Abstract
The diversity of the retinal-containing proteins (rhodopsins) in nature is extremely large. Fundamental similarity of the structure and photochemical properties unites them into one family. However, there is still a debate about the origin of retinal-containing proteins: divergent or convergent evolution? In this review, based on the results of our own and literature data, a comparative analysis of the similarities and differences in the photoconversion of the rhodopsin of types I and II is carried out. The results of experimental studies of the forward and reverse photoreactions of the bacteriorhodopsin (type I) and visual rhodopsin (type II) rhodopsins in the femto- and picosecond time scale, photo-reversible reaction of the octopus rhodopsin (type II), photovoltaic reactions, as well as quantum chemical calculations of the forward photoreactions of bacteriorhodopsin and visual rhodopsin are presented. The issue of probable convergent evolution of type I and type II rhodopsins is discussed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
INTRODUCTION
Family of the retinal-containing proteins (RCPs) include three types of rhodopsin: microbial (type I), animal (type II), and recently discovered heliorhodopsins (type III) [1-7]. Despite the fact that their functions are very diverse, fundamental similarity of the structure, 7-α-helical topography of the protein part and retinal as a chromophore group, as well as of their photochemical and spectral properties is astounding. Obviously, this raises the question of their evolutionary origin. Bearing in mind conflicting opinions on their divergent [8-10] or convergent [11-15] evolution, we consider the latter more favorable. In other words, it has been suggested that there is no common ancestor for all three types of rhodopsins. And, if this is true, most likely, the pressure of external factors (Darwin’s natural selection) and physiological need resulted in such amazing similarity of such unrelated RCPs. In this regards comparison and understanding of evolution of each type of rhodopsins seem to be an independent topic that attract a lot of interest among the researchers. The ever-increasing number of publications on this topic testifies to this [3-5, 11, 16-18].
It became clear recently that the diversity of RCPs is very large. At present they have been found in all domains of life – bacteria, archaea, and eukaryotes, as well as in giant viruses. Type I rhodopsins are characteristic for bacteria, archaea, viruses, and lower eukaryotes; they have very diverse functions with main ones including light-driven energy (ion pumps) and photo-information (sensor rhodopsins, cation, and anion channels) functions [6]. Type II rhodopsins are characteristic for higher animals; in the majority of cases, they are represented by the specialized G-protein-coupled receptors, which ensure mainly photo-information functions, predominantly vision function [1, 2]. Type III rhodopsins are widely available in live organisms from the same domains of life as type I rhodopsins, and they probably perform photo-information functions [7].
Initially the term rhodopsin was referred to only proteins mediating vision. The protein discovered in 1876 by Ferenz Boll was first named a vision compound (Sehestoff), next, due to its color, “visual purple” (Sehpurpur), and later “rhodopsin” from the Greek words “rhodo” – pink and “opsis” – sight. The retinal-containing proton pump of halophilic archaeon (Halobacterium salinarum) discovered by Walther Stoeckenius and Dieter Oesterhelt almost 100 years later was named bacteriorhodopsin by analogy with the visual rhodopsin [19]. At present the name rhodopsin is applied to all members of type I, II, and III RCPs. Their common features are, first, the structure of apo-protein, opsin, with seven transmembrane α-helices, and, second, cofactor (chromophore) – retinal that absorbs light quanta. It becomes more obvious with time that the 7-α-helical protein domain of RCPs is both very conserved and, at the same time, it is characterized with very high plasticity. As to retinal, it is, as a rule, covalently bound via the protonated Schiff-base (RPSB) with the lysine amino acid residue of opsin in the seventh transmembrane α-helix (TM7), and exist as all-trans (types I and III rhodopsins) or 11-cis (type II rhodopsins) isomeric forms. It should be noted that the chromophore center is the most conserved domain of opsin. Protein environment of RPSB is crucial for both spectral tuning of the rhodopsin molecule, and for facilitating ultrafast and efficient photochemical reaction of the chromophore isomerization, which is the basis for functioning of all RCPs. Considering the role of protein environment of RPSB in the chromophore center of opsin, it is acceptable to consider the process of photoisomerization as catalyzed by the protein. Furthermore, the issue of the nature of protein–chromophore interactions (steric, electrostatic, hydrogen-bonding, and hydrophobic) is actively investigated [20].
With regards to membrane topology of the protein part of the molecule, it was established that in types I and II rhodopsins N-end is facing outside the cell and C-end – inside, while the opposite is observed in type III rhodopsins with N-end facing inside the cell and C-end – outside [4, 7, 21]. The reason and biological significance of such arrangement of type III rhodopsin is not yet clear. It is also surprizing that the “inverted” topology of the protein part of the molecule has been also observed in the olfactory G-protein-binding receptors of insects [22].
Type I rhodopsins of bacteria and archaea, including bacteriorhodopsin that performs the simplest photosynthesis, are among the most ancient proteins in the biosphere, they emerged in prokaryotic cells around 3.8 billion years ago. Type I rhodopsins of single-cell eukaryotes emerged 3.2 billion years ago, while type II rhodopsins of higher animals, including visual rhodopsin, appeared in multi-cellular eukaryotes less than 1 billion years ago [5, 11, 18, 23-25].
It has been suggested in the framework of the theory of convergent evolution of rhodopsins that the lysosomal cysteine transporters with 7-α-helical structure were predecessors of the microbial rhodopsins [15]. The cAMP-dependent G-protein-coupled receptors with classic 7-α-helical structure, i.e., not containing retinal receptors, are considered as predecessors of animal rhodopsins [12-14], and retinal chromophore was inserted in the chromophore centers of opsins later.
It has been suggested in one of the studies in the framework of the theory of divergent evolution that, first, type II rhodopsins originated from the cAMP receptors (same as other G-protein-coupled receptors of class A), from which later type I rhodopsins were generated via horizontal gene transfer from eukaryotes to prokaryotes [9]. In another work, based on optimization of electron properties of the chromophore without considering clear differences in amino acid sequences, the authors suggest existence of the divergent pathway of evolution, but in this case from type I rhodopsins to type II rhodopsins [8]. In one other study the authors suggested their common origin based on comparison of the structures of Na-binding centers in the conserved amino acid region of the TM6 and on the functionally significant tilt of this helix in the microbial rhodopsins and in the G-protein-coupled receptors [10].
With regards to the recently discovered type III rhodopsins, their origin remains obscure. According to the suggestion of the authors of one study, heliorhodopsins originated from type II eukaryotic rhodopsins, which were next captured by the giant viruses and were transferred to prokaryotic cells [16]. This implies, similar to one of the hypotheses of the origin of type I rhodopsins [9], unusual direction of evolution from eukaryotes to prokaryotes.
In any case, the whole array of the data accumulated so far allows to favor the notion on convergent independent evolution of all types of RCPs. In this review similarities and differences in molecular mechanisms of photochemical reaction in type I and type II rhodopsins are considered. Based on our own experimental data and quantum-chemical calculations we present our arguments in favor of the convergent evolution of RCPs.
COMPARISON OF THE PRIMARY PHOTOREACTIONS IN TYPE I AND TYPE II RHODOPSINS
Rhodopsin functioning is based on the photochemical reaction of isomerization of the RPSB chromophore group (all-trans → 13-cis in type I rhodopsins and 11-cis → all-trans in type II rhodopsins) (Fig. 1a) [1, 2, 26-30]. Photoreaction occurs in the excited state and is characterized with unique parameters, which are determined by both chemical properties of the chromophore itself and effect of protein environment on the chromophore [26, 31-33]. In the process, the energy of light quantum is stored as the energy of the highly twisted configuration of the isomerized RPSB in the chromophore center [34]. Moreover, in the case of type I rhodopsins additional contribution to the energy storage is provided by the change of the hydrogen bond network in the RPSB region [27]. These processes occur on a femto- and early-picosecond time scale [27]. Next, transformation of the strained chromophore conformation into a relaxed state occurs accompanied by the release of stored energy, which, in turn, leads to re-arrangement of the protein environment in the chromophore center. This process eventually initiates global structural changes of the entire protein part of the molecule required for its functioning.
Physicochemical characteristics of the chromophore group in type I and type II rhodopsins. a) Chemical structure of the rhodopsin chromophore group – all-trans RPSB in type I rhodopsins (1) and 6-s-cis–11-cis RPSB in type II rhodopsins (2). Reactive bond is shown in bold. b) Stationary absorption spectra of the suspension of purple membranes containing bacteriorhodopsin of the Halobacterium salinarum archaeon (BR) (1) and of the detergent extract of the visual rhodopsin of Bos taurus (Rh) (2) normalized to α-band absorption.
Mechanisms of photoinduced reactions of type I and type II rhodopsins are presented in this section of the review using the most studied representatives of these two classes of RCPs, proton pump – bacteriorhodopsin of the H. salinarum archaeon (BR) and G-protein coupled receptor – bovine visual rhodopsin of Bos taurus (Rh), as examples.
Stationary absorption spectra of BR and Rh are shown in Fig. 1b, which consist of α-, β-, and γ-bands with the latter defined by the opsin absorption at 280 nm. α- and β-bands are associated with the absorption of RPSB as a part of the chromophore center. Depending on the isomeric form, protonation state of the Schiff base, and on peculiarities of the chromophore center structure the spectral, photochemical, and a number of other functionally important properties of the molecule change significantly [35]. Position of the α-band absorption determines spectral range of rhodopsin functioning (300-700 nm) [2]. In the case of BR, maximum of the α-band absorption is at 568 nm [Fig. 1b (1)], and in the case of Rh – at 498 nm [Fig. 1b (2)]. Position of the β-ionone ring significantly affects position of the RPSB absorption maximum [36]. In BR the chromophore group has planar trans-configuration of the C5 = C6 and C7 = C8 bonds relative to the C6–C7 single bond [6-s-trans; Fig. 1a (1)], while in Rh it has cis-configuration [6-s-cis; Fig. 1a (2)], which is not planar due to steric hindrance causing decrease of the conjugation length in the chromophore and shift of the Rh absorption maximum towards shorter wavelengths.
Forward photoreaction of type I and type II rhodopsins. Absorption of a quantum of light results in photochemical reaction of RPSB isomerization and formation of the primary photoproducts with spectral properties different from the initial state. In this review our own results on the dynamics of the primary processes of phototransformation of BR and Rh obtained using method of femtosecond absorption laser spectroscopy [37-46] and supplemented with the data of quantum-chemical calculations are presented [32, 47-50].
Difference spectra and kinetic curves of the photoinduced absorption of BR and Rh presented in Fig. 2 are produced with probing pulse delay time of up to 10 ps. The first photoinduced signals observed in the early femtosecond time scale include absorption and stimulated emission from the S1 excited state [Fig. 2a (2 and 3); Fig. 2b (2)]. These signals are followed by absorption of the first photoproduct in the ground electronic state S0, and bleaching appears in the region of absorption of rhodopsin in the ground state [Fig. 2a (5), 600-700 nm; Fig. 2b (4), 540-700 nm]. Within several picoseconds the absorption band of the first photoproduct shifts slightly towards shorter wavelengths, which reflects formation of the next product as a result of the processes of vibrational relaxation of the chromophore group and its amino acid environment [Fig. 2a (6); Fig. 2b (6)]. A fraction of the excited BR and Rh molecules returns back into the initial state with non-isomerized RPSB, which determined quantum yield of the reaction.
Forward photoreactions of BR and Rh. a) Spectra of photoinduced absorption of BR recorded with delay times: –0.2 (1), 0.05 (2), 0.12 (3), 0.5 (4), 1 (5), and 10 (6) ps. b) Spectra of photoinduced absorption of Rh recorded with delay times: –0.2 (1), 0.03 (2), 0.1 (3), 0.2 (4), 0.8 (5), and 10 (6) ps. Stationary absorption spectra of BR (a) and Rh (b) are presented with negative sign (7). c, d) Normalized kinetic curves of photoinduced absorption of BR (1) and Rh (2) recorded at the absorption band of the excited state (c) with probing wavelengths of 470 (BR) and 410 (Rh) nm and in the absorption band of the photoreaction products (d) with probing wavelengths of 640 (BR) and 580 (Rh) nm. Kinetic curves are shown in the linear scale of delay time up to 2 ps and further in the logarithmic scale. The figure is adapted from [44] with permission.
Comparative analysis of the photoinduced absorption spectra of BR and Rh demonstrates differences in the position of absorption bands and stimulated emission, as well as in the time of formation of the primary products. In the case of BR, the signals of the excited state (I460) appear after delay time of 100 fs (Fig. 2a (2 and 3); 400-540 nm and 700-880 nm) [42-44]. Within 1 ps these signals practically completely disappear and are replaced with the positive absorption signal of the first product (J625) [Fig. 2a (5)], which contains RPSB in 13-cis-configuration. The next product (K590) is formed within a picosecond time interval [Fig. 2a (6)].
In the case of Rh, the signals from the excited state (Rh*510) appear within <30 fs (Fig. 2b (2); 410-480 nm and 620-720 nm) [41, 42], which is significantly faster than in the case of BR. At the delay time of 100 fs these signals already disappear and are replaced with the positive signal of the first product (Photo570), which is completely formed by the time of 200 fs after absorption of the light quantum [Fig. 2b (3 and 4)] [37, 38, 41, 42]. The next product (Batho535) is formed within several picoseconds [Fig. 2b (5 and 6)].
Kinetic curves of the photoinduced absorption of BR and Rh are presented in Fig. 2, which reflect formation and decay of the excited state (I460 and Rh*510, respectively; Fig. 2c) and formation of the first product of photoreaction (J625 and Photo570, respectively; Fig. 2d). In the case of Rh, photoreaction proceeds much faster (60 fs) in comparison with BR (480 fs) [40, 42-44], which is in good agreement with the data reported by Kochendoerfer and Mathies [51], Polli et al. [52], and Johnson et al. [53, 54]. Time of formation of the second product of the BR reaction (K590) is estimated as 1.8 ps, and of the second product of the Rh reaction (Batho535) – as 2.2 ps [44]. Analysis of dynamics of the excited state decay (both of Rh and BR) reveals that in a small fraction of the molecules (~4%) lifetime of the excited state is much longer (2.4 ps), and its decay results in formation of only initial state of the RCP, hence, this pathway has been termed non-reactive [44]. Existence of several pathways of the excited state decay, some of which could be non-reactive, is, in general, typical for type I rhodopsins [55-58], and in a lesser degree – for type II rhodopsins [59]. This has been associated with the split of the reaction pathway in the Frank–Condon state (FC) or with the initial heterogeneity of the protein part of the molecule [28, 60]. The latter suggestion has been confirmed for the sodium pump KR2 of the Krokinobacter eikastus bacterium [49, 57] and for the proton pumps proteorhodopsin and BR [58]. The ratio of the fraction of the excited molecules undergoing isomerization and the fraction of the excited molecules returning to the initial state via the reactive and non-reactive pathways determines quantum yield of isomerization, which is 0.64 for BR [61] and 0.65 for Rh [62]. Kinetic scheme and lifetimes of the observed processes are presented in Fig. 3, a and b.
Kinetic schemes of elementary reaction of photoisomerization in type I rhodopsin – BR (a) and in type II rhodopsin – Rh (b). Lifetimes of the intermediate states are presented in the schemes as well as reactive (r) and non-reactive (nr) decay pathways of the excited state are marked. Potential energy surfaces (S0 and S1) of type I rhodopsins exemplified by BR (c) and type II rhodopsins exemplified by Rh (d) show the reaction pathways of their forward (dark gray color) and reverse (gray color) photoreactions in femto- and pico-second time scale. FC, Franck–Condon state; CI, conical intersection. Figure is adapted from [42] with permission.
One of distinctive features of the photoreaction of RPSB in rhodopsins and in solution is existence of the vibrational component in its dynamics in the early stages [29, 42, 53, 63, 64]. Phases and amplitudes of these vibrations allowed to interpret their appearance as a result of non-stationary oscillatory motion in the excited and ground states of the reagent and reaction products. Contrary to the classic notions, formation of the product in this type of photochemical reactions occurs via coordinated motion of the molecule nuclei and ends faster than the processes of vibrational relaxation and vibrational dephasing in the excited state [65, 66]. Such reactions are termed coherent and could be described as a motion of the wave packet (set of coherent excited vibrational states) first down the S1 potential energy surface (PES) and next down the S0 PES along the reaction coordinate. Such fast S1 → S0 transition is possible due to existence of the multidimensional conical intersection region (CI) of S1/S0 PESs, owing to which effective and ultrafast transformation of light energy into chemical energy occurs [31, 67].
The main differences in the dynamics of forward photoreactions of BR and Rh are associated with the structures of PESs of the S0, S1, and S2 states of these rhodopsins (Fig. 3, c and d), which is determined by both isomeric form of RPSB, and by the effect of specific protein environment of the chromophore.
In the case of BR, similar to the other type I rhodopsin, the forward photoreaction is described by the 3-state model including S0, S1, and S2 states [26, 28, 55, 68], which postulates existence of a small barrier on the decay pathway of the S1 state due to interaction with the S2 PES, which significantly affects dynamics of the reaction (Fig. 3c). Excitation of the BR results in formation of the FC state on the S1 PES, which, as a result of the wave packet motion along all-symmetrical (C=C and C–C) vibrational modes of RPSB, is transferred into the excited state I460 within 40 fs. Next, the wave packet moves along the reactive hydrogen out-of-plane (HOOP) and torsional vibrational modes and by overcoming a small barrier on the S1 PES reaches the CI region within the characteristic time of 480 fs. In the CI region the wave packed is divided into two subpackets, with one of them is transferred to the S0 PES of the J625 product, and the second one – to the S0 PES of the initial state BR568. As a result of overcoming the barrier on the S1 PES the wave packet loses its coherent properties, which is obvious from the absence of clearly pronounced oscillations in the time-resolved signals of the product J625 absorption [Fig. 2d (1)] [29, 42-44, 55, 56]. This type of dynamics is termed diffusive [26].
In the case of Rh, same as for other type II rhodopsins, forward photoreaction is described by the 2-state model (S0 and S1) (Fig. 3d) [2, 26, 28, 69]. As a result of excitation of the Rh molecule a FC state is formed on the S1 PES, which is transformed within less than 30 fs into the excited state Rh*510 as a result of motion of the wave packet along the all-symmetric (C=C and C–C) vibrational modes of RPSB, similarly to the case of BR. Next, the wave packet moves along the out-of-plane (HOOP and torsional) vibrational modes, reaches the CI region within ≈60 fs, where it is divided into two subpackets. The first subpacket moves along the S0 PES of Photo570, which is accompanied by the vibrational relaxation of this product visualized by the clearly pronounced oscillations of these kinetic curves (Fig. 2d), while the second subpacket is transferred to the S0 PES of the initial state Rh498 continuing its motion. Hence, the S1 → S0 transition of the wave packet in the process of photoreaction of Rh occurs very fast, without deviations and barriers thus preserving the significant portion of coherence [37, 38, 42, 63]. Such type of dynamics is termed ballistic or impulsive [26, 70].
In order to explain differences in the dynamics of photoinduced processes and specificity of the reaction of photoisomerization in BR and Rh, quantum-chemical calculations of the structures of these RCPs were performed, and activity of the vibrational modes during S0 → S1 excitation and their link with the reactive modes during non-radiative transition S1 → S0 were analyzed.
Optimized structures of the all-trans and 11-cis RPSB in the ground electronic state in the protein environment of BR and Rh, respectively, obtained with the help of combined method of quantum and molecular mechanics (QM/MM) are presented in Fig. 4 [48].
In the protein environment the chromophores are found to have reactive bonds significantly twisted (Fig. 4, a and c). On the other hand, these twists are absent outside of the protein and the π-conjugated system is planar with exception of the double bond in the β-ionone ring. This bond, depending on the RPSB configuration, is either in the conjugation plane of all other five double bonds [6-s-trans; Fig. 1a (1)], or is out of the conjugation plane [6-s-cis; Fig. 1a (2)]. It should be noted that in the protein environment the out-of-conjugation-plane angle of the double bond in the β-ionone ring in BR practically does not change, while in Rh it increases significantly [48].
Hence, the protein environment indeed promotes change of conformation and ‘twisting’ of the chromophore group, which is due to its interaction with the closest amino acid residues of the chromophore center. This is mainly mediated by formation of hydrogen bond between the protonated Schiff base and primary counterion, which affects significantly torsion angle of the reactive bond. Furthermore, attention should be also paid to steric interaction in the area of β-ionone ring of retinal. As expected, geometry of the primary photoproducts was found to be even more twisted around the reactive bonds due to their inability to relax in the protein environment, which, on picosecond time scale, remains more optimal for the initial isomers of RPSB (Fig. 4, b and d).
Interestingly enough, rotation barrier around the single bond C6–C7 does not exceed 3 kcal/mol in the gas phase [50], which facilitates practically free transition between the 6-s-trans and 6-s-cis conformations in the RPSB. As was mentioned above, length of the π-conjugation chain is an important factor affecting position of the absorption maximum wavelength. Intramolecular rotation around the single C6–C7 bond results in the abnormally wide absorption spectrum of the RPSB isomers in the gas phase [71], while protein environment could easily affect RPSB conformation in the region of β-ionone ring, and, depending on the value of dihedral angle of the C6–C7 bond, could control the length of π-conjugation in the chromophore thus adjusting its absorption to the certain range. Such mechanism of regulation of photophysical properties of RPSB could be a basis for spectral tuning in type II rhodopsins. Noteworthy, position of the β-ionone ring in BR does not change neither in comparison with the gas phase, nor during transition into the primary photoproducts, while its conformation changes significantly in Rh. Hence, conformation of the β-ionone ring is less significant in type I rhodopsins in comparison with type II rhodopsins.
Effects of twisting of the reactive bonds in different RPSB isomers on the dynamics of isomerization in the protein environment could be also illustrated by comparing it with the dynamics of photoresponse in the gas phase. As was mentioned above, the RPSB isomers in the isolated state have planar structure of the main part of the π-conjugated system. It was shown with the help of femtosecond spectroscopy and quantum-chemical calculations that the relaxation dynamics of the electronic excited state S1 of the 11-cis RPSB occurs within the sub-picosecond timeframe (400 fs) [32], which is comparable with the ultrafast times of photoisomerization of the chromophore in Rh (50-100 fs) [40, 42, 51-54]. At the same time, specificity of the reaction and average lifetimes of the excited state of the all-trans RPSB differ significantly in the isolated state (3 ps) [32] and in the protein environment in BR (≈500 fs) [29, 55, 56, 58]. It is important to mention that photoisomerization of the planar all-trans-isomer of RPSB occurs slowly in the gas phase, but selectively. The lowest barrier of isomerization in the S1 state has been observed in the case of the C11=C12 bond, but not in the case of the C13=C14 bond as in BR.
Hence, the effect of protein environment is a key factor in the case of type I rhodopsins. While in the case of type II rhodopsins, more advanced (from the point of view of photochemical properties) 11-cis-isomer of RPSB is used. Moreover, the reaction in Rh proceeds ultrafast, which also implies significance of the effect of protein environment of the chromophore center. This difference explains the clearly visible coherent mode of the primary photochemical reaction in type II rhodopsins.
The effect of conformation of the reactive bonds in RPSB on the dynamics of photoisomerization has been independently confirmed during investigation of the chemically modified all-trans-chromophore twisted around one of the double bonds. Change of the RPSB conformation at the reactive bond results in the significant reduction of the photoisomerization barrier and sub-picosecond times, which is typical for the reaction in the protein environment [47].
Twisting of the reactive bond in the electronic state S0 not only changes the PES topography in the state S1 by decreasing the barrier of photoisomerization, but also facilitates simultaneous excitation of certain vibrational modes on transition S0 → S1 [48, 49]. In the process, excitation of both valent vibrations, localized predominantly on the reactive double bond, and of HOOP-vibrations at this bond occur. BR protein environment in this case facilitates excitation of vibrations of the C13=C14 bond, while in the case of Rh, protein environment facilitates excitation of the C11=C12 bond. Activity of the vibrational modes during photoexcitation could be estimated from the shift of minima of the PES of the S0 and S1 states along these modes. The shifts characterize changes of geometry of the chromophore in the S1-state in comparison with the S0 state. The larger is the shift along certain vibration mode the more active is vibration upon photoexcitation. The so-called Huang–Rhys factor associated with the square of the shift along the normal mode in a harmonic approximation is a useful dimensionless parameter allowing evaluation of the activity of the vibrational mode in electronic transition. Values of the Huang–Rhys factor for the all-trans and 11-cis RPSB in the protein environment and in the gas phase, calculated with the help of QM/MM method using multiconfigurational quasidegenerate perturbation theory of the second order, are presented in Fig. 5 [48, 49].
Two high-frequency valent vibrations of the bonds C11=C12 and C13=C14 are active in the planar all-trans and 11-cis RPSB upon excitation to the S1 state in the gas phase. In the case of BR, activity of the vibrational mode localized at the C13=C14 bond becomes significantly higher in comparison with the gas phase, while the valent vibration of the C11=C12 bond disappears entirely. On the contrary, in the case of Rh, intensity of the mode associated with vibrations of the C11=C12 bond increases significantly, while intensity of the C13=C14 mode decreases. Moreover, HOOP at the C11=C12 bond in Rh also become active.
Hence, increase of twisting of the reactive bond promotes increase of activity of the corresponding valent vibration during photoexcitation. It should be mentioned that specificity of the photo-response of RPSB and activity of the vibrations facilitating photoisomerization at the certain double bond (in this case valent and HOOP vibrations) are more characteristic exactly for Rh. This is in agreement with very high rate of the photochemical reaction in this type II rhodopsin. The possibility of excitation of various type of reactive modes in the same phase, which promotes coherency of the reaction in Rh, is also an important factor.
Hence, the results of quantum-chemical calculations provided explanation to the experimentally observed difference in the dynamics of the photoinduced processes in BR and Rh. The main factor affecting these processes in both cases is protein environment of the RPSB. Photoresponse of the chromophore group in type I and type II rhodopsins becomes highly specific in comparison with the gas phase already at the early times of the reaction resulting in excitation of certain vibrational modes, which facilitate isomerization of the reactive bond. The 11-cis RPSB chromophore in Rh is faster and more selective in comparison with the all-trans RPSB in BR. Moreover, protein environment in Rh facilitates excitation of not only valent vibrations of the reactive bond, but also of the out-of-plane vibrations of hydrogen atoms at this bond, which directly leads to the reaction of photoisomerization. It must be also mentioned that conformation of the β-ionone ring in the 6-s-cis-11-cis RPSB isomer, most likely, is an important factor in regulation of photophysical properties of the chromophore group in type II rhodopsins, which is associated with the mechanism of spectral tuning.
It could be concluded that, most likely, the evolutionary “younger” type II rhodopsins have more advanced chromophore center in the opsin and isomeric form of the chromophore, which allow more efficient forward photochemical reaction.
Photochromism of type I and type II rhodopsins. Rhodopsins exhibit photochromic properties [30, 34, 61, 72], i.e., have ability to realize phototransitions from the intermediate products of the forward photoreaction back to the initial state. This ability realized in nature in a number of rhodopsins starting with the nanosecond time scale serves for performing certain physiological functions in such type I rhodopsins as sensory rhodopsin I of H. salinarum [73], sensory rhodopsin of cyanobacteria Anabaena sp. [29], and channel rhodopsin of the single-cell alga Chlamydomonas reinhardtii [74], and in such type II rhodopsins as visual rhodopsins of invertebrates [75-77] and closely related to them melanopsins of vertebrates [78].
Despite the fact that at first stages of rhodopsin phototransformation reverse phototransitions are practically not existing in nature, investigation of such ultrafast photochromism under in vitro conditions could provide additional information on molecular mechanisms of the photochemical reaction of these RCPs. In this review the results of our studies investigating reverse photoreactions of BR and Rh initiated in femto- and early picosecond timeframes are presented [39, 41, 42].
Spectrum of photoinduced absorption of BR recorded with the delay time of 100 ps after the first pump pulse with wavelength of 560 nm (pulse 1) that consists of an absorption band of the product K590 and bleaching band of the initial state BR568 is presented in Fig. 6 [Fig. 6a (1)]. The second pump pulse with wavelength of 680 nm (pulse 2) and delay time of 5 ps excites K590 molecules, part of which returns to BR568 as a result of reverse photoreaction (13-cis → all-trans RPSB). In the process, dark formation of K590 decreases and absorption of BR568 increases [Fig. 6a (2)]. Quantum yield of the reverse reaction of BR568 formation from the product K590 is 0.81 [16], which is in agreement with the results reported by Kim et al. [61] and Balashov et al. [79]. Reverse phototransition could occur not only from the product K590 [Fig. 6a (inset: 3 and 5 ps)], but also from the product J625 [Fig. 6a (inset: 1 ps)] with approximately equal efficiency [42].
Reverse photoreactions of BR and Rh. a, b) Spectra of photoinduced absorption of BR (a) and Rh (b) recorded with delay time of 100 ps after the action of one (1) and two (2) pump pulses with delay of the second pump pulse of 5 ps (a) and 200 fs (b). In the spectral regions of pump pulses experimental curves were completed using modeling (dashed lines). Inset: values of photoinduced absorption of BR (a) and Rh (b) recorded at probing wavelengths of 635 nm (a) and 560 nm (b) before and after the second pump pulse depending on its delay time. c, d) Kinetic curves of photoinduced absorption of BR (c) and Rh (d) recorded after the first pump pulse at probing wavelengths of 680 nm (c) and 620 nm (d). Figure is adapted from [41, 42] with permission.
In the case of Rh action of the pulse 2 (620 nm) that follows with delay time after pulse 1 (500 nm) of 0.2-3.8 ps also induces reverse photoreaction [Fig. 6b] [39, 41, 42, 46]. Depending on the pulse 2 delay the reverse phototransition occurs (all-trans → 11-cis RPSB) from the Photo570 product [Fig. 6b (inset: 0.2-2 ps)] or from the Batho535 product [Fig. 6b (inset: 3-3.8 ps)]. Efficiency of the reverse photoreaction of Rh from the Photo570 product depends of the oscillation phase of the time-resolved absorption signals of this product [Fig. 6b (inset); Fig. 6d], and time of this photoreaction is comparable or less than the time of forward photoreaction [41]. Quantum yield of the reverse photoreaction of Rh from the Batho535 product is 0.15 [41, 42, 46], which is significantly less than the quantum yield value (0.5) at 77 K reported previously by Suzuki and Callender [80].
The reverse photochemical process in BR and Rh initiated at early stages could be described as a motion of the wave packet along the right branch of the S1 PES of the product of initial photochemical reaction towards the CI region of the S1/S0 PES (Fig. 3, c and d). Theoretical calculations for Rh demonstrated that this is the same CI region, which participates in the forward photoreaction [81]. Transition of the molecules from the excited state S1 to the S0 PES via the CI region results in formation of the same states as in the forward reaction: BR568 and J625 – in the case of BR; Rh498 and Photo570 – in the case of Rh. While efficiency of the reverse photoreaction of BR practically does not depend on the delay of the pulse 2 [Fig. 6a (inset)], for Rh it correlates directly with the dynamics of the wave packet in the Photo570 product [Fig. 6b (inset); Fig. 6d], which is associated with clearly pronounced coherence of the forward photoreaction of Rh. In this case, efficiency of the reverse photoreaction correlates with the number of molecules of the Photo570 product excited by the pulse 2.
The rate of reverse photochemical reaction in rhodopsins is strongly affected by the particular isomeric form of RPSB. Phototransition from trans- to cis-form usually is slower than phototransition cis → trans, and this is true for protein environment, gas phase, as well as solution [26, 29, 32, 52, 57, 58, 82]. This difference is due to the structures of S0, S1, and S2 PES, as described above using BR and Rh as examples [Fig. 3, c and d]. It was shown by Gai et al. [56] that, in the case of BR, during excitation of the K590 product the reverse photochemical reaction from 13-cis- to all-trans-form of RPSB occurs within the time period of ~100 fs, which is significantly faster that the time of forward photoreaction (480 fs). It can be suggested that the reverse dependence would be observed for Rh. However, as has been shown experimentally for the Photo570 product [41] and theoretically for the Batho535 product [81], time of the reverse phototransition from all-trans- to 11-cis-form RPSB is comparable with the time of forward phototransition (60 fs) or less. It could be suggested that the rate (and selectivity) of the reverse photochemical reaction of BR and Rh is affected by the significant twist of the reactive bond in the chromophore in their primary products by the angle from –39° to –20° and from –36° to –34.8°, respectively [31, 83]. This allows fast and barrier-less transition to the CI region even in the case of trans → cis isomerization as in the reverse photoreaction of Rh.
It must be emphasized that while quantum yields of the forward photoreaction in BR and Rh are practically the same (≈0.65), quantum yields of their reverse photoreactions differ significantly (0.81 and 0.15, respectively). According to the theoretical calculations [31, 81], quantum yield of the product formation both in the forward and reverse photoreactions of rhodopsins is determined by the ratio of phases of HOOP and torsional vibration modes of RPSB at the moment of the S1 → S0 transition. Initial population of the C=C and HOOP vibrational modes at the reactive bond on excitation also plays an important role in efficiency of the photoreaction, as was mentioned above. In methanol solution the 13-cis → all-trans and all-trans → 11-cis RPSB photoreactions proceed with very close quantum yields (0.11 and 0.14, respectively) [84]. Based on this, it could be suggested that the significant difference in the quantum yields of the analogous reverse reactions in BR and Rh is due to the effects of protein environment at the chromophore center.
It could be mentioned that, from the functional point of view, the low quantum yield of the reverse reaction in Rh in comparison with BR is an advantage facilitating more reliable realization of the forward reaction initiating the process of phototransduction. Light absorption by type I rhodopsins serves primarily the energy conversion function, as in the case of BR, and reverse photoreactions likely cannot significantly affect their efficiency. However, in the case of type II rhodopsins, which are G-protein-coupled receptors, 11-cis RPSB acts as an effective ligand antagonist maintaining low thermal “dark” noise of the photoreceptor cell. On absorption of light, the isomerized all-trans RPSB becomes a powerful ligand-agonist, and the activated Rh initiates the process of phototransduction. In this case the possibility of emergence of the reverse photoreaction could significantly reduce efficiency of initiation of the phototransduction process.
Hence, lower efficiency of the reverse photoreaction of Rh, which increases reliability of the forward photoreaction, could be considered as one of the arguments in favor of selection in the course of evolution of the 11-cis-isomer as a chromophore group in type II rhodopsins. This indicates more advanced structure of the chromophore center in Rh and evolutionary selection of such chromophore (11-cis RPSB), which allows efficient phototransduction process. Obviously, under real physiological conditions, the probability of the second light quantum hitting the so short-lived intermediate of Rh photolysis as Batho535 and more so Photo570 is extremely low, however, potential possibility of reducing the probability of reverse photoreaction in the structure of Rh chromophore center exists.
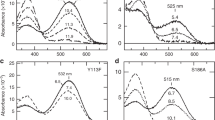
At the same time, physiological significance of the reverse photochemical reactions of type II rhodopsins in invertebrate animals from the late intermediate stages of phototransformation is obvious. In the majority of invertebrate animals this is one of the main ways for regeneration (photoregeneration) of the visual pigment [75, 77]. In particular, final product of the forward photoreaction of the octopus rhodopsin is the so-called acidic metarhodopsin, and of the reverse reaction – again rhodopsin (Fig. 7a) [76].
Photochromic reaction of octopus rhodopsin. a) Difference spectra of two consecutive cycles of octopus rhodopsin transformations demonstrating reversible conversion of rhodopsin into acidic metarhodopsin: metarhodopsin minus rhodopsin (1 and 3, after illumination with blue light) and photoregeneration of rhodopsin: rhodopsin minus metarhodopsin (2 and 4, after illumination with red light). b) Photoreversibility of octopus rhodopsin recorded at wavelength of maximum absorption of acidic metarhodopsin (528 nm) under illumination with blue light (1), red light (2), and in the dark (3). Figure is adapted from [76] with permission.
In the course of forward photoreaction, several intermediates with different absorption spectra are formed, and in the course of reverse photoreaction – there are only two states corresponding to conformational changes first in the vicinity of chromophore, and, next, in the entire protein, which return rhodopsin into initial state. As we have demonstrated with the detergent extracts of the octopus rhodopsin, the photoreversible reaction of type II rhodopsin in invertebrates is extremely stable, and under in vitro conditions it can be repeated multiple times for a significantly long period of time (Fig. 7b) [76].
Comparison of mechanisms of phototransformation of type I and type II rhodopsins. Comparison of the primary photoreactions of BR and Rh reveals both common and distinguishing features. Model kinetic schemes (Fig. 3, a and b) have much in common, and reflect the suggestion that heterogeneity of the excited states of the investigated rhodopsins is associated with heterogeneity of their initial states. In the course of BR and Rh reactions intermediates are formed that have similar properties: I460, J625, K590, and Rh*510, Photo570, Batho535, respectively. Elementary act of isomerization of RPSB proceeds via transition through the CI of the S1/S0 PES, and time of this transition is in femtosecond time scale. The first products of photoinduced transformations of BR and Rh with isomerized RPSB (J625 and Photo570, respectively) are formed in the ground (S0) state, and are vibrationally excited precursors of the following products (K590 and Batho535, respectively) formed in the timeframe of several picoseconds. At this stage isomerization of RPSB is completed, and part of the light quantum energy is preserved in the strained structures of the products K590 and Batho535. Quantum yields of the forward photoreaction of BR and Rh are practically the same (~0.65). Reaction of photoisomerization of RPSB in BR and Rh, same as in other rhodopsins, as well as in solution, is coherent due to the inherent properties of the chromophore.
Dynamics and efficiency of the RPSB photoreaction in rhodopsins depends on its isomeric form (through the length of conjugated π-bonds) and on interactions with the protein environment, which results not only in the additional twisting of the chromophore, but also in the strong electrostatic interactions with the counterion. These factors affect the structure of the S0, S1, and S2 PES of the chromophore, by, for example, decreasing barrier in the S1 PES of type I rhodopsins, which significantly affects rate and coherence of the reaction. Activity of certain vibrational modes also changes during electronic transition S0 → S1. Selective and simultaneous excitation of the vibrational modes associated with the reactive modes of the non-radiative transition S1 → S0 into the reaction products leads at the early stages of photoinduced dynamics to high speed and high quantum yield of the reaction of photoisomerization.
It can be concluded that interaction of RPSB with opsin significantly increases the rate and quantum yield of the photoreaction in comparison with the parameters observed in gas phase and in solution, and selectivity of the reaction reaches 100%. The finest regulation of these parameter is achieved in type II rhodopsins, in which the reaction has more pronounced coherence. The chromophore–protein interactions also could be the cause of heterogeneity of the initial state of RCPs, because it is not observed in the gas phase [32].
Decrease of the length of π-conjugation in the 6-s-cis–11-cis RPSB in comparison with the all-trans-isomer also leads to the shift of absorption maximum of rhodopsin towards blue region of the spectrum and, which is very important, to increase of the barrier of thermal 11-cis → trans isomerization [26]. The first effect is actively used by type II rhodopsins as one of the factors of spectral tuning. The second factor is crucial for reduction of the ‘dark’ noise of the photoreceptor cell, which is also much facilitated by the limited volume of the chromophore center [85]. At the same time, in the case of BR and some other type I rhodopsins, the chromophore center is rather spacious and does not prevent thermal trans → 13-cis isomerization occurring in the dark.
All abovementioned features of the 11-cis RPSB are extremely important for functioning of type II rhodopsins that perform photo-information function. This is, likely, the reason why the 11-cis RPSB was selected in the course of evolution as a chromophore group of visual rhodopsins in both invertebrate and vertebrate animals.
It should be emphasized that although the reaction of photoisomerization is common for both type I and type II rhodopsins, the final stages of the photoactivated processes differ significantly. In type I rhodopsins, the 13-cis RPSB is subjected at one of the final stages to thermal isomerization into the all-trans-form thus closing the photoactivated processes into a photocycle (Fig. 8a). In type II rhodopsins of the majority of invertebrates, absorption of the second light quantum by one of the long-lived intermediate products of phototransformation is required to close the cycle. The cycle is not closed in type II rhodopsins of the vertebrates, and the isomerized all-trans-retinal is released from the chromophore center of the opsin, which is called photobleaching or rhodopsin photolysis (Fig. 8b). In other words, type II rhodopsin molecule of vertebrate animals – is a single-use molecule. For further functioning – rhodopsin regeneration – a new retinal molecule in 11-cis conformation should be delivered to the chromophore center (see details in the review by Ostrovsky and Feldman [20]).
General similarities of the primary photochemical reactions of type I and type II rhodopsins and their significant differences in the following stages of phototransformation have been manifested clearly in their photoelectric reactions. In the end of 1970s and beginning of 1980s using the method developed by L. A. Drachev [86], we in collaborations with Drachev and the Skulachev group compared in detail generation of photopotential for Rh on a photoreceptor disk membrane from rods and for BR on a purple membrane of haloarchaea [87-89]. As expected, both transition all-trans → 13-cis in BR, and transition 11-cis → all-trans in Rh were accompanied by the emergence of fast photoelectric response. However, there was a significant difference: while potential generation on the purple membrane was accompanied also by the proton transfer, which is essential for the photoenergetic function of BR, in the case of Rh such transfer was absent. It is exactly the moment when physiological pathways of type I and type II rhodopsins diverge. Absence of the proton transfer across the photoreceptor disk membrane was demonstrated experimentally in our later study [90].
In the case of Rh, conformational changes in the protein part of the molecule occur simultaneously with the charge separation, which first results, at the metarhodopsin II stage, in rapid deprotonation of RPSB followed by slow release of the proton into external medium [91]. As a result of these conformational rearrangements, Rh at the stage of metarhodopsin II acquires ability to interact with G-protein (transducin) and to initiate the process of phototransduction [24, 92]. Based on its characteristic times and polarity, the rapid component of the Rh photoresponse is, in essence, the so-called early receptor potential, which is observed during regular electrophysiological separation from the retina [93].
While in the case of BR, with its photoactivated processes connected into a cycle, stable electric potential is generated on the purple membrane during all period of illumination, in the case of Rh, which is undergoing photolysis, rapid photoelectric response irreversibly drops long before the light is switched off, and there is no response to the repeated illumination [88, 89].
CONCLUSIONS
Hence, it can be concluded that the similarities of type I and type II rhodopsins are associated with the nature of their common chromophore group, retinal, which, to a large extent, determines the mechanism of photoreaction as a key stage of RCP functioning. And the observed differences in the nature of photoreactions of type I and type II rhodopsins most likely are explained by the different isomeric form of the chromophore selected in the course of evolution in accordance with their functions. It can be suggested in the framework of convergent evolution of type I and type II rhodopsins that the similar and at the same time plastic 7-α-helical protein structures of these two types evolved independently driven by the need to optimize electronic properties of their chromophore groups in the all-trans and 11-cis isomeric forms.
Similarity of the structure and mechanism of actions of such complex molecular system as retinal-containing transmembrane light-sensitive proteins is due to the pressure of natural selection with (must be emphasized) certain physicochemical limitations. The selection itself was pursuing the most efficient function of each type of rhodopsins.
It is absolutely clear that the mechanisms of evolution of all three currently known types of RCPs [microbial (type I), animal (type II), and recently discovered heliorhodopsins (type III)] require further detailed investigation.
Abbreviations
- RCP:
-
retinal-containing protein
- RPSB:
-
retinal protonated Schiff-base
- BR:
-
bacteriorhodopsin of Halobacterium salinarum archaeon
- Rh:
-
bovine visual rhodopsin of Bos taurus
- FC:
-
Franck–Condon state
- wave packet:
-
a set of coherent excited vibrational states
- CI:
-
conical intersection of potential energy surfaces
- HOOP:
-
hydrogen-out-of-plane vibrations
- PES:
-
potential energy surface
References
Spudich, J. L., Yang, C.-S., Jung, K.-H., and Spudich, E. N. (2000) Retinylidene proteins: structures and functions from archaea to humans, Annu. Rev. Cell Dev. Biol., 16, 365-392, https://doi.org/10.1146/annurev.cellbio.16.1.365.
Ernst, O. P., Lodowski, D. T., Elstner, M., Hegemann, P., Brown, L. S., and Kandori, H. (2014) Microbial and animal rhodopsins: structures, functions, and molecular mechanisms, Chem. Rev., 114, 126-163, https://doi.org/10.1021/cr4003769.
Nagata, T., and Inoue, K. (2021) Rhodopsins at a glance, J. Cell Sci., 134, jcs258989, https://doi.org/10.1242/jcs.258989.
Rozenberg, A., Inoue, K., Kandori, H., and Béjà, O. (2021) Microbial rhodopsins: The last two decades, Annu. Rev. Microbiol., 75, 427-447, https://doi.org/10.1146/annurev-micro-031721-020452.
Gordeliy, V., Kovalev, K., Bamberg, E., Rodriguez-Valera, F., Zinovev, E., Zabelskii, D., Alekseev, A., Rosselli, R., Gushchin, I., and Okhrimenko, I. (2022) Microbial rhodopsins, in Rhodopsin (Gordeliy, V., ed.) Methods Mol. Biol., 2501, 1-52, Humana, New York, NY, https://doi.org/10.1007/978-1-0716-2329-9_1.
Kandori, H. (2020) Biophysics of rhodopsins and optogenetics, Biophys. Rev., 12, 355-361, https://doi.org/10.1007/s12551-020-00645-0.
Pushkarev, A., Inoue, K., Larom, S., Flores-Uribe, J., Singh, M., Konno, M., Tomida, S., Ito, S., Nakamura, R., Tsunoda, S. P., Philosof, A., Sharon, I., Yutin, N., Koonin, E. V., Kandori, H., and Béjà, O. (2018) A distinct abundant group of microbial rhodopsins discovered using functional metagenomics, Nature, 558, 595-599, https://doi.org/10.1038/s41586-018-0225-9.
Luk, H. L., Melaccio, F., Rinaldi, S., and Olivucci, M. (2015) Molecular bases for the selection of the chromophore of animal rhodopsins, Proc. Natl. Acad. Sci. USA, 112, 15297-15302, https://doi.org/10.1073/pnas.1510262112.
Mackin, K. A., Roy, R. A., and Theobald, D. L. (2014) An empirical test of convergent evolution in rhodopsins, Mol. Biol. Evol., 31, 85-95, https://doi.org/10.1093/molbev/mst171.
Shalaeva, D. N., Galperin, M. Y., and Mulkidjanian, A. Y. (2015) Eukaryotic G protein-coupled receptors as descendants of prokaryotic sodium-translocating rhodopsins, Biol. Forward, 10, 63, https://doi.org/10.1186/s13062-015-0091-4.
Kojima, K., and Sudo, Y. (2023) Convergent evolution of animal and microbial rhodopsins, RSC Adv., 13, 5367-5381, https://doi.org/10.1039/d2ra07073a.
Krishnan, A., Almen, M. S., Fredriksson, R., and Schioth, H. B. (2012) The origin of GPCRs: identification of mammalian like rhodopsin, adhesion, glutamate and frizzled GPCRs in fungi, PLoS One, 7, e29817, https://doi.org/10.1371/journal.pone.0029817.
Feuda, R., Hamilton, S. C., McInerney, J. O., and Pisani, D. (2012) Metazoan opsin evolution reveals a simple route to animal vision, Proc. Natl. Acad. Sci. USA, 109, 18868-18872, https://doi.org/10.1073/pnas.1204609109.
Feuda, R., Menon, A. K., and Göpfert, M. C. (2022) Rethinking opsins, Mol. Biol. Evol., 39, 3, https://doi.org/10.1093/molbev/msac033.
Zhai, Y., Heijne, W. H., Smith, D. W., and Saier, M. H., Jr. (2001) Homologues of archaeal rhodopsins in plants, animals and fungi: structural and functional predications for a putative fungal chaperone protein, Biochim. Biophys. Acta, 1511, 206-223, https://doi.org/10.1016/s0005-2736(00)00389-8.
Bulzu, P.-A., Kavagutti, V. S., Andrei, A.-S., and Ghai, R. (2022) The evolutionary kaleidoscope of rhodopsins, mSystems, 7, e00405-22, https://doi.org/10.1128/msystems.00405-22.
Govorunova, E. G., Sineshchekov, O. A., and Spudich, J. L. (2022) Emerging diversity of channelrhodopsins and their structure-function relationships, Front. Cell. Neurosci., 15, 800313, https://doi.org/10.3389/fncel.2021.800313.
Ostrovsky, M. A. (2017) Rhodopsin: evolution and comparative physiology, Paleontol. J., 51, 562-572, https://doi.org/10.1134/S0031030117050069.
Oesterhelt, D., and Stoeckenius, W. (1971) Rhodopsin-like protein from the purple membrane of Halobacterium halobium, Nat. New Biol., 233, 149-152, https://doi.org/10.1038/newbio233149a0.
Ostrovsky, M. A. and Feldman, T. B. (2012) Chemistry and molecular physiology of vision: light-sensitive protein rhodopsin, Russ. Chem. Rev., 81, 1071-1090, https://doi.org/10.1070/RC2012v081n11ABEH004309.
Kovalev, K., Volkov, D., Astashkin, R., Alekseev, A., Gushchin, I., Haro-Moreno, J. M., Chizhov, I., Siletsky, S., Mamedov, M., Rogachev, A., Balandin, T., Borshchevskiy, V., Popov, A., Bourenkov, G., Bamberg, E., Rodriguez-Valera, F., Büldt, G., and Gordeliy, V. (2020) High-resolution structural insights into the heliorhodopsin family, Proc. Natl. Acad. Sci. USA, 117, 4131-4141, https://doi.org/10.1073/pnas.1915888117.
Benton, R., Sachse, S., Michnick, S. W., and Vosshall, L. B. (2006) Atypical membrane topology and heteromeric function of Drosophila odorant receptors in vivo, PLoS Biol., 4, e20, https://doi.org/10.1371/journal.pbio.0040020.
Lamb, T. D., Collin, S. P., and Pugh, E. N., Jr. (2007) Evolution of the vertebrate eye: Opsins, photoreceptors, retina and eye cup, Nat. Rev. Neurosci., 8, 960-976, https://doi.org/10.1038/nrn2283.
Hofmann, K. P., and Lamb, T. D. (2023) Rhodopsin, light-sensor of vision, Prog. Retin. Eye Res., 93, 101116, https://doi.org/10.1016/j.preteyeres.2022.101116.
Rozanov, A. Yu. (2009) Conditions of life on early earth after 4.0 billion years ago, in Problems of Origin of Life, PIN RAN, Moscow, pp. 185-201.
Gozem, S., Luk, H. L., Schapiro, I., and Olivucci, M. (2017) Theory and simulation of the ultrafast double-bond isomerization of biological chromophores, Chem. Rev., 117,13502-13565, https://doi.org/10.1021/acs.chemrev.7b00177.
Kandori, H. (2011) Protein-controlled ultrafast photoisomerization in rhodopsin and bacteriorhodopsin, in Supramolecular Photochemistry: Controlling Photochemical Processes, Chapter 14, 571-595, https://doi.org/10.1002/9781118095300.ch14.
Diller, R. (2008) Primary reactions in retinal proteins, in Ultrashort laser pulses in biology and medicine. Biological and Medical Physics, Biomedical Engineering, (Braun M., Gilch P., Zinth W., eds), Springer, Berlin. Heidelberg, Germany, Chapter 10, 243-277, https://doi.org/10.1007/978-3-540-73566-3_10.
Wand, A., Gdor, I., Zhu, J., Sheves, M., and Ruhman, S. (2013) Shedding new light on retinal protein photochemistry, Annu. Rev. Phys. Chem., 64, 437-458, https://doi.org/10.1146/annurev-physchem-040412-110148.
Hampp, N. (2000) Bacteriorhodopsin as a photochromic retinal protein for optical memories, Chem. Rev., 100, 1755-1776, https://doi.org/10.1021/cr980072x.
Schapiro, I., Melaccio, F., Laricheva, E. N., and Olivucci, M. (2011) Using the computer to understand the chemistry of conical intersections, Photochem. Photobiol. Sci., 10, 867-886, https://doi.org/10.1039/c0pp00290a.
Kiefer, H. V., Gruber, E., Langeland, J., Kusochek, P. A., Bochenkova, A. V., and Andersen, L. H. (2019) Intrinsic photoisomerization dynamics of protonated Schiff-base retinal, Nat. Commun., 10, 1210, https://doi.org/10.1038/s41467-019-09225-7.
Agathangelou, D., Roy, P. P., del Carmen Marin, M., Ferre, N., Olivucci, M., Buckup, T., Léonard, J., and Haacke, S. (2021) Sub-picosecond C=C bond photo-isomerization: evidence for the role of excited state mixing, Comptes Rendus Physique, 22, 111-138, https://doi.org/10.5802/crphys.41.
Birge, R. R., Cooper, T. M., Lawrence, A. F., Masthay, M. B., Vasilakis, C., Fan Zhang, C., and Zidovetzki, R. (1989) A spectroscopic, photocalorimetric, and theoretical investigation of the quantum efficiency of the primary event in bacteriorhodopsin, J. Am. Chem. Soc., 111, 4063-4074, https://doi.org/10.1021/ja00193a044.
Wang, W., Geiger, J. H., and Borhan, B. (2013) The photochemical determinants of color vision, BioEssays, 36, 65-74, https://doi.org/10.1002/bies.201300094.
Okada, T., Sugihara, M., Bondar, A.-N., Elstner, M., Entel, P., and Buss, V. (2004) The retinal conformation and its environment in rhodopsin in light of a new 2.2 Å crystal structure, J. Mol. Biol., 342, 571-583, https://doi.org/10.1016/j.jmb.2004.07.044.
Smitienko, O. A., Shelaev, I. V., Gostev, F. E., Feldman, T. B., Nadtochenko, V. A., Sarkisov, O. M., and Ostrovsky, M. A. (2008) Coherent processes in formation of primary products of rhodopsin photolysis, Dokl. Biochem. Biophys., 421, 194-198.
Smitienko, O. A., Mozgovaya, M. N., Shelaev, I. V., Gostev, F. E., Feldman, T. B., Nadtochenko, V. A., Sarkisov, O. M., and Ostrovsky, M. A. (2010) Femtosecond formation dynamics of primary photoproducts of visual pigment rhodopsin, Biochemistry (Moscow), 75, 25-35, https://doi.org/10.1134/s0006297910010049.
Mozgovaya, M. N., Smitienko, O. A., Shelaev, I. V., Gostev, F. E., Feldman, T. B., Nadtochenko, V. A., Sarkisov, O. M., and Ostrovsky, M. A. (2010) Photochromism of visual pigment rhodopsin on the femtosecond time scale: coherent control of retinal chromophore isomerization, Dokl. Biochem. Biophys., 435, 302-306.
Nadtochenko, V. A., Smitienko, O. A., Feldman, T. B., Mozgovaya, M. N., Shelaev, I. V., Gostev, F. E., Sarkisov, O. M., and Ostrovsky, M. A. (2012) Conical intersection participation in femtosecond dynamics of visual pigment rhodopsin chromophore cis-trans photoisomerization, Dokl. Biochem. Biophys., 446, 242-246.
Smitienko, O., Nadtochenko, V., Feldman, T., Balatskaya, M., Shelaev, I., Gostev, F., Sarkisov, O., and Ostrovsky, M. (2014) Femtosecond laser spectroscopy of the rhodopsin photochromic reaction: A concept for ultrafast optical molecular switch creation (ultrafast reversible photoreaction of rhodopsin), Molecules, 19, 18351-18366, https://doi.org/10.3390/molecules191118351.
Feldman, T. B., Smitienko, O. A., Shelaev, I. V., Gostev, F. E., Nekrasova, O. V., Dolgikh, D. A., Nadtochenko, V. A., Kirpichnikov, M. P., and Ostrovsky, M. A. (2016) Femtosecond spectroscopic study of photochromic reactions of bacteriorhodopsin and visual rhodopsin, J. Photochem. Photobiol. B, 164, 296-305, https://doi.org/10.1016/j.jphotobiol.2016.09.041.
Smitienko, O. A., Nekrasova, O. V., Kudryavtsev, A. V., Yakovleva, M. A., Shelaev, I. V., Gostev, F. E., Dolgikh, D. A., Kolchugina, I. B., Nadtochenko, V. A., Kirpichnikov, M. P., Feldman, T. B., and Ostrovsky, M. A. (2017) Femtosecond and picosecond dynamics of recombinant bacteriorhodopsin primary reactions compared to the native protein in trimeric and monomeric forms, Biochemistry (Moscow), 82, 490-500, https://doi.org/10.1134/S0006297917040113.
Smitienko, O., Feldman, T., Petrovskaya, L., Nekrasova, O., Yakovleva, M., Shelaev, I., Gostev, F., Cherepanov, D., Kolchugina, I., Dolgikh, D., Nadtochenko, V., Kirpichnikov, M., and Ostrovsky, M. (2021) Comparative femtosecond spectroscopy of primary photoreactions of Exiguobacterium sibiricum rhodopsin and Halobacterium salinarum bacteriorhodopsin, J. Phys. Chem. B, 125, 995-1008, https://doi.org/10.1021/acs.jpcb.0c07763.
Medvedeva, A. S., Smitienko, O. A., Feldman, T. B., and Ostrovsky, M. A. (2020) Comparative study of photochemistry of microbial rhodopsins (type I) and animal’s rhodopsins (Type II), Zh. Evol. Biokhim. Fiziol., 56, 519-523, https://doi.org/10.31857/S0044452920070943.
Ostrovsky, M. A., and Nadtochenko, V. A. (2021) Femtochemistry of rhodopsins, Russ. J. Phys. Chem. B, 15, 344-351, https://doi.org/10.1134/S1990793121020226.
Gruber, E., Kabylda, A. M., Nielsen, M. B., Rasmussen, A. P., Teiwes, R., Kusochek, P. A., Bochenkova, A. V., and Andersen, L. H. (2022) Light driven ultrafast bioinspired molecular motors: Steering and accelerating photoisomerization dynamics of retinal, J. Am. Chem. Soc., 144, 69-73, https://doi.org/10.1021/jacs.1c10752.
Kusochek, P. A., Logvinov, V. V., and Bochenkova, A. V. (2021) Role of the protein environment in photoisomerization of type I and type II rhodopsins: a theoretical perspective, Moscow Univ. Chem. Bull., 76, 407-416, https://doi.org/10.3103/S0027131421060110.
Kusochek, P. A., Scherbinin, A. V., and Bochenkova, A. V. (2021) Insights into the early-time excited-state dynamics of structurally inhomogeneous rhodopsin KR2, J. Phys. Chem. Lett., 12, 8664-8671, https://doi.org/10.1021/acs.jpclett.1c02312.
Toker, Y., Svendsen, A., Bochenkova, A. V., and Andersen, L. H. (2012) Probing the barrier for internal rotation of the retinal chromophore, Angew. Chem. Int. Ed. Engl., 51, 8757-8761, https://doi.org/10.1002/anie.201203746.
Kochendoerfer, G. G., and Mathies, R. A. (1996) Spontaneous emission study of the femtosecond isomerization dynamics of rhodopsin, J. Phys. Chem., 100, 14526-14532, https://doi.org/10.1021/jp960509+.
Polli, D., Altoè, P., Weingart, O., Spillane, K. M., Manzoni, C., Brida, D., Tomasello, G., Orlandi, G., Kukura, P., Mathies, R. A., Garavelli, M., and Cerullo, G. (2010) Conical intersection dynamics of the primary photoisomerization event in vision, Nature, 467, 440-443, https://doi.org/10.1038/nature09346.
Johnson, P. J. M., Halpin, A., Morizumi, T., Prokhorenko, V. I., Ernst, O. P., and Miller, R. J. D. (2015) Local vibrational coherences drive the primary photochemistry of vision, Nat. Chem., 7, 980-986, https://doi.org/10.1038/nchem.2398.
Johnson, P. J. M., Farag, M. H., Halpin, A., Morizumi, T., Prokhorenko, V. I., Knoester, J., Jansen, T. L. C., Ernst, O. P., and Miller, R. J. D. (2017) The primary photochemistry of vision occurs at the molecular speed limit, J. Phys. Chem. B, 121, 4040-4047, https://doi.org/10.1021/acs.jpcb.7b02329.
Hasson, K. C., Gai, F., and Anfinrud, P. A. (1996) The photoisomerization of retinal in bacteriorhodopsin: experimental evidence for a three-state model, Proc. Natl. Acad. Sci. USA, 93, 15124-15129, https://doi.org/10.1073/pnas.93.26.15124.
Gai, F., Hasson, K. C., McDonald, J. C., and Anfinrud, P. A. (1998) Chemical dynamics in proteins: the photoisomerization of retinal in bacteriorhodopsin, Science, 279, 1886-1891, https://doi.org/10.1126/science.279.5358.1886.
Tahara, S., Takeuchi, S., Abe-Yoshizumi, R., Inoue, K., Ohtani, H., Kandori, H., and Tahara, T. (2018) Origin of the reactive and nonreactive excited states in the primary reaction of rhodopsins: pH dependence of femtosecond absorption of light-driven sodium ion pump rhodopsin KR2, J. Phys. Chem. B, 122, 4784-4792, https://doi.org/10.1021/acs.jpcb.8b01934.
Chang, C.-F., Kuramochi, H., Singh, M., Abe-Yoshizumi, R., Tsukuda, T., Kandori, H., and Tahara, T. (2022) A unified view on varied ultrafast dynamics of the primary process in microbial rhodopsins, Angew. Chem. Int. Ed. Engl., 61, e202111930, https://doi.org/10.1002/anie.202111930.
Kandori, H., Futurani, Y., Nishimura, S., Shichida, Y., Chosrowjan, H., Shibata, Y., and Mataga, N. (2001) Excited-state dynamics of rhodopsin probed by femtosecond fluorescence spectroscopy, Chem. Phys. Lett., 334, 271-276, https://doi.org/10.1016/S0009-2614(00)01457-3.
Kochendoerfer, G. G., and Mathies, R. A. (1995) Ultrafast spectroscopy of rhodopsins – photochemistry at its best!, Isr. J. Chem., 35, 211-226, https://doi.org/10.1002/ijch.199500028.
Kim, J. E., Tauber, M. J., and Mathies, R. A. (2001) Wavelength dependent cis-trans isomerization in vision, Biochemistry, 40, 13774-13778, https://doi.org/10.1021/bi0116137.
Govindjee, R., Balashov, S. P., and Ebrey, T. G. (1990) Quantum efficiency of the photochemical cycle of bacteriorhodopsin, Biophys. J., 58, 597-608, https://doi.org/10.1016/S0006-3495(90)82403-6.
Wang, Q., Shoenlein, R. W., Peteanu, L. A., Mathies, R. A., and Shank, C. V. (1994) Vibrationally coherent photochemistry in the femtosecond primary event of vision, Science, 266, 422-424, https://doi.org/10.1126/science.7939680.
Liebel, M., Schnedermann, C., Bassolino, G., Taylor, G., Watts, A., and Kukura, P. (2014) Direct observation of the coherent nuclear response after the absorption of a photon, Phys. Rev. Lett., 112, 238301, https://doi.org/10.1103/PhysRevLett.112.238301.
Zewail, A. H. (2000) Femtochemistry: atomic-scale dynamics of the chemical bond, J. Phys. Chem. A, 104, 5660-5694, https://doi.org/10.1021/jp001460h.
Sarkisov, O. M., and Umanskii, S. Ya. (2001) Femtochemistry, Russ. Chem. Rev., 70, 449-469, https://doi.org/10.1070/RC2001v070n06ABEH000664.
Klessinger, M. (1995) Conical intersections and the mechanism of singlet photoreactions, Angew. Chem. Int. Ed. Engl., 34, 549-551, https://doi.org/10.1002/anie.199505491.
Gozem, S., Johnson, P. J. M., Halpin, A., Luk, H. L., Morizumi, T., Prokhorenko, V. I., Ernst, O. P., Olivucci, M., and Miller, R. J. D. (2020) Excited-state vibronic dynamics of bacteriorhodopsin from two-dimensional electronic photon echo spectroscopy and multiconfigurational quantum chemistry, J. Phys. Chem. Lett., 11, 3889-3896, https://doi.org/10.1021/acs.jpclett.0c01063.
Rivalta, I., Nenov, A., Weingart, O., Cerullo, G., Garavelli, M., and Mukamel, S. (2014) Modelling time-resolved two-dimensional electronic spectroscopy of the primary photoisomerization event in rhodopsin, J. Phys. Chem. B, 118, 8396-8405, https://doi.org/10.1021/jp502538m.
Hayashi, S., Tajkhorshid, E., and Schulten, K. (2009) Photochemical reaction dynamics of the primary event of vision studied by means of a hybrid molecular simulation, Biophys. J., 96, 403-416, https://doi.org/10.1016/j.bpj.2008.09.049.
Rajput, J., Rahbek, D. B., Andersen, L. H., Hirshfeld, A., Sheves, M., Altoè, P., Orlandi, G., and Garavelli, M. (2010) Probing and modeling the absorption of retinal protein chromophores in vacuo, Angew. Chem. Intl. Ed. Engl., 49, 1790-1793, https://doi.org/10.1002/anie.200905061.
Yoshizawa, T., and Wald, G. (1963) Pre-lumirhodopsin and the bleaching of visual pigments, Nature, 197, 1279-1286, https://doi.org/10.1038/1971279a0.
Takahashi, T., Mochizuki, Y., Kamo, N., and Kobatake, Y. (1985) Evidence that the long-lifetime photointermediate of s-rhodopsin is a receptor for negative phototaxis in Halobacterium halobium, Biochem. Biophys. Res. Commun., 127, 99-105, https://doi.org/10.1016/s0006-291x(85)80131-5.
Bruun, S., Stoeppler, D., Keidel, A., Kuhlmann, U., Luck, M., Diehl, A., Geiger, M. A., Woodmansee, D., Trauner, D., Hegemann, P., Oschkinat, H., Hildebrandt, P., and Stehfest, K. (2015) Light-dark adaptation of channelrhodopsin involves photoconversion between the all-trans and 13-cis retinal isomers, Biochemistry, 54, 5389-5400, https://doi.org/10.1021/acs.biochem.5b00597.
Dixon, S. F., and Cooper, A. (1987) Quantum efficiencies of the reversible photoreaction of octopus rhodopsin, Photochem. Photobiol., 46, 115-119, https://doi.org/10.1111/j.1751-1097.1987.tb04744.x.
Ostrovsky, M. A., and Weetall, H. H. (1998) Octopus rhodopsin photoreversibility of a crude extract from whole retina over several weeks’ duration, Biosens. Bioelectron., 13, 61-65, https://doi.org/10.1016/S0956-5663(97)00078-X.
Inoue, K., Tsuda, M., and Terazima, M. (2007) Photoreverse reaction dynamics of octopus rhodopsin, Biophys. J., 92, 3643-3651, https://doi.org/10.1529/biophysj.106.101741.
Koyanagi, M., and Terakita, A. (2008) Gq-coupled rhodopsin subfamily composed of invertebrate visual pigment and melanopsin, Photochem. Photobiol., 84, 1024-1030, https://doi.org/10.1111/j.1751-1097.2008.00369.x.
Balashov, S. P., Imasheva, E. S., Govindjee, R., and Ebrey, T. G. (1991) Quantum yield ratio of the forward and back light reactions of bacteriorhodopsin at low temperature and photosteady-state concentration of the bathoproduct K, Photochem. Photobiol., 54, 955-961, https://doi.org/10.1111/j.1751-1097.1991.tb02116.x.
Suzuki, T., and Callender, R. H. (1981) Primary photochemistry and photoisomerization of retinal at 77 degrees K in cattle and squid rhodopsins, Biophys. J., 34, 261-270, https://doi.org/10.1016/S0006-3495(81)84848-5.
Schapiro, I., Ryazantsev, M. N., Frutos, L. M., Ferré, N., Lindh, R., and Olivucci, M. (2011) The ultrafast photoisomerizations of rhodopsin and bathorhodopsin are modulated by bond length alternation and HOOP driven electronic effects, J. Am. Chem. Soc., 133, 3354-3364, https://doi.org/10.1021/ja1056196.
Schoenlein, R. W., Peteanu, L. A., Mathies, R. A., and Shank, C. V. (1991) The first step in vision: Femtosecond isomerization of rhodopsin, Science, 254, 412-415, https://doi.org/10.1126/science.1925597.
Altoè, P., Cembran, A., Olivucci, M., and Garavelli, M. (2010) Aborted double bicycle-pedal isomerization with hydrogen bond breaking is the primary event of bacteriorhodopsin proton pumping, Proc. Natl. Acad. Sci. USA, 107, 20172-20177, https://doi.org/10.1073/pnas.1007000107.
Koyama, Y., Kubo, K., Komori, M., Yasuda, H., and Mukai, Y. (1991) Effect of protonation on the isomerization properties of n-butylamine Schiff base of isomeric retinal as revealed by direct HPLC analyses: Selection of isomerization pathways by retinal proteins, Photochem. Photobiol., 54, 433-443, https://doi.org/10.1111/j.1751-1097.1991.tb02038.x.
Ikuta, T., Shihoya, W., Sugiura, M., Yoshida, K., Watari, M., Tokano, T., Yamashita, K., Katayama, K., Tsunoda, S. P., Uchihashi, T., Kandori, H., and Nureki, O. (2020) Structural insights into the mechanism of rhodopsin phosphodiesterase, Nat. Commun., 11, 5605, https://doi.org/10.1038/s41467-020-19376-7.
Drachev, L. A., Kaulen, A. D., and Skulachev, V. P. (1977) Temporal characteristics of bacteriorhodopsin as a molecular biological generator of current, Mol. Biol. (Moscow), 11, 1377-1387.
Bol’shakov, V. I., Drachev, L. A., Kalamkarov, G. R., Kaulen, A. D., and Skulachev, V. P. (1979) Community of properties of bacterial and visual rhodopsins: light energy conversion to electric potential difference, Dokl. Akad. Nauk SSSR, 249, 1462-1466.
Drachev, L. A., Kalamkarov, G. R., Kaulen, A. D., Ostrovsky, M. A., and Skulachev, V. P. (1981) Fast stages of photoelectric processes in biological membranes. II. Visual rhodopsin, Eur. J. Biochem., 117, 471-481, https://doi.org/10.1111/j.1432-1033.1981.tb06362.x.
Drachev, L. A., Kaulen, A. D., Khitrina, L. V., and Skulachev, V. P. (1981) Fast stages of photoelectric processes in biological membranes. I. Bacteriorhodopsin, Eur. J. Biochem., 117, 461-470, https://doi.org/10.1111/j.1432-1033.1981.tb06361.x.
Shevchenko, T. F., Kalamkarov, G. R., and Ostrovsky, M. A. (1987) The lack of H+ transfer across the photoreceptor membrane during rhodopsin photolysis [in Russian], Sensory Sistems, 1, 117-126.
Ostrovsky, M. A., Fedorovich, I. B., and Poliak, S. E. (1968) Change in pH of the medium during illumination of the retina and of a suspension of outer segments of photoreceptors, Biofizika, 13, 338-339.
Kalamkarov, G. R., and Ostrovsky, M. A. (2002) Molecular mechanisms of visual reception, Moscow: Nauka, pp. 1-280.
Brindley, G. S., and Gardner-Medwin, A. R. (1966) The origin of the early receptor potential of the retina, J. Physiol., 182, 185-194, https://doi.org/10.1113/jphysiol.1966.sp007817.
Acknowledgments
The authors are grateful to Dr. P. A. Kusochek and V. V. Logvinov for performing part of quantum-chemical calculations. The calculations were carried out using the HPC resources of the Laboratory of Quantum Photodynamics (RSC Tornado) provided through the Lomonosov Moscow State University Program of Development.
Funding
This work was financially supported by the Ministry of Science and Higher Education of the Russian Federation (agreement no. 075-15-2020-795, inner no. 13.1902.21.0027).
Author information
Authors and Affiliations
Contributions
M.A.O. concept and supervision of the study; M.A.O., O.A.S., A.V.B., and T.B.F. writing and editing of the paper.
Corresponding author
Ethics declarations
The authors declare no conflict of interests in financial or any other sphere. This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
Open access. This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit https://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ostrovsky, M.A., Smitienko, O.A., Bochenkova, A.V. et al. Similarities and Differences in Photochemistry of Type I and Type II Rhodopsins. Biochemistry Moscow 88, 1528–1543 (2023). https://doi.org/10.1134/S0006297923100097
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006297923100097