Abstract
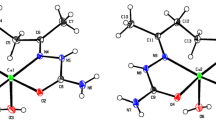
Nickel, copper, and zinc complexes with salicylidenehydrazone of iminodiacetic acid (H4L): Cu2L · 2Py · 2CH3OH and Cu2ML(CH3COO)2 (M= Ni, Cu, Zn) were synthesized and studied by thermo-gravimetric analysis and IR and ESR spectroscopy. The crystal structure of the complex Cu2ZnL(CH3COO)2 · 4Py · CH3OH was studied by X-ray diffraction: space group C2/c, a = 31.6974(12) Å, b = 10.3023(4) Å, c = 16.4714(7) Å; β = 119.7460(10)°; Z = 4; 3513 reflections with I > 2σ(I); R = 0.0382, R w = 0.1010. The temperature dependences of the ESR spectra of liquid solutions and the magnetic susceptibility of polycrystalline samples were studied. The ESR spectra of the binuclear copper complex and the trinuclear dicopper zinc complex exhibited hyperfine structure of seven lines, indicating exchange interaction of unpaired electrons with two equivalent copper nuclei.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
W. Luo, X.-G. Meng, J.-F. Xiang, et al., Inorg. Chim. Acta 361, 2667 (2008).
O. Das, E. Zangrando, and T. K. Paine, Inorg. Chim. Acta 362, 3617 (2009).
E. I. Solomon, U. M. Sundaram, and T. E. Machonkin, Chem. Rev. 96, 2563 (1996).
L. M. Mirica and T. D. P. Stack, Inorg. Chem. 44, 2131 (2005).
J. Yoon and E. I. Solomon, Coord. Chem. Rev. 251, 379 (2007).
H. Borzel, P. Comba, and H. Pritzkow, Chem. Commun., No. 1, 97 (2001).
S. S.-Y. Chui, S. M.-F. Lo, J. P. H. Charmant, et al., Science 283(5405), 1148 (1999).
X. Chen, S. Zhan, C. Hu, et al., J. Chem. Soc., Dalton Trans., No. 2, 245 (1997).
V. A. Milway, L. Zhao, T. S. M. Abedin, et al., Polyhedron 22, 1271 (2003).
H. Adams, D. E. Fenton, and G. Minardi, Inorg. Chem. Commun. 3, 24 (2000).
Yu. V. Rakitin, G. M. Larin, and V. V. Minin, Interpretation of EPR Spectra of Coordination Compounds (Nauka, Moscow, 1993) [in Russian].
Yu. V. Rakitin and V. T. Kalinnikov, Advanced Magnetochemistry (Nauka, St. Petersburg, 1994) [in Russian]
SMART (Control) and SAINT (Integration) Software, Version 5.0, Bruker AXS, Inc., Madison (WI), 1997.
G. M. Sheldrick, Acta Crystallogr., Sect. A 64, 112 (2008).
S. E. Nefedov, Yu. T. Struchkov, O. V. Konnik, et al., Ukr. Khim. Zh. 57, 685 (1991).
O. V. Konnik, V. F. Shul’gin, S. E. Nefedov, et al., Zh. Neorg. Khim. 36, 630 (1991).
G. Orpen, L. Brammer, F. H. Allen, et al., J. Chem. Soc., Dalton Trans. Suppl., S1 (1989).
F. H. Allen, O. Kennard, D. G. Watson, et al., J. Chem. Soc., Perkin Trans. II, Suppl. S1 (1987).
A. W. Addison and T. N. Rao, J. Chem. Soc., Dalton Trans., 1349 (1984).
B. Chiari, W. E. Hatfild, O. Piovesana, et al., Inorg. Chem. 22, 1468 (1983).
V. F. Shul’gin, O. V. Konnik, A. S. Bogomyakov, et al., Russ. J. Inorg. Chem. 57, 615 (2012).
A. Yatani, M. Fujii, Y. Nakao, et al., Inorg. Chim. Acta 316, 127 (2001).
R.-J. Tao, C.-Z. Mei, S.-Q. Zang, et al., Inorg. Chim. Acta 357, 1985 (2004).
S. Thakurta, C. Rizzoli, R. J. Butcher, et al., Inorg. Chim. Acta 363, 1395 (2010).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © O.V. Konnik, V.F. Shul’gin, E.A. Zamnius, A.N. Gusev, V.V. Minin, 2015, published in Zhurnal Neorganicheskoi Khimii, 2015, Vol. 60, No. 5, pp. 664–671.
Rights and permissions
About this article
Cite this article
Konnik, O.V., Shul’gin, V.F., Zamnius, E.A. et al. Trinuclear copper(II) complexes based on iminodiacetic acid salicylidenehydrazone. Russ. J. Inorg. Chem. 60, 595–601 (2015). https://doi.org/10.1134/S0036023615050125
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023615050125