Abstract
Transcutaneous electrical spinal cord stimulation (SсТS) was carried out in different phases of the stepping cycle in order to control the kinematic parameters of the step in healthy subjects walking on a treadmill. ScTS during the swing phase at the T11–T12 level activated the flexor motor pools and caused a change in the motion amplitude in the hip, knee and ankle joints, as well as increased the height of leg elevation. ScTS at the stance phase at the L1–L2 level, addressed to the extensor motor pools, did not affect the kinematics of stepping movements. A shift in the start of T11–T12 stimulation to 100–150 ms before the initiation of the swing phase or its prolongation by 100 ms after the end of the swing phase caused significant changes in the kinematics of stepping movements. Essential for the start of stimulation is the moment of pushing the leg off the support a little earlier than beginning of the swing phase . Prolongation of the stimulation period in the swing phase allows to increase ankle join flexion The choice of the optimal algorithm of phase-dependent ScTS for the activation of flexor and extensor motor pools during the stepping cycle increases the efficiency of stimulation in motor function rehabilitation techniques.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
INTRODUCTION
Electrical spinal cord stimulation is one of the possible methods for the external control of human motor functions lost due to a disease or spinal cord trauma [1]. In the recent years, an active search is in progress for noninvasive techniques to affect neural structures of the spinal cord, and one of such techniques is transcutaneous electrical spinal cord stimulation (ScTS). This method is being successfully applied in complex rehabilitation of human and animal motor functions in clinical and experimental studies [2–5]. Currently, a new approach to using ScTS is being developed, namely, the possibility of a targeted impact on certain neural structures of the spinal cord. Thus, while stimulating various spinal cord levels (T10–L1), a relative selectivity of recruiting different motoneuron pools that innervate leg muscles was demonstrated, and the possibility of selective activation of flexor or extensor motor pools was suggested [6]. Since locomotion is a steady cyclic sequence of leg movements with alternating stance and swing phases that differ in functions and the degree of muscle activation, then to control the parameters of stepping movements, stimulatory effects should be targeted to the motor pools that provide a corresponding motor function. In this work, phase-dependent ScTS was applied at different levels of the spinal cord in the zone of input of the dorsal roots in order to control the kinematic parameters of human movements during walking. The present study aims to find the most adequate algorithm for such stimulation, namely, the answer to the question of how a change in the time limits of ScTS within the phases of the stepping cycle affects the motor response.
MATERIALS AND METHODS
Experiments involved healthy male volunteers (n = 5, aged 19–35 years). In accordance with the principles of the Declaration of Helsinki and the Russian Federation’s and international standards, written informed consent was obtained from each of the subjects to participate in the research. During the study, the subjects walked on a treadmill (h/p/cosmos gaitway®, Germany) moving at a speed 1.5–1.7 km/h. Against the background of a quiet walk, 30–60 s thereafter, ScTS started first for 30 s at a frequency of 15 Hz during the stance phase at the vertebral L1–L2 level to affect motor pools of the extensor muscles, then for the next 30 s, at a frequency of 30 Hz during the swing phase at the T11–T12 level to affect motor pools of the flexor muscles. Then ScTS was performed with alternating stimulation at L1 and T11 vertebral levels in the corresponding phases of the step cycle, i.e. alternate activation of the extensor and flexor muscles was performed. The detection of the stance and swing phases to trigger stimulation was carried out in real time while walking using specially designed sensors [7].
To execute ScTS, a five-channel electrical stimulator Bio-Stim-5 (Kosima Ltd.) [8] was used. Rectangular pulses with a duration of 1 ms were modulated by a frequency of 5 kHz; a stimulation frequency of 15 Hz for extensor pools and 30 Hz for flexor pools, and a stimulation intensity of 15–90 mA were used. Movements were video recorded using a 3D video analysis system (Qualisys, Sweden). Light-reflecting markers were fixed at the following bilateral anthropometric points: humeral acromial (shoulder joint), trochanteric (hip joint), upper tibia (knee joint), lower tibia (ankle joint), terminal (toe or hallux). The coordinates of the markers were used to calculate the trajectory of the leg movement and the following kinematic parameters of the step: changes in the angle amplitude in the hip, knee and ankle joints, as well as height of leg elevation determined by the coordinates of displacement of the marker fixed on the toe. Mathematical data processing was performed using original computer programs and Microsoft Excel. The average values and standard deviations of the parameters were calculated for each subject and compared to the values under initial conditions, i.e. during the first 30 s of the study, before the start of ScTS. The calculated relative values were averaged across all subjects. The statistical significance of changes in the parameters was assessed using the Student’s t-test, the differences were considered significant at p < 0.05.
RESULTS AND DISCUSSION
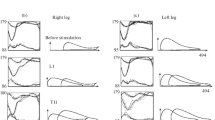
During experiments, it was established that ScTS influences on the kinematic parameters of stepping, while the ScTS’s effects at the L1 level in the stance phase and at the T11 level in the swing phase were expressed to a varying degree. Figure 1 shows the change in the join angles and the height of leg elevation during ScTS while walking, as recorded in one subject. The motion amplitude in joints, as well as the height of leg elevation, increased to a significantly larger extent with ScTS at T11 in the swing phase than with ScTS at L1 in the stance phase; a largest effect was observed at an alternating stimulation (L1 + T11).
Since the step is implemented by consecutive activation of different motor pools that innervate leg muscles, while the moments of the start and the end of stimulation within both phases can influence the kinematics of movement, we analyzed the dependence of the height of leg elevation on phase shifts of the ScTS. It is in changes in this parameter where the influence of ScTS parameters can manifest itself because, while walking on the moving treadmill, other kinematic parameters of movement are largely determined by the speed of the treadmill track.
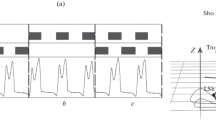
In the practice of phase-dependent ScTS, when the phase detection is performed in a real time, it is not always possible to accurately hit within the phase boundaries of the stepping cycle, due to which some shifts in the start and the end of stimulation arise relative to the stepping cycle phases (Fig. 2); the value of these shifts relies on the capabilities of the equipment. Since in our studies all the shifts were distributed randomly, with separate stimulations they could be considered as mutually independent characteristics, and the influence of each of the shifts on the stepping parameters could be analyzed separately.
The results of statistical analysis of changes in the height of leg elevation for each type of shift are shown in Fig. 3.
It can be seen that the effect of a height of leg elevation is present to a different ranges practically with all the shifts observed, however, the elevation value depends on shift values of the start and the end of ScST within the phase boundaries. For instance, during stimulation at L1 (stance phase), the shifts in the start of stimulation relative to the initiation of the stance phase have a negligible effect, while a delay in the end of stimulation, when it may already overlap the swing phase, causes a considerable decrease in the height of leg elevation, sometimes even stronger than while free walking. The maximum elevation is observed in the case of the end of stimulation 100–200 ms before the termination of the stance phase (–250…–150 ms in Fig. 3).
During stimulation in the swing phase (ScTS at T11), the effect is most pronounced when the start of stimulation precedes the beginning of swing phase by 200 ms (–250…–150 ms in Fig. 3) and the end is later by 100 ms (+50…+150 ms in Fig. 3). Combining the data of delays in different phases, we can conclude that stimulation in the stance phase should be finished 100 ms earlier, while in the swing phase, it should be started 100 ms earlier and continued for 100 ms longer.
As is well known, the stance phase accounts for 60%, and the swing phase 40% of the step cycle time. However, using multivariate analysis, 6 biomechanical phases are distinguished in the human walking stepping cycle. [9]. In the 4th phase (51–66% of the stepping cycle), the leg pushes off from the ground; this moment slightly precedes the beginning of the swing phase and, according to our data, is important for the start of stimulation. On the curve describing the vertical component of the stance reaction of the leg while walking, this time interval corresponds to the second maximum which reflects an active pushing-off of the leg from the surface, which causes the body movement upward and forward [10].
It is known that in the swing phase, at first a flexion of the hip joint, then a flexion of the knee joint with a coordinated action of the biceps femoris and semitendinosus, and finally a flexion of the ankle occur [11]. From our data, it follows that when stimulation is prolonged in the swing phase by 100 ms, the impact on these muscles increases, and the maximum muscular activity falls at the end and beggining of the stance phase. Extending of the stimulation period allows to affect the tibialis anterior muscle and increase plantar flexion.
A greater effect of stimulation in the swing phase can be explained by the fact that the extensor muscles provide a force component while the flexor muscles are corrective components of the locomotor synergy. According to their functions, the extensor muscles have a more rigid innervation program of walking while the flexor muscles have a more adaptive one. It was shown that proprioceptive afferents evoke monosynaptic responses in extensor neurons and activate flexor neurons through polysynaptic pathways [12].
In a number of works [9–11], functional electrical stimulation (FES) was applied to the muscles active in certain phases of the stepping cycle. FES was developed on the basis of gait biomechanics and neurophysiology and showed good results in correcting gait parameters in patients with motor disorders [10, 11, 13]. Currently, the gait analysis is used as one of the key methods for diagnosing motor pathology in clinical researches [14, 15]. In contrast to FES, phase-dependent ScTS, as a method of artificial movement correction, activates motor pools of the muscle complex, affecting the locomotor synergy control system, while with multisegmental ScTS, descending and ascending influences converge onto the neural networks responsible for postural and locomotor functions [16].
Phase-dependent ScTS enables a combination of natural and artificial activation of motor pools without disturbing the walking automatism, while the development of a correct algorithm opens new avenues for controlling the step characteristics, which can be used in developing new rehabilitation programs to restore human motor functions.
Changes in angular displacement of the hip (HIP), knee (KNEE) and ankle (ANKLE) joints and the magnitude of the elevation height of the right toe (based on the vertical toe coordinates, HRT) at ScTS while walking and stimulation at L1 (swing phase), T11 (stance phase), and alternating stimulation at L1 and T1. The moments of the beginning of stimulation are marked with arrows; subject Z.Yu., right leg.
Dependences of mean values of the ipsilateral leg elevation height within the step (h, %) on the shifts of the beginning and end of ScTS, as determined in 100-ms ranges relative to the phase boundaries; coincidence with the phase boundary corresponds to 0 (abscissa). The mean leg elevation height in steps before stimulation was taken as 100%. The data were averaged over 5 subjects; by 30 steps per stimulation for each subject. The top row: stimulation during the stance phase; the bottom row: stimulation during the swing phase.
REFERENCES
Harkema, S., Hodes, J., Angeli, C., Chen, Y., Ferreira, C., Willhite, A., Gerasimenko, Y.P., Edgerton, V. R., Burdick, J., Rejc, E., and Grossman, R.G., Effect of epidural stimulation of the lumbosacral spinal cord on voluntary movement, standing, and assisted stepping after motor complete paraplegia: a case study, Lancet, 2011, vol. 377, p. 1938. https://doi.org/10.1016/S0140-6736(11)60547-3
Dy, C.J., Gerasimenko, Y.P., Edgerton, V.R., Dyhre-Poulsen, P., Courtine, G., and Harkema, S.J., Phase-dependent modulation of percutaneously elicited multisegmental muscle responses after spinal cord injury, J. Neurophysiol., 2010, vol. 103(5), pp. 2808–2820. https://doi.org/10.1152/jn.00316.2009
Hofstoetter, U.S., Freundl, B., Binder, H., and Minassian, K., Recovery cycles of posterior root-muscle reflexes evoked by transcutaneous spinalcord stimulation and of the H reflex in individuals with intact and injured spinal cord, PLoS One, 2019, vol. 14(12), e0227057. https://doi.org/10.1371/journal.pone.0227057
Sayenko, D.G., Rath, M., Edgerton, V.R., Gerasimenko, Y.P., Ferguson, A.R., Burdick, J.W., Havton, L.A., Self-assisted standing enabled by non-invasive spinal stimulation after spinal cord injury, J. Neurotrauma, 2019, vol. 36(9), pp. 1435–1450. https://doi.org/10.1089/neu.2018.5956
Musienko, P.E., Bogacheva, I.N., Savochin, A.A., Kilimnik, V.A., Gorsky, O.V., Nikitin, O.A., and Gerasimenko, Yu.P., Non-invasive transcutaneous spinal cord stimulation facilitates locomotor activity in decerebrated and spinal cats, Russ. J. Physiol., 2013, vol. 99(8), pp. 917–927.
Sayenko, D.G., Atkinson, D.A., Dy, C.J., Gurley, K.M., Smith, V.L., Angeli, C., Harkema, S.J., Edgerton, V.R., and Gerasimenko, Y.P., Spinal segment-specific transcutaneous stimulation differentially shapes activation pattern among motor pools in humans, J. Appl. Physiol., 2015, vol. 118, pp. 1364–1374. https://doi.org/10.1152/japplphysiol.01128.2014
Grishin, A.A., Bobrova, E.V., Reshatnikova, V.V., Moshonkina, T.R., and Gerasimenko, Yu.P., The system for detecting the phases of stepping cycle and stimulating the spinal cord as instrument for human locomotion control, Med. Equipment, 2020, vol. 5, pp. 10–13.
Grishin, A.A., Moshonkina, T.R., and Solopova, I.A., The device for non-invasive electrical stimulation of the spinal cord, Med. Equipment, 2016, vol. 5, pp. 8–11.
Vitenzon, A.S. and Petrushanskaya, K.A., Physiological substantiation of the method of artificial movement correction by means of electrical muscle stimulation under walking, Russ. J. Biomech., 2010, vol. 14(48), pp. 7–27.
Ugnivenko, V.I., Foot kinematics and dynamics under walking, 2012. http://www.tri.by/News/Details/271.
Vitenzon, A.S. and Petrushanskaya, K.A., Phase analysis of walking and some rhythmical motions of man, Russ. J. Biomech., 2005, vol. 9(1), pp. 19–35.
Wagner, F.B., Mignardot, J.B., Le Goff-Mignardot, C.G., Demesmaeker, R., Komi, S., Capogrosso, M., Rowald, A., Seáñez, I., Caban, M., Pirondini, E., Vat, M., McCracken, L.A., Heimgartner, R., Fodor, I., Watrin, A., Seguin, P., Paoles, E., Van Den Keybus, K., Eberle, G., Schurch, B., Pralong, E., Becce, F., Prior, J., Buse, N., Buschman, R., Neufeld, E., Kuster, N., Carda, S., von Zitzewitz, J., Delattre, V., Denison, T., Lambert, H., Minassian, K., Bloch, J., and Courtine, G., Targeted neurotechnology restores walking in humans with spinal cord injury, Nature, 2018, vol. 563(7729), pp. 65–71.
Burkin, A.N., Features of kinematics and dynamics of the foot, requiring consideration in the development of methods and means for assessing the properties of shoe sole materials during bending, Scientific Works of BSEU, 2013, pp. 42–47.
Skvortsov, D.V., Diagnostika dvigatelnoi patologii instrumentalnymi metodami: analiz pokhodki, stabilometriya (Diagnosis of Motor Pathology by Instrumental Methods: Gait Analysis, Stabilometry), Moscow, 2007.
Malouin, F., Potvin, M., Prevost, J., Ri-chards, C.L., Wood-Dauphinee, S., Giuliani, C.A., and Duncan, P.W., Use of an intensive task-oriented gait training program in a series of patients with acute cerebrovascular accidents, Phys. Ther., 1992, vol. 72(11), pp. 781–793.
Saenko, D.G., Alkinson, D.F., Floyd, T.C., Gorodnichev, R.M., Moshonkina, T.R., Harkema, S.J., Edgerton, V.R., and Gerasimenko, Y.P., Effects of paired transcutaneous electrical stimulation delivered at single and dual sites over limbosacral spinal cord, Neurosci. Letters, 2005, (609), pp. 229–234. https://doi.org/10.1016/j.neulet.2015.10.005CAPTIONS
Funding
This study was supported by grant No. 2/2019 of 12.01.2019 from Kosima Ltd. (project the “Designing a neurostimulation device to regulate motor functions in patients with consequences of acute impairment of cerebral circulation”) and the Program “Fundamental studies for the long-term development and provision of competitiveness of the society and the State” (project No. АААА-А18-118050890115-9).
Author information
Authors and Affiliations
Contributions
I.N. Bogacheva—manuscript writing, survey of relevant publications; N.A. Shcherbakova—data analysis, survey of relevant publications; A.A. Grishin—data obtaining; Yu.P. Gerasimenko—experiment design, data obtaining.
Corresponding author
Ethics declarations
COMPLIANCE WITH ETHICAL STANDARDS
In accordance with the principles of the Declaration of Helsinki and the Russian Federation’s and international standards, written informed consent was obtained from each of the subjects to participate in the research.
CONFLICT OF INTEREST
Authors of this study have no conflict of interest.
Additional information
Russian Text © The Author(s), 2021, published in Rossiiskii Fiziologicheskii Zhurnal imeni I.M. Sechenova, 2021, Vol. 57, No. 3, pp. 374–381https://doi.org/10.31857/S0869813921030031.
Translated by A. Polyanovsky
Rights and permissions
About this article
Cite this article
Bogacheva, I.N., Shcherbakova, N.A., Grishin, A.A. et al. Effects of Phase Shifts of Transcutaneous Electrical Spinal Cord Stimulation on the Kinematic Characteristics of Stepping Movements in Humans. J Evol Biochem Phys 57, 319–324 (2021). https://doi.org/10.1134/S0022093021020137
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022093021020137