Abstract
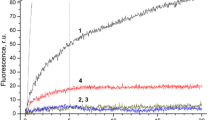
Plastoquinone bound with decyltriphenylphosphonium cation (SkQ1) penetrating through the membrane in nanomolar concentrations inhibited H2O2 generation in cells of epidermis of pea seedling leaves that was detected by the fluorescence of 2′,7′-dichlorofluorescein. Photosynthetic electron transfer in chloroplasts isolated from pea leaves is suppressed by SkQ1 at micromolar concentrations: the electron transfer in chloroplasts under the action of photosystem II or I (with silicomolybdate or methyl viologen as electron acceptors, respectively) is more sensitive to SkQ1 than under the action of photosystem II + I (with ferricyanide or p-benzoquinone as electron acceptors). SkQ1 reduced by borohydride is oxidized by ferricyanide, p-benzoquinone, and, to a lesser extent, by silicomolybdate, but not by methyl viologen. SkQ1 is not effective as an electron acceptor supporting O2 evolution from water in illuminated chloroplasts. The data on suppression of photosynthetic O2 evolution or consumption show that SkQ1, similarly to phenazine methosulfate, causes conversion of the chloroplast redox-chain from non-cyclic electron transfer mode to the cyclic mode without O2 evolution. Oxidation of NADH or succinate in mitochondria isolated from pea roots is stimulated by SkQ1.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- Asc:
-
ascorbate
- BQ:
-
p-benzoquinone
- DCF:
-
2′,7′-dichlorofluorescein
- DCMU:
-
3-(3,4-dichlorophenyl)-1,1-dimethylurea
- DTPP+ :
-
decyltriphenylphosphonium cation
- EC:
-
epidermal cells
- FeCy:
-
ferricyanide
- GC:
-
guard cells
- MitoQ:
-
10-(6′-ubiquinonyl)decyltriphenylphosphonium
- MV:
-
methyl viologen
- PCD:
-
programmed cell death
- PMS:
-
phenazine methosulfate
- PS I(II):
-
photosystem I(II)
- ROS:
-
reactive oxygen species
- SH:
-
salicylhydroxamate
- SiMo:
-
silicomolybdate
- SkQ1:
-
10-(6′-plastoquinonyl)decyltriphenylphosphonium
- SkQ3:
-
10-(6′-methylplastoquinonyl)decyltriphenylphosphonium
- SkQR1:
-
10-(plastoquinonyl)decylrhodamine
- TMPD:
-
N,N,N′,N′-tetramethyl-p-phenylenediamine
- Δψ:
-
electric potential transmembrane difference
References
Skulachev, V. P. (2006) Bioenergetic aspects of apoptosis, necrosis and mitoptosis, Apoptosis, 11, 473–485.
Samuilov, V. D., Lagunova, E. M., Dzyubinskaya, E. V., Izyumov, D. S., Kiselevsky, D. V., and Makarova, Y. V. (2002) Involvement of chloroplasts in the death of plant cells, Biochemistry (Moscow), 67, 627–634.
Samuilov, V. D., Lagunova, E. M., Kiselevsky, D. B., Dzyubinskaya, E. V., Makarova, Y. V., and Gusev, M. V. (2003) Participation of chloroplasts in plant apoptosis, Biosci. Rep., 23, 103–117.
Liberman, E. A., and Skulachev, V. P. (1970) Conversion of biomembrane-produced energy into electric form. IV. General discussion, Biochim. Biophys. Acta, 216, 30–42.
Kelso, G. F., Porteous, C. M., Coulter, C. V., Hughes, G., Porteous, W. K., Ladgerwood, E. C., Smith, R. A., and Murphy, M. P. (2001) Selective targeting of a redox-active ubiquinone to mitochondria within cells. Antioxidant and antiapoptotic properties, J. Biol. Chem., 276, 4588–4596.
Dhanasekaran, A., Kotamraju, S., Kalivendi, S. V., Matsunaga, T., Shang, T., Keszler, A., Joseph, J., and Kalyanaraman, B. (2004) Supplementation of endothelial cells with mitochondria-targeted antioxidants inhibit peroxide-induced mitochondrial iron uptake, oxidative damage, and apoptosis, J. Biol. Chem., 279, 37575–37587.
James, A. M., Cocheme, H. M., Smith, R. A., and Murphy, M. P. (2005) Interactions of mitochondria-targeted and untargeted ubiquinones with the mitochondrial respiratory chain and reactive oxygen species. Implications for the use of exogenous ubiquinones as therapies and experimental tools, J. Biol. Chem., 280, 21295–21312.
Skulachev, V. P. (2007) A biochemical approach to the problem of aging: “megaproject” on membrane-penetrating ions. The first results and prospects, Biochemistry (Moscow), 72, 1385–1399.
Vasil’ev, E. V., Dzyubinskaya, E. V., Kiselevsky, D. B., Shestak, A. A., and Samuilov, V. D. (2011) Programmed cell death in plants: protective effect of mitochondrial-targeted quinines, Biochemistry (Moscow), 76, 1120–1132.
Barsky, E. L., Gubanova, O. N., and Samuilov, V. D. (1991) Inhibition of photosynthetic electron transfer in chloroplasts by carbonyl cyanide m-chlorophenylhydrazone, Biokhimiya, 56, 434–438.
Millenaar, F. F., Benschop, J. J., Wagner, A. M., and Lambers, H. (1998) The role of the alternative oxidase in stabilizing the in vivo reduction state of the ubiquinone pool and the activation state of the alternative oxidase, Plant Physiol., 118, 599–607.
Arnon, D. I. (1949) Copper enzymes in isolated chloroplasts. Polyphenol oxidase in Beta vulgaris, Plant Physiol., 24, 1–15.
Smith, P. K., Krohn, R. I., Hermanson, G. T., Mallia, A. K., Gartner, F. H., Provenzano, M. D., Fujimoto, E. K., Goeke, N. M., Olson, B. J., and Klenk, D. C. (1985) Measurement of protein using bicinchoninic acid, Anal. Biochem., 150, 76–85.
LeBel, C. P., Ischiropoulos, H., and Bondy, S. C. (1992) Evaluation of the probe 2′,7′-dichlorofluorescin as an indicator of reactive oxygen species formation and oxidative stress, Chem. Res. Toxicol., 5, 227–231
Wrona, M., Patel, K., and Wardman, P. (2005) Reactivity of 2′,7′-dichlorodihydrofluorescein and dihydrorhodamin 123 and their oxidized forms toward carbonate, nitrogen dioxide, and hydroxyl radicals, Free Radic. Biol. Med., 38, 262–270.
Vasil’ev, L. A., Dzyubinskaya, E. V., Zinovkin, P. A., Kiselevsky, D. B., Lobesheva, N. V., and Samuilov, V. D. (2009) Chitosan-induced programmed cell death in plants, Biochemistry (Moscow), 74, 1035–1044.
Sagi, M., and Fluhr, R. (2006) Production of reactive oxygen species by plant NADPH oxidases, Plant Physiol., 141, 336–340.
Graan, T. (1986) The interaction of silicomolybdate with the photosystem II herbicide-binding site, FEBS Lett., 206, 9–14.
Fujii, T., Yokoyama, E., Inoue, K., and Sakurai, H. (1990) The sites of electron donation of photosystem I to methyl viologen, Biochim. Biophys. Acta, 1015, 41–48.
Braun, G., Driesenaar, A. R. J., Shalgi, E., and Malkin, S. (1992) Manipulation of the imbalance for linear electron flow activities between photosystems I and II of photosynthesis by cyclic electron flow cofactors, Biochim. Biophys. Acta, 1099, 57–66.
Moore, A. L., and Siedow, J. N. (1991) The regulation and nature of the cyanide-resistant alternative oxidase of plant mitochondria, Biochim. Biophys. Acta, 1059, 121–140.
Severina, I. I., Severin, F. F., Korshunova, G. A., Sumbatyan, N. V., Ilyasova, T. M., Simonyan, R. A., Rogov, A. G., Trendeleva, T. A., Zvyagilskaya, R. A., Dugina, V. B., Domnina, L. V., Fetisova, E. K., Lyamzaev, K. G., Vyssokikh, M. Yu., Chernyak, B. V., Skulachev, M. V., Skulachev, V. P., and Sadovnichii, V. A. (2013) In search of novel highly active mitochondria-targeted antioxidants: thymoquinone and its cationic derivatives, FEBS Lett., 587, 2018–2024.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V. D. Samuilov, D. B. Kiselevsky, 2015, published in Biokhimiya, 2015, Vol. 80, No. 4, pp. 489–496.
Originally published in Biochemistry (Moscow) On-Line Papers in Press, as Manuscript BM14-231, February 22, 2015.
Rights and permissions
About this article
Cite this article
Samuilov, V.D., Kiselevsky, D.B. Effect of cationic plastoquinone SkQ1 on electron transfer reactions in chloroplasts and mitochondria from pea seedlings. Biochemistry Moscow 80, 417–423 (2015). https://doi.org/10.1134/S0006297915040045
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006297915040045