complex as a normal intracomplex salt of composition Th(L3)2 where two bivalent anions of ligand acid are coordinated via all four POO− groups to Th(IV) cation. Sorption of U(VI), Th(IV), and REE(III) by impregnated sorbent based on LPS-500 polymer with 1,5-bis[2-(oxyethoxyphosphoryl-4-ethyl)phenoxy]-3-oxapentane from 0.052 and 3.52 mol/L HNO3 has been studied. It has been established that the obtained sorbent shows high selectivity in the separation of U(VI), Th(IV), and REE(III). The separation factor for uranium(VI) and europium(III) (βU/Eu) has been found to be 202 and ∼55000 upon sorption from 3.52 and 0.052 mol/L HNO3, respectively, at V/m = 500 mL/g. At thorium(IV) sorption from 3.52 and 0.052 mol/L HNO3, βTh/U = 66 and ∼5050, respectively.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
M. Y. Alyapyshev, V. A. Babain, and Y. A. Ustynyuk, Russ. Chem. Rev. 85, 943 (2016). doi https://doi.org/10.1070/rcr4589
K. L. Nash, R. E. Barrans, R. Chiarizia, et al., Solvent Extr. Ion Exch. 18, 605 (2000). doi https://doi.org/10.1080/07366290008934700
O. B. Mokhodoeva, G. V. Myasoedova, and E. A. Zakharchenko, Radiochemistry 53, 35 (2011). doi https://doi.org/10.1134/S106636221101005X
G. V. Myasoedova, Ross. Khim. Zh. 49, 72 (2005).
G. V. Myasoedova, E. A. Zakharchenko, N. P. Molochnikova, et al., Radiochemistry 50, 482 (2008). doi https://doi.org/10.1134/S1066362208050093
A. N. Turanov, V. K. Karandashev, A. N. Yarkevich, et al., Radiochemistry 44, 559 (2002). doi https://doi.org/10.1023/A:1022376225458
E. P. Horwitz, D. R. McAlister, and M. L. Dietz, Sep. Sci. Technol. 41, 2163 (2006). doi https://doi.org/10.1080/01496390600742849
I. Boulenouar, M. KaiD, O. Abbers, et al., Solvent Extr. Res. Develop., Japan 23, 51 (2016). doi https://doi.org/10.15261/serdj.23.51
M. Jensen, R. Chiarizia, J. S. Ulicki, et al., Solvent Extr. Ion Exch. 33, 329 (2015). doi https://doi.org/10.1080/07366299.2015.1046292
R. Chiarizia, Jr. R. E. Barrans, J. R. Ferraro, et al., Sep. Sci. Technol. 36, 687 (2001). doi https://doi.org/10.1081/SS100103615
R. Chiarizia, D. R. Mcalister, and A. W. Herlinger, Sep. Sci. Technol. 40, 69 (2005). doi https://doi.org/10.1081/SS200041762
J. Wang, G. Chen, S. Xu, and L. Li, Hydrometallurgy 154, 129 (2015). https://doi.org/10.1016/j.hydromet.2015.04.001.
A. N. Turanov, V. K. Karandashev, A. N. Yarkevich, et al., Radiochemistry 45, 568 (2003). doi https://doi.org/10.1023/B:RACH.0000015753.31087.ef
G. J. Lumetta, S. I. Sinkov, J. A. Krause, et al., Inorg. Chem. 55, 1633 (2016). doi https://doi.org/10.1021/acs.inorgchem.5b02524
E. P. Horwitz, R. Chiarizia, and M. L. Dietz, React. Funct. Polym. 33, 25 (1997).
R. Chiarizia, A. W. Herlinger, Y. D. Cheng, et al., Solvent Extr. Ion Exch. 16, 505 (1998). doi https://doi.org/10.1080/07366299808934537
V. E. Baulin, V. Kh. Syundyukova, and E. N. Tsvetkov, Russ. J. Gen. Chem., 59, 62 (1989).
A. N. Turanov, V. K. Karandashev, V. E. Baulin, et al., Radiochemistry 56, 22 (2014). doi https://doi.org/10.1134/S1066362214010056
A. M. Safiulina, A. G. Matveeva, and D. V. Ivanets, et al., Russ. Chem. Bull. 64, 161 (2015). doi https://doi.org/10.1007/s11172-015-0837-2
E. V. Chukhlantseva, A. N. Usolkin, O. V. Kovalenko, et al., Analitika I Kontrol’ 17, 219 (2013). doi https://doi.org/10.15826/analitika.2013.17.2
E. V. Chukhlantseva, A. N. Usolkin, V. E. Baulin, et al., Analitika i Kontrol’ 17, 314 (2013). doi https://doi.org/10.15826/analitika.2013.17.3.007
V. E. Baulin, O. V. Kovalenko, A. Yu. Tsivadze, et al., Radiochemistry 57, 61 (2015). doi https://doi.org/10.1134/S1066362215010099
V. E. Baulin, I. P. Kalashnikova, and O. V. Kovalenko, et al., Prot. Met. Phys. Chem. Surf. 52, 996 (2016). doi https://doi.org/10.7868/S0044185616060085
A. M. Safiulina, A. G. Matveeva, and D. V. Ivanets, et al., Russ. Chem. Bull. 64, 169 (2015). doi https://doi.org/10.1007/s11172-015-0838-1
S. B. Savvin, Organic Reagents of Arsenazo III Group (Atomizdat, Moscow, 1971) [in Russian].
OST 95 10353-2008. Branch Standard. Branch System for the Unity of Measurements. Algorithms for Assessment of Metrological Characteristics on Measurement Procedures Attestation. Accepted and Put into Action by the Federal Atomic Energy Agency on 2008 [in Russian].
Yu. V. Egorov, N. D. Betenekov, and V. D. Puzako, Methods for Preconcentration and Separation of Radionuclides: A Tutorial (Ural Univ., Yekaterinburg, 2016) [in Russian].
A. M. Rozen and B. V. Krupnov, Russ. Chem. Rev. 65, 973 (1996). doi https://doi.org/10.1070/RC1996v065n11ABEH000241
G. I. Timofeeva, A. G. Matveeva, and A. M. Safiulina, et al., Russ. Chem. Bull. 64, 224 (2015). doi https://doi.org/10.1007/s11172-015-0847-0
STO RosGeo 08-002-98. Technological Methods for the Investigation of Mineral Raw Materials. Text (RosGeo, Moscow, 1998), p. 33 [in Russian].
Acknowledgments
This work was performed under the State Assignment (theme no. 0090-2017-0024) and supported in part by the Presidium of the RAS (Fundamental Research Program no. 34 “Actual problems of surface physical chemistry and design of novel composites”) as well as by the Russian Foundation for Basic Research (project no. 18-29-24069).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Safiulina, A.M., Ivanets, D.V., Kudryavtsev, E.M. et al. Liquid- and Solid-Phase Extraction of Uranium(VI), Thorium(IV), and Rare Earth Elements(III) from Nitric Acid Solutions Using Acid-Type Phosphoryl-Containing Podands. Russ. J. Inorg. Chem. 64, 536–542 (2019). https://doi.org/10.1134/S0036023619040181
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023619040181