Abstract
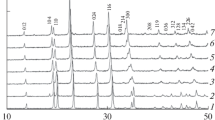
Mg0.5Zr2(AsO4)x(PO4)3 − x arsenate phosphates have been prepared by a sol–gel process and characterized by X-ray diffraction, IR spectroscopy, and impedance spectroscopy. The results indicate the formation of a continuous series of solid solutions with the Sc2(WO4)3 structure (sp. gr. P21/n) in the composition range 0 < x < 3. The unit-cell parameters of the solid solutions increase linearly with composition, as a consequence of arsenic substitution for phosphorus, which has a smaller ionic radius. The number of observed stretching and bending bands of the AsO 3-4 and PO 3-4 ions in the IR spectra of the solid solutions agrees with that predicted by factor group analysis for space group P21/n. The observed gradual shift of the absorption bands of the AsO4 tetrahedra to lower frequencies with increasing arsenic content on the tetrahedral site supports the X-ray diffraction evidence of the formation of substitutional solid solutions. The cation conductivity of Mg0.5Zr2(AsO4)x(PO4)3 − x with 0 ≤ х ≤ 1 has been shown to exceed the conductivity of the parent magnesium zirconium arsenate.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Pet’kov, V.I., Complex phosphates formed by metal cations in oxidation states I and IV, Russ. Chem. Rev., 2012, vol. 81, no. 7, pp. 606–637.
Yaroslavtsev, A.B. and Stenina, I.A., Complex phosphates with the NASICON structure (MxA2(PO4)3), Russ. J. Inorg. Chem., 2006, vol. 51, suppl. 1, pp. S97–S116.
Ivanov–Shitz, A.K. and Murin, I.V., Ionika tverdogo tela (Solid–State Ionics), St. Petersburg: S.–Peterburg. Gos. Univ., 2001, vol. 1.
Aono, H., Sugimoto, E., Sadaoka, Y., Imanaka, N., and Adachi, G., Ionic–conductivity of solid electrolytes based on lithium titanium phosphate, J. Electrochem. Soc., 1990, vol. 137, no. 4, pp. 1023–1027.
Best, A.S., Forsyth, M., and MacFarlane, D.R., Stoichiometric changes in lithium conducting materials based on Li1 + xAlxTi2 − x(PO4)3: impedance, X–ray and NMR studies, Solid State Ionics, 2000, vols. 136–137, pp. 339–344.
Adachi, G., Imanaka, N., and Aono, H., Fast Li+ conducting ceramic electrolytes, Adv. Mater., 1996, vol. 8, no. 2, pp. 127–135.
Stenina, I.A., Kislitsyn, M.N., Pinus, I.Yu., Arkhangel’skii, I.V., Zhuravlev, N.A., and Yaroslavtsev, A.B., Phase transformations and cation mobility in NASICON lithium zirconium double phosphates Li1 ± xZr2 − x–Mx(PO4)3 (M = Sc, Y, In, Nb, Ta), Russ. J. Inorg. Chem., 2005, vol. 50, no. 6, pp. 906–911.
Muldoon, J., Bucur, C.B., Oliver, A.G., Sugimoto, T., Matsui, M., Kim, H.S., Allred, G.D., Zajicek, J., and Kotani, Y., Electrolyte roadblocks to a magnesium rechargeable battery, Energy Environ. Sci., 2012, vol. 5, pp. 5941–5950.
Yaroslavtsev, A.B., Solid electrolytes: main prospects or research and development, Russ. Chem. Rev., 2016, vol. 85, no. 11, pp. 1255–1276.
Tamura, S., Yamane, M., Hoshino, Y., and Imanaka, N., Highly conducting divalent Mg2+ cation solid electrolytes with well–ordered three–dimensional network structure, J. Solid State Chem., 2016, vol. 235, no. 1, pp. 7–11.
Anuar, N.K., Adnan, S.B.R.S., and Mohamed, N.S., Characterization of Mg0.5Zr2(PO4)3 for potential use as electrolyte in solid state magnesium batteries, Ceram. Int., 2014, vol. 40, no. 8, pp. 13 719–13 727.
Anuar, N.K. and Mohamed, N.S., Structural and electrical properties of novel Mg0.9 + 0.5yZn0.4AlyZr1.6 − y(PO4)3 ceramic electrolytes synthesized via nitrate sol–gel method, J. Sol–Gel Sci. Technol., 2016, vol. 80, pp. 249–258.
Anuar, N.K., Adnan, S.B.R.S., Jaafar, M.H., and Mohamed, N.S., Studies on structural and electrical properties of Mg0.5 + y(Zr2 − yFey)2(PO4)3 ceramic electrolytes, Ionics, 2016, no. 22, pp. 1125–1133.
Halim, Z.A., Adnan, S.B.R.S., and Mohamed, N.S., Effect of sintering temperature on the structural, electrical and electrochemical properties of novel Mg0.5Si2(PO4)3 ceramic electrolytes, Ceram. Int., 2016, vol. 42, no. 3, pp. 4452–4461.
Pet’kov, V.I., Sukhanov, M.V., Shipilov, A.S., Kurazhkovskaya, V.S., Borovikova, E.Yu., Pinus, I.Yu., and Yaroslavtsev, A.B., Synthesis and properties of LiZr2(AsO4)3 and LiZr2(AsO4)x(PO4)3 − x, Inorg. Mater., 2014, vol. 50, no. 3, pp. 263–272.
Jouanneaux, A., Verbaere, A., Piffard, Y., Fitch, A.N., and Kinoshita, M., How to distinguish between monoclinic distortions of Nasicon and Sc2(WO4)3 structure types from X–ray–powder patterns—crystal–structure of Ni0.5Zr2(PO4)3, Eur. J. Solid State Inorg. Chem., 1991, vol. 28, pp. 683–699.
Pet’kov, V.I., Shipilov, A.S., Borovikova, E.Yu., Boldyrev, K.N., and Koval’skii, A.M., Synthesis and X–ray crystallographic and IR–spectroscopic study of complex alkaline earth zirconium arsenates, Russ. J. Inorg. Chem., 2017, vol. 62, no. 2, pp. 218–223.
Pet’kov, V.I., Kurazhkovskaya, V.S., Orlova, A.I., and Spiridonova, M.L., Synthesis and crystal chemical characteristics of the structure of M0.5Zr2(PO4)3 phosphates, Crystallogr. Rep., 2002, vol. 47, no. 5, pp. 736–743.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V.I. Pet’kov, A.S. Shipilov, E.Yu. Borovikova, A.M. Kovalskii, I.A. Stenina, A.B. Yaroslavtsev, 2018, published in Neorganicheskie Materialy, 2018, Vol. 54, No. 10, pp. 1079–1084.
Rights and permissions
About this article
Cite this article
Pet’kov, V.I., Shipilov, A.S., Borovikova, E.Y. et al. Synthesis, Structure, IR-Spectroscopic Characterization, and Ionic Conductivity of Mg0.5Zr2(AsO4)x(PO4)3 − x. Inorg Mater 54, 1021–1026 (2018). https://doi.org/10.1134/S0020168518100138
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0020168518100138