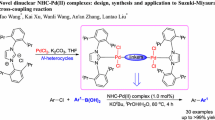
Abstract
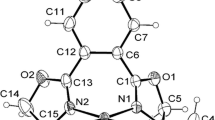
The preparation and characterization of a series of new 2-phenylpyridine derivative ligands consisting of 2-(R) pyridine (R = mesityl (L1), 2,6-dimethylphenyl (L2), o-tolyl (L3), m-tolyl (L4), p-tolyl (L5), o-methoxyphenyl (L6), and p-methoxyphenyl (L7)) and their Pd complexes [PdCl2L2] (L1–L7) is investigated using a combination of X-ray diffraction spectroscopy, GC-MS, and NMR. The crystal structures show that the Pd complexes adopt a square planar geometry, and the monodentate ligand is coordinated through the N donor of the pyridine ring to the Pd atom. The catalytic activities of the synthesized complexes are investigated. The square planar Pd complex trans-[(2-mesitylpy)2PdCl2)] shows a high efficiency in promoting Suzuki-Miyaura cross coupling in an aqueous solvent under aerobic conditions.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Handbook of OrganoPd Chemistry for Organic Synthesis/Ed. E.-i Negishi.; Wiley-Interscience: New York, 2002.
N. Miyaura. Cross-Coupling Reactions: A Practical Guide. Springer: New York, 2002.
A. Suzuki and H. C. Brown. Organic Synthesis via Boranes. Aldrich Chemical Co: Milwaukee, WI, 2002, 3.
A. Suzuk. Angew. Chem. Int., Ed., 2011, 50, 6722–6737.
H. Valdés, D. Canseco-González, J. M. Germán-Acacio, and D. Morales-Morales. J. Organomet. Chem., 2018, 867, 51.
K. O. Marichev, S. A. Patil, and A. Bugarin. Tetrahedron, 2018, 74, 2523.
Y. Zhang and G. Lavigne. Eur. J. Inorg. Chem., 2015, 2042–2050.
M. E. Hanhan. Appl. Organomet. Chem., 2008, 22, 270–275.
Y. Lai, Z. Zong, Y. Tang, W. Mo, N. Sun, B. Hu, Z. Shen, L. Jin, W. Sun, and X. Hu. Beilstein J. Org. Chem., 2017, 13, 213–221.
S. Ghosh, A. S. Kumar, and G. N. Mehta. Beilstein J. Org. Chem., 2010, 4, 4–7.
L. J. Goossen and B. Melzer. J. Org. Chem., 2007, 7473–7476.
H. Khojasteh, V. Mirkhani, M. Moghadam, S. Tangestaninejad, and I. Mohammadpoor-Baltork. J. Nano Struct., 2015, 5, 271.
I. Maluenda and O. Navarro. Molecules, 2015, 20, 7528.
Y.-N. Yu and M.-H. Xu. Acta Chim. Sin., 2017, 75, 655–670.
R. E. Andrew, L. Gonzalez-Sebastian, and A. B. Chaplin. Dalton Trans., 2016, 45, 1299–1305.
A. Fihri, P. Meunier. and J. Hierso. Coord. Chem. Rev., 2007, 251, 2017–2055.
W. A. Herrmann and C. Köcher. Angew. Chem., Int. Ed. Engl., 1997, 36, 2162.
D. Bourissou, O. Guerret, F. P. Gabbaï, and G. Bertrand. Chem. Rev., 2000, 100, 39.
I. J. B. Lin and C. S. Vasam. Coord. Chem. Rev., 2007, 251, 642–670.
C. E. Willans, K. M. Anderson, M. J. Paterson, P. C. Junk, L. J. Barbour, and J. W. Steed. Eur. J. Inorg. Chem., 2009, 2835.
Z. Guan, J. Hu, Y. Gu, H. Zhang, G. Li, and T. Li. Angew. Chem., Int. Ed., 2006, 45, 1282–1284.
Z. Z. Zhou, F. S. Liu, D. S. Shen, C. Tan, and L. Y. Luo. Inorg. Chem. Commun., 2011, 14, 659.
I. D. Kostas, A. G. Coutsolelos, G. Charalambidis, and A. Skondra. Tetrahedron Lett., 2007, 48, 6688–6691.
D. H. Lee and M. J. Jin. Org. Lett., 2011, 13, 252–255.
E. Amadio, M. Bertoldini, A. Scrivanti, G. Chessa, V. Beghetto, U. Matteoli, R. Bertani, and A. Dolmella. Inorg. Chim. Acta, 2011, 370, 388–393.
E. Amadio, A. Scrivanti, G. Chessa, U. Matteoli, V. Beghetto, M. Bertoldini, M. Rancan, A. Dolmella, A. Venzo, and R. Bertani. J. Organomet. Chem., 2012, 716, 193–200.
P. Liu, M. Yan, and R. He. Appl. Organomet. Chem., 2010, 24, 131–134.
L. Shen, S. Huang, Y. Nie, and F. Lei. Molecules, 2013, 18, 1602–1612.
V. Montoya, J. Pons, V. Branchadell, J. Garcia-Antón, X. Solans, M. Font-Bardia, and J. Ros. Organometallics, 2008, 27, 1084–1091.
J. A. Perez, J. Pons, X. Solans, M. Font-Bardia, and J. Ros. Inorg. Chim. Acta, 2005, 358, 617–622.
I. A. Mkhalid and H. F. Al-shaikh. Asian J. Chem., 2014, 26, 2077–2082.
A. Suzuki. In: Handbook of OrganoPd Chemistry for Organic Synthesis/Ed. E.-i. Negishi. Wiley Interscience: New-York, 2002, Ch. III.2.2.
J. C. Anderson, H. Namli, and C. A. Roberts. Tetrahedron, 1997, 53, 15123–15134.
H. Tatamidani, F. Kakiuchi, and N. Chatani. Org. Lett., 2004, 6, 3597–3599.
G. A. Anderson and M. Lie. Inorg. Synthesis, 1990, 28, 61.
Agilent. CrysAlis PRO. Agilent Technologies, Yarnton, England, 2012.
G. M. Sheldrick. SHELXS, SHELXL. Acta Crystallogr., 2008, A64, 112.
L. J. Barbour. J. Supramol. Chem., 2001, 1, 189.
O. V. Dolomanov, L. J. Bourhis, R. J. Gildea, J. A. K. Howard, and H. Puschmann. J. Appl. Crystallogr., 2009, 42, 339–341.
C. A. Tolman. Chem. Rev., 1977, 77, 313–348.
A. de Meijere, F. Diederich. Metal-catalyzed Cross-coupling Reactions. Wiley-VCH: Weinheim, 2004.
N. J. Whitcome, K. K. Hii, and S. E. Gibson. Tetrahedron, 2001, 57, 7449–7476.
Acknowledgments
The authors acknowledge DSR for technical and financial support. Abdulmuizz Adamson is thankful to King Abdulaziz University for providing a scholarship.
Funding
This project was funded by the Deanship of Scientific Research (DSR) at King Abdulaziz University, Jeddah, under grant no. 1438/130/227.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interests.
Additional information
Text © The Author(s), 2020, published in Zhurnal Strukturnoi Khimii, 2020, Vol. 61, No. 3, pp. 489–497.
Rights and permissions
About this article
Cite this article
Adamson, A., Budiman, Y.P., Mkhalid, I. et al. Palladium (II) Complexes Containing 2-Phenylpyridine Derivatives: Synthesis, Molecular Structures, and Catalytic Activity for Suzuki–Miyaura Cross-Coupling Reactions. J Struct Chem 61, 466–475 (2020). https://doi.org/10.1134/S0022476620030130
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476620030130