Abstract
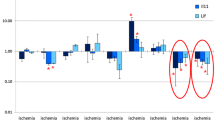
Neurotrophins stimulate the regeneration of neural tissue after lesions. It is also known that the sources of neurogenesis and cerebral function recovery are predominantly located in subcortical brain structures. The effects of ischemia on the expression of genes that encode neurotrophins (Bdnf, Ngf, Nt-3) and their receptors (TrkB, TrkA, TrkC, p75) in brain structures outside the lesion site were studied 3, 24, and 72 h after irreversible unilateral occlusion of the middle cerebral artery in rats. Changes in the mRNA expression of these genes were assessed by relative quantification using real-time RT-PCR. Sham surgery was found to stimulate the expression of genes that encode neurotrophins (Bdnf, Ngf) and their receptor (p75). It has been shown that ischemia influenced the expression of neurotrophins (Bdnf, Ngf, Nt-3) and their receptors (TrkB, TrkA, TrkC, p75) in brain structures outside the lesion focus, including the contralateral hemisphere. The downregulation of Bdnf and TrkB transcripts and Ngf and TrkA upregulation in the contralateral cortex on the first day of ischemia obviously reflected stress response. On day 3, Nt-3 transcription increased in all investigated structures outside the lesion focus. In the contralateral hemisphere, relative levels of TrkA and TrkC mRNA expression increased, while p75 expression decreased. Presumably, the observed changes in gene transcription serve to facilitate neuroplasticity and neural tissue regeneration.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Temple S., Qian X. 1995. bFGF, neurotrophins, and the control or cortical neurogenesis. Neuron. 15 (2), 249–252.
Beck T., Lindholm D., Castrén E., Wree A. 1994. Brain-derived neurotrophic factor protects against ischemic cell damage in rat hippocampus. J. Cereb. Blood Flow Metab. 14 (4), 689–692.
Hayashi T., Abe K., Itoyama Y. 1998. Reduction of ischemic damage by application of vascular endothelial growth factor in rat brain after transient ischemia. J. Cereb. Blood Flow Metab. 18 (8), 887–895.
Wang J.M., Hayashi T., Zhang W.R., Sakai K., Shiro Y., Abe K. 2000. Reduction of ischemic brain injury by topical application of insulin-like growth factor-I after transient middle cerebral artery occlusion in rats. Brain Res. 859 (2), 381–385.
Schäbitz W.R., Sommer C., Zoder W., Kiessling M., Schwaninger M., Schwab S. 2000. Intravenous brainderived neurotrophic factor reduces infarct size and counterregulates Bax and Bcl-2 expression after temporary focal cerebral ischemia. Stroke. 31 (9), 2212–2217.
Daniela F., Vescovi A.L., Bottai D. 2007. The stem cells as a potential treatment for neurodegeneration. Methods Mol. Biol. 399, 199–213.
Gritti A., Frölichsthal-Schoeller P., Galli R., Parati E.A., Cova L., Pagano S.F., Bjornson C.R., Vescovi A.L. 1999. Epidermal and fibroblast growth factors behave as mitogenic regulators for a single multipotent stem cell-like population from the subventricular region of the adult mouse forebrain. J. Neurosci. 19 (9), 3287–3297.
Zhang R.L., Zhang Z.G., Zhang L., Chopp M. 2001. Proliferation and differentiation of progenitor cells in the cortex and the subventricular zone in the adult rat after focal cerebral ischemia. Neuroscience. 105 (1), 33–41.
Zhang R.L., Chopp M., Roberts C., Liu X., Wei M., Nejad-Davarani S.P., Wang X., Zhang Z.G. 2014. Stroke increases neural stem cells and angiogenesis in the neurogenic niche of the adult mouse. PLOS ONE. 9 (12), e113972.
Craig C.G., Tropepe V., Morshead C.M., Reynolds B.A., Weiss S., van der Kooy D. 1996. In vivo growth factor expansion of endogenous subependymal neural precursor cell populations in the adult mouse brain. J. Neurosci. 16 (8), 2649–2658.
Fallon J., Reid S., Kinyamu R., Opole I., Opole R., Baratta J., Korc M., Endo T.L., Duong A., Nguyen G., Karkehabadhi M., Twardzik D., Patel S., Loughlin S. 2000. In vivo induction of massive proliferation, directed migration, and differentiation of neural cells in the adult mammalian brain. Proc. Natl. Acad. Sci. U. S. A. 97 (26), 14686–14691.
Kokaia Z., Gidö G., Ringstedt T., Bengzon J., Kokaia M., Siesjö B.K., Persson H., Lindvall O. 1993. Rapid increase of BDNF mRNA levels in cortical neurons following spreading depression: Regulation by glutamatergic mechanisms independent of seizure activity. Brain Res. Mol. Brain Res. 19 (4), 277–286.
Branston N.M., Strong A.J., Symon L. 1977. Extracellular potassium activity, evoked potential and tissue blood flow. Relationships during progressive ischaemia in baboon cerebral cortex. J. Neurol Sci. 32 (3), 305–321.
Kitagawa K., Matsumoto M., Kuwabara K., Tagaya M., Ohtsuki T., Hata R., Ueda H., Handa N., Kimura K., Kamada T. 1991. Ischemic tolerance phenomenon detected in various brain regions. Brain Res. 561 (2), 203–211.
Dietrich W.D., Feng Z.C., Leistra H., Watson B.D., Rosenthal M. 1994. Photothrombotic infarction triggers multiple episodes of cortical spreading depression in distant brain regions. J. Cereb. Blood Flow Metab. 14 (1), 20–28.
Gusev E.I., Skvortsova V.I. 2001. Ishemiya golovnogo mozga (Cerebral Ischemia). Moscow: Meditsina.
Holtzman D.M., Sheldon R.A., Jaffe W., Cheng Y., Ferriero D.M. 1996. Nerve growth factor protects the neonatal brain against hypoxic-ischemic injury. Ann. Neurol. 39 (1), 114–122.
Wu D. 2005. Neuroprotection in experimental stroke with targeted neurotrophins. NeuroRx. 2 (1), 120–128.
Zhang Z.H., Wang R.Z., Wang R.Z., Li G.L., Wei J.J., Li Z.J., Feng M., Kang J., Du W.C., Ma W.B., Li Y.N., Yang Y., Kong Y.G. 2008. Transplantation of neural stem cells modified by human neurotrophin-3 promotes functional recovery after transient focal cerebral ischemia in rats. Neurosci. Lett. 444 (3), 227–230.
Kitagawa H., Abe K., Hayashi T., Mitsumoto Y., Koga N., Itoyama Y. 1998. Ameliorative effect of glial cell linederived neurotrophic factor on brain edema formation after permanent middle cerebral artery occlusion in rats. Neurol. Res. 20 (4), 333–336.
Kitagawa H., Hayashi T., Mitsumoto Y., Koga N., Itoyama Y., Abe K. 1998. Reduction of ischemic brain injury by topical application of glial cell line-derived neurotrophic factor after permanent middle cerebral artery occlusion in rats. Stroke. 29 (7), 1417–1422.
Dmitrieva V.G., Povarova O.V., Skvortsova V.I., Limborska S.A., Myasoedov N.F., Dergunova L.V. 2010. Semax and Pro-Gly-Pro activate the transcription of neurotrophins and their receptor genes after cerebral ischemia. Cell. Mol. Neurobiol. 30 (1), 71–79.
Povarova O.V., Garibova T.L., Kalenikova E.I., Galaeva I.P., Kraineva V.A., Medvedev O.S., Voronina T.A. 2004. Effect of phenyl-tert-butylnitrone, mexidol and nooglutil on the ischemic lesion zone and memory in rats following middle cerebral artery occlusion. Eksp. Klin. Farmakol. 67 (1), 3–6.
Schwarting R., Huston J.P. 1987. Short-term effects of ether, equithesin and droperidol/fentanyl on catecholamine and indolamine metabolism in the brain of the rat, Neuropharmacol. 26 (5), 457–461.
Schultz V.L., Boass A., Garner S.C., Toverud S.U. 1995. Several anesthetics, but not diethyl ether, cause marked elevation of serum parathyroid hormone concentration in rats. J. Bone Miner. Res. 10 (9), 1298–1302.
Dmitrieva V.G., Torshina E.V., Yuzhakov V.V., Povarova O.V., Skvortsova V.I., Limborska S.A., Dergunova L.V. 2008. Expression of sphingomyelin synthase 1 gene in rat brain focal ischemia. Brain Res. 1188, 222–227.
Chomczynski P., Sacchi N. 1987. Single-step method of RNA isolation by acid guanidinium thiocyanate–phenol–chloroform extraction. Anal. Biochem. 162, 156–159.
Maniatis T., Fritsch E.F., Sambrook J. 1982. Molecular Cloning: A Laboratory Manual. Cold Spring Harbor, NY: Cold Spring Harbor Lab. Press.
Stavchansky V.V., Tvorogova T.V., Botsina A.Yu., Skvortsova V.I., Limborska S.A., Myasoedov N.F., Dergunova L.V. Effect of Semax and its C-terminal peptide PGP on expression of neurotrophins and their receptors in rat brain during incomplete global ischemia. Mol. Biol. (Moscow). 45 (6), 941–949.
Medhurst A.D., Harrison D.C., Read S.J., Campbell C.A., Robbins M.J., Pangalos M.N. 2000. The use of Taq Man RT-PCR assays for semiquantitative analysis of gene expression in CNS tissues and disease models. J. Neurosci. Methods. 98, 9–20.
Harrison D.C., Medhurst A.D., Bond B.C., Campbell C.A., Davis R.P., Philpott K.L. 2000. The use of quantitative RT-PCR to measure mRNA expression in a rat model of focal ischemia: Caspase 3 as a case study. Mol. Brain. Res. 75, 143–149.
Pfaffl M.W. 2001. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 29 (9), 2002–2007.
Pfaffl M.W., Horgan G.W., Dempfle L. 2002. Relative expression software tool (REST) for group-wise comparison and statistical analysis of relative expression results in real-time PCR. Nucleic Acids Res. 30 (9), 36.
Smith M.A., Makino S., Kvetnansky R., Post R.M. 1995. Stress and glucocorticoids affect the expression of brain-derived neurotrophic factor and neurotrophin-3 mRNAs in the hippocampus. J. Neurosci. 15 (3, Pt. 1), 1768–1777.
Molteni R., Calabrese F., Cattaneo A., Mancini M., Gennarelli M., Racagni G., Riva M.A. 2009. Acute stress responsiveness of the neurotrophin BDNF in the rat hippocampus is modulated by chronic treatment with the antidepressant duloxetine. Neuropsychopharmacology. 34 (6), 1523–1532.
Lee T.H., Kato H., Chen S.T., Kogure K., Itoyama Y. 1998. Expression of nerve growth factor and trkA after transient focal cerebral ischemia in rats. Stroke. 29 (8), 1687–1696.
Lindvall O., Ernfors P., Bengzon J., Kokaia Z., Smith M.L., Siesjö B.K., Persson H. 1992. Differential regulation of mRNAs for nerve growth factor, brain-derived neurotrophic factor, and neurotrophin 3 in the adult rat brain following cerebral ischemia and hypoglycemic coma. Proc. Natl. Acad Sci. U. S. A. 89 (2), 648–652.
Takeda A., Onodera H., Sugimoto A., Kogure K., Obinata M., Shibahara S. 1993. Coordinated expression of messenger RNAs for nerve growth factor, brain-derived neurotrophic factor and neurotrophin-3 in the rat hippocampus following transient forebrain ischemia. Neuroscience. 55 (1), 23–31.
Tokumine J., Kakinohana O., Cizkova D., Smith D.W., Marsala M. 2003. Changes in spinal GDNF, BDNF, and NT-3 expression after transient spinal cord ischemia in the rat. J. Neurosci Res. 74 (4), 552–561.
Shimazu K., Zhao M., Sakata K., Akbarian S., Bates B., Jaenisch R., Lu B. 2006. NT-3 facilitates hippocampal plasticity and learning and memory by regulating neurogenesis. Learn. Mem. 13 (3), 307–315.
Kim M.W., Bang M.S., Han T.R., Ko Y.J., Yoon B.W., Kim J.H., Kang L.M., Lee K.M., Kim M.H. 2005. Exercise increased BDNF and trkB in the contralateral hemisphere of the ischemic rat brain. Brain Res. 1052 (1), 16–21.
Kokaia Z., Zhao Q., Kokaia M., Elmér E., Metsis M., Smith M.L., Siesjo B.K., Lindvall O. 1995. Regulation of brain-derived neurotrophic factor gene expression after transient middle cerebral artery occlusion with and without brain damage. Exp. Neurol. 136 (1), 73–88.
Merlio J.P., Ernfors P., Kokaia Z., Middlemas D.S., Bengzon J., Kokaia M., Smith M.L., Siesjö B.K., Hunter T., Lindvall O., Persson H. 1993. Increased production of the TrkB protein tyrosine kinase receptor after brain insults. Neuron. 10 (2), 151–164.
Arai S., Kinouchi H., Akabane A., Owada Y., Kamii H., Kawase M., Yoshimoto T. 1996. Induction of brainderived neurotrophic factor (BDNF) and the receptor trk B mRNA following middle cerebral artery occlusion in rat. Neurosci. Lett. 211 (1), 57–60.
Majda B.T., Meloni B.P., Rixon N., Knuckey N.W. 2001. Suppression subtraction hybridization and northern analysis reveal upregulation of heat shock, trkB, and sodium calcium exchanger genes following global cerebral ischemia in the rat. Brain Res. Mol. Brain Res. 93 (2), 173–179.
Chung J.Y., Kim M.W., Bang M.S., Kim M. 2010. The effect of exercise on trkA in the contralateral hemisphere of the ischemic rat brain. Brain Res. 1353, 187–193.
Roux P.P., Barker P.A. 2002. Neurotrophin signaling through the p75 neurotrophin receptor. Prog. Neurobiol. 67 (3), 203–233.
Yoon S.O., Casaccia-Bonnefil P., Carter B., Chao M.V. 1998. Competitive signaling between TrkA and p75 nerve growth factor receptors determines cell survival. J. Neurosci. 18 (9), 3273–3281.
Kokaia Z., Andsberg G., Martinez-Serrano A., Lindvall O. 1998. Focal cerebral ischemia in rats induces expression of P75 neurotrophin receptor in resistant striatal cholinergic neurons. Neuroscience. 84 (4), 1113–1125.
Ibánez C.F., Simi A. 2012. p75 neurotrophin receptor signaling in nervous system injury and degeneration: Paradox and opportunity. Trends Neurosci. 35 (7), 431–440.
Kokaia Z., Andsberg G., Yan Q., Lindvall O. 1998. Rapid alterations of BDNF protein levels in the rat brain after focal ischemia: Evidence for increased synthesis and anterograde axonal transport. Exp. Neurol. 154 (2), 289–301.
Issa R., Krupinski J., Bujny T., Kumar S., Kaluza J., Kumar P. 1999. Vascular endothelial growth factor and its receptor, KDR, in human brain tissue after ischemic stroke. Lab. Invest. 79 (4), 417–425.
Keyvani K., Witte O.W., Paulus W.J. 2002. Gene expression profiling in perilesional and contralateral areas after ischemia in rat brain. Cereb. Blood Flow Metab. 22 (2), 153–160.
Keyvani K., Witte O.W., Reinecke S., Paulus W. 2002. Use-related gene expression patterns of rat motor cortex. NeuroReport. 13 (7), 953–956.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V.G. Dmitrieva, V.V. Stavchansky, O.V. Povarova, V.I. Skvortsova, S.A. Limborska, L.V. Dergunova, 2016, published in Molekulyarnaya Biologiya, 2016, Vol. 50, No. 5, pp. 775–784.
Rights and permissions
About this article
Cite this article
Dmitrieva, V.G., Stavchansky, V.V., Povarova, O.V. et al. Effects of ischemia on the expression of neurotrophins and their receptors in rat brain structures outside the lesion site, including on the opposite hemisphere. Mol Biol 50, 684–692 (2016). https://doi.org/10.1134/S0026893316030067
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0026893316030067