Abstract
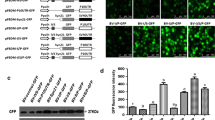
Mammalian T7 polymerase-based cytoplasmic expression systems are common tool for molecular studies. The majority of these systems include the internal ribosome entry site (IRES) of the encephalomyocarditis virus (EMCV). To carry out a cap-independent translation process, this type of IRES might require the expression of an extensive array of host factors, what is a disadvantage. Other IRESes might be less dependent on the host cell factors, but their biology is characterized to a lesser degree. Here, we compare the translational efficiencies of bovine viral diarrhea virus (BVDV) IRES with that of ECMV. Both IRESes were tested in reporter vectors containing the T7 promoter, an IRES of choice and the coding sequence of the enhanced green fluorescent protein (EGFP). To provide for the expression of T7 RNA polymerase, the corresponding gene was isolated from Escherichia coli and inserted into pCDNA3.1-Hygro(+). After co-transfection of the T7 RNA polymerase encoding vector with either of the two IRES-containing reporter vectors into T7 baby hamster kidney (T7-BHK), human embryonic kidney (HEK) 293T, chinese hamster ovary (CHO) and HeLa cells, the translational efficiency of the reporter construct was studied by fluorescence microscopy and flow cytometry. In T7-BHK, HEK 293T and HeLa cells the translational efficiency of BVDV IRES was two to three times higher than that of EMCV IRES. In CHO cells, BVDV IRES and EMCV IRES were equally efficient. An analysis of the secondary structure of respective mRNAs showed that their ΔG values were–544.00 and–469.40 kcal/mol for EMCV IRES and BVDV IRES harboring molecules, respectively. As EMCV IRES-containing mRNA is more stable, it is evident that other, still unidentified factors should be held responsible for the enhanced translational efficiency of BDVD IRES. Taken together, our results indicate the potential of BVDV IRES as a replacement for EMCV IRES, which is now commonly used for T7 polymerase driven cytoplasmic expression of genes of interest or virus cDNA rescue experiments.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- BVDV:
-
bovine viral diarrhea virus
- EMCV:
-
encephalomyocarditis virus
- HCV:
-
hepatitis C virus
- IRES:
-
internal ribosome entry site
- MFE:
-
minimal free energy
- MFI:
-
mean fluorescence intensity
References
Bruter A.V., Avdeev A.V., Belyavsky A.V 2013. Regulated expression systems for gene therapy. Mol. Biol. (Moscow). 47, 321–342.
Lavigne M.D., Gorecki D.C 2006. Emerging vectors and targeting methods for nonviral gene therapy. Exp. Opin. Emerg. Drugs. 11, 541–557.
Dean D.A., Strong D.D., Zimmer W.E 2005. Nuclear entry of nonviral vectors. Gene Ther. 12, 881–890.
Finn J., Lee A.C., MacLachlan I., Cullis P 2004. An enhanced autogene-based dual-promoter cytoplasmic expression system yields increased gene expression. Gene Ther. 11, 276–283.
Aubry F., Nougairède A., Gould E.A., de Lamballerie X 2015. Flavivirus reverse genetic systems, construction techniques and applications: A historical perspective. Antivir. Res. 114, 67–85.
Farrow P.J., Barrett L.B., Stevenson M., Fisher K.D., Finn J., Spice R., Allan M.A., Berry M., Logan A., Seymour L.W., Read M.L 2006. Cytoplasmic expression systems triggered by mRNA yield increased gene expression in post-mitotic neurons. Nucleic Acids Res. 34, 80.
Structural Genomics Consortiu 2008. Protein production and purification. Nat. Methods. 5, 135–146.
Ghaderi M., Sabahi F., Sadeghi-Zadeh M., Khanlari Z., Jamaati A., Mousavi-Nasab D., Majidi-Gharenaz N., Ajorloo M., Fazeli M 2014. Construction of an eGFP expression plasmid under control of T7 promoter and IRES sequence for assay of T7 RNA polymerase activity in mammalian cell lines. Iran J. Cancer Prev. 7, 137–141.
Chen, X., Li Y., Xiong K., Wagner T.E 1994. A selfinitiating eukaryotic transient gene expression system based on contransfection of bacteriophage T7 RNA polymerase and DNA vectors containing a T7 autogene. Nucleic Acids Res. 22, 2114–2120.
Zheng, H., Tian H., Jin Y., Wu J., Shang Y., Yin S., Liu X., Xie Q 2009. Development of a hamster kidney cell line expressing stably T7 RNA polymerase using retroviral gene transfer technology for efficient rescue of infectious foot-and-mouth disease virus. J. Virol. Methods. 156, 129–137.
Kohl A., Billecocq A., Préhaud C., Yadani F.Z., Bouloy M 1999. Transient gene expression in mammalian and mosquito cells using a recombinant Semliki Forest virus expressing T7 RNA polymerase. Appl. Microbiol. Biotechnol. 53, 51–56.
Belsham G.J., Sonenberg N 2000. Picornavirus RNA translation: Roles for cellular proteins. Trends Microbiol. 8, 330–335.
Merrick W.C 2004. Cap-dependent and cap-independent translation in eukaryotic systems. Gene. 332, 1–11.
Komar A.A., Hatzoglou M 2011. Cellular IRES-mediated translation: The war of ITAFs in pathophysiological states. Cell Cycle. 10, 229–240.
Belsham G.J 2009. Divergent picornavirus IRES elements. Virus Res. 139, 183–192.
Niepmann M 2009. Internal translation initiation of picornaviruses and hepatitis C virus. Biochim. Biophys. Acta. 1789, 529–541.
Elroy-Stein O., Fuerst T.R., Moss B 1989. Cap-independent translation of mRNA conferred by encephalomyocarditis virus 5’ sequence improves the performance of the vaccinia virus/bacteriophage T7 hybrid expression system. Proc. Natl. Acad. Sci. U. S. A. 86, 6126–6130.
Borman A.M., Bailly J.L., Girard M., Kean K.M 1995. Picornavirus internal ribosome entry segments: Comparison of translation efficiency and the requirements for optimal internal initiation of translation in vitro. Nucleic Acids Res. 23, 3656–3663.
Bouabe H., Fassler R., Heesemann J 2008. Improvement of reporter activity by IRES-mediated polycistronic reporter system. Nucleic Acids Res. 36, 28.
Scheper G.C., Thomas A.A., Voorma H.O 1991. The 5’ untranslated region of encephalomyocarditis virus contains a sequence for very efficient binding of eukaryotic initiation factor eIF-2/2B. Biochim. Biophys. Acta. 1089, 220–226.
Brown E.A., Zajac A.J., Lemon S.M 1994. In vitro characterization of an internal ribosomal entry site (IRES) present within the 5’ nontranslated region of hepatitis A virus RNA: Comparison with the IRES of encephalomyocarditis virus. J. Virol. 68, 1066–1074.
Borman, A.M., Le Mercier P., Girard M., Kean K.M 1997. Comparison of picornaviral IRES-driven internal initiation of translation in cultured cells of different origins. Nucleic Acids Res. 25, 925–932.
Bochkov Y.A., Palmenberg A.C 2006. Translational efficiency of EMCV-IRES in bicistronic vectors is dependent upon IRES sequence and gene location. Biotechniques. 41, 283–288.
Hennecke M., Kwissa M., Metzger K., Oumard A., Kröger A., Schirmbeck R., Reimann J., Hauser H 2001. Composition and arrangement of genes define the strength of IRES-driven translation in bicistronic mRNAs. Nucleic Acids Res. 29, 3327–3334.
Ngoi S.M., Chien A.C., Lee C.G 2004. Exploiting internal ribosome entry sites in gene therapy vector design. Curr. Gene Ther. 4, 15–31.
Lancaster A.M., Jan E., Sarnow P 2006. Initiation factor-independent translation mediated by the hepatitis C virus internal ribosome entry site. RNA. 12, 894–902.
Chon S.K., Perez D.R., Donis R.O 1998. Genetic analysis of the internal ribosome entry segment of bovine viral diarrhea virus. Virology. 251, 370–382.
Moes L., Wirth M 2007. The internal initiation of translation in bovine viral diarrhea virus RNA depends on the presence of an RNA pseudoknot upstream of the initiation codon. Virol. J. 4, 124.
Il’ichev A.A., Melamed N.V., Razumov I.A., Maksiutov A.Z., Pereboev A.V., Zakabunin A.I., Loktev V.B 1996. Expression of fragments of Venezuelan equine encephalomyelitis virus E2 protein in a system based on phage T7 RNA polymerase. Mol. Biol. (Moscow). 30, 76–83.
Baghbani-Arani F., Roohvand F, Aghasadeghi M.R., Eidi A., Amini S., Motevalli F., Sadat S.M., Memarnejadian A., Khalili G 2012. Expression and characterization of Escherichia coli derived hepatitis C virus ARFP/F protein. Mol. Biol. (Moscow). 46, 226–235.
Forster A.C 2012. Synthetic biology challenges longheld hypotheses in translation, codon bias and transcription. Biotechnol J. 7, 835–845.
Zavil’gel’skii G.B., Rastorguev S.M 2009. Antirestriction proteins ardA and Ocr as effective inhibitors of the type I restriction-modification enzymes. Mol. Biol. (Moscow). 43, 241–248.
Herfst S., de Graaf M., Schickli J.H., Tang R.S., Kaur J., Yang C.F., Spaete R.R., Haller A.A., Van den Hoogen B.G., Osterhaus A.D., Fouchier R.A 2004. Recovery of human metapneumovirus genetic lineages a and B from cloned cDNA. J. Virol. 78, 8264–8270.
Jiang Y., Liu H., Liu P., Kong X 2009. Plasmids driven minigenome rescue system for Newcastle disease virus V4 strain. Mol. Biol. Rep. 36, 1909–1914.
de Wit E., Spronken M.I., Vervaet G., Rimmelzwaan G.F., Osterhaus A.D., Fouchier R.A 2007. A reverse-genetics system for influenza A virus using T7 RNA polymerase. J. Gen. Virol. 88, 1281–1287.
Hobl B., Hock B., Schneck S., Fischer R., Mack M 2013. Bacteriophage T7 RNA polymerase-based expression in Pichia pastoris. Protein Expr. Purif. 92, 100–104.
Xiu M.H., Wang Q., Tang L.H., Cao S.C., Li W.H., Wei Y., Lu P., Liang M.F., Li D.X 2007. Rescue of minireplicon by using the cell line stably expressing the T7 RNA polymerase. Bing Du Xue Bao. 23, 326–330.
Sambrook J., Fritsch E.F., Maniatis T 1989. Molecular Cloning. A Laboratory Manual. Cold Spring Harbor, NY: Cold Spring Harbor Laboratory Press, vol. 2.
Rasekhian M., Roohvand F., Teimoori-Toolabi L., Amini S., Azadmanesh K 2014. Application of the 3’-noncoding region of poliovirus RNA for cell-based regulation of mRNA stability: Implication for biotechnological applications. Biotechnol. Appl. Biochem. 61, 699–706.
Regier N., Frey B 2010. Experimental comparison of relative RT-qPCR quantification approaches for gene expression studies in poplar. BMC Mol. Biol. 11, 57.
Kaufman J., Cho M W., Lee K M., Shiloach J 2002. Large-scale production of HIV-I gp120 using recombinant vaccinia virus in packed-bed bioreactor. In: Animal Cell Technology: Products from Cells, Cells as Products. Bernard A. et al. Eds. Netherlands: Springer, pp. 289–291.
Wong E.T., Ngoi S.M., Lee C.G 2002. Improved coexpression of multiple genes in vectors containing internal ribosome entry sites (IRESes) from human genes. Gene Ther. 9, 337–344.
Ghim C.-M., Lee S.K., Takayama S., Mitchell R.J 2010. The art of reporter proteins in science: Past, present and future applications. BMB Rep. 43, 451–460.
Jiang T., Xing B., Rao J 2008. Recent developments of biological reporter technology for detecting gene expression. Biotechnol. Genet. Eng. Rev. 25, 41–76.
Hoffmann B., Depner K., Schirrmeier H., Beer M 2006. A universal heterologous internal control system for duplex real-time RT-PCR assays used in a detection system for pestiviruses. J. Virol. Methods. 136, 200–209.
Gao X., Huang L 1993. Cytoplasmic expression of a reporter gene by co-delivery of T7 RNA polymerase and T7 promoter sequence with cationic liposomes. Nucleic Acids Res. 21, 2867–2872.
Chu L., Robinson D.K 2001. Industrial choices for protein production by large-scale cell culture. Curr. Opin. Biotechnol. 12, 180–187.
Jayapal K.P., Wlaschin K.F., Hu W.S., Yap M.C.S 2007. Recombinant protein therapeutics from CHO cells: 20 years and counting, Chem. Engin. Progr. 103(10), 40–47.
Nevins T.A., Harder Z.M., Korneluk R.G., Holcík M 2003. Distinct regulation of internal ribosome entry site-mediated translation following cellular stress is mediated by apoptotic fragments of eIF4G translation initiation factor family members eIF4GI and p97/DAP5/NAT1. J. Biol. Chem. 278, 3572–3579.
Pause A., Méthot N., Svitkin Y., Merrick W.C., Sonenberg N 1994. Dominant negative mutants of mammalian translation initiation factor eIF-4A define a critical role for eIF-4F in cap-dependent and cap-independent initiation of translation. EMBO J. 13, 1205–1215.
Pestova T.V., Shatsky I.N., Fletcher S.P., Jackson R.J., Hellen C.U 1998. A prokaryotic-like mode of cytoplasmic eukaryotic ribosome binding to the initiation codon during internal translation initiation of hepatitis C and classical swine fever virus RNAs. Genes Dev. 12, 67–83.
Masuda K., Marasa B., Martindale J.L., Halushka M.K., Gorospe M 2009. Tissue-and age-dependent expression of RNA-binding proteins that influence mRNA turnover and translation. Aging (Albany, NY). 1, 681–698.
Landgraf P., Rusu M., Sheridan R., Sewer A., Iovino N., Aravin A., Pfeffer S., Rice A., Kamphorst A.O., Landthaler M., Lin C., Socci N.D., Hermida L., Fulci V., Chiaretti S., et al 2007. A mammalian microRNA expression atlas based on small RNA library sequencing. Cell. 129, 1401–1414.
Schult Ph., Lohmann V 2015. Dissecting the roles of of miR122 in different steps of HCV life cycles. Proc. 22nd International Symposium on HCV and Related Viruses, Strasbourg, France (October 9–13, 2015).
Brown D.M., Kauder S.E., Cornell C.T., Jang G.M., Racaniello V.R., Semler B.L 2004. Cell-dependent role for the poliovirus 3’ noncoding region in positivestrand RNA synthesis. J. Virol. 78, 1344–1351.
Sanderbrand S.A., Tautz N., Thiel H.J., Ochs K., Beck E., Niepmann M 2000. Translation from the internal ribosome entry site of bovine viral diarrhea virus is independent of the interaction with polypyrimidine tract-binding protein. Vet. Microbiol. 77, 215–227.
Ray P.S., Das S 2002. La autoantigen is required for the internal ribosome entry site-mediated translation of Coxsackievirus B3 RNA. Nucleic Acids Res. 30, 4500–4508.
Ali N., Siddiqui A 1997. The La antigen binds 5’ noncoding region of the hepatitis C virus RNA in the context of the initiator AUG codon and stimulates internal ribosome entry site-mediated translation. Proc. Natl. Acad. Sci. U. S. A. 94, 2249–2254.
Myers T.M., Kolupaeva V.G., Mendez E., Baginski S.G., Frolov I., Hellen C.U., Rice C.M 2001. Efficient translation initiation is required for replication of bovine viral diarrhea virus subgenomic replicons. J. Virol. 75, 4226–4238.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Molekulyarnaya Biologiya, 2017, Vol. 51, No. 2, pp. 324–333.
The article is published in the original.
Rights and permissions
About this article
Cite this article
Ghassemi, F., Madadgar, O., Roohvand, F. et al. Translational efficiency of BVDV IRES and EMCV IRES for T7 RNA polymerase driven cytoplasmic expression in mammalian cell lines. Mol Biol 51, 283–292 (2017). https://doi.org/10.1134/S002689331702011X
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S002689331702011X