Abstract
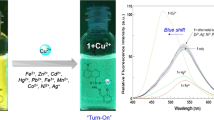
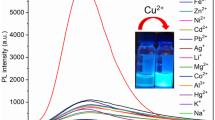
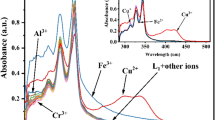
A new terephthaldehyde-based chemosensor 1 bearing an aminophenol recognition unit has been synthesized and applied to the fluorescent sensing of metal ions. Molecular system 1 acts as a highly selective and sensitive fluorescence turn-on sensor for Cu2+. The sensing mechanism has been explored. It is proposed that Cu2+ binds with the imine and hydroxyl moiety of 1 in 1: 2 binding stoichiometry, thereby enhancing the fluorescence at 386 nm. The detection limit and association constant (Ka) of 1 with Cu2+ were found to be 0.62 μM and 6.67 × 104 M−1, respectively. Chemosensor 1 has shown excellent specificity towards Cu2+ and has been successfully applied to the determination of Cu2+ in live L929 cells.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
A. P. de Silva, H. Q. N. Gunaratne, T. Gunnlaugsson, A. J. M. Huxley, C. P. McCoy, J. T. Rademacher, and T. E. Rice, Chem. Rev., 1997, 97, 1515.
L. Prodi, F. Bolletta, M. Montalti, and N. Zaccheroni, Coord. Chem. Rev., 2000, 205, 59.
C. Bargossi, M. C. Fiorini, M. Montalti, L. Prodi, and N. Zaccheroni, Coord. Chem. Rev., 2000, 208, 17.
R. L. Lieberman, and A. C. Rosenzweig, Nature, 2005, 434, 177.
K. Piontek, M. Antorini, and T. Choinowski, J. Biol. Chem., 2002, 277, 37663.
K. C. Ko, J. S. Wu, H. J. Kim, P. S. Kwon, J. W. Kim, R. A. Bartsch, J. Y. Lee, and J. S. Kim, Chem. Commun., 2011, 47, 3165.
M. F. Yardim, T. Budinova, E. Ekinci, N. Petrov, M. Razvigorova, and V. Minkova, Chemosphere, 2003, 52, 835.
M. Royzen, Z. Dai, and J. W. Canary, J. Am. Chem. Soc., 2005, 127, 1612.
E. L. Que, D. W. Domaille, and C. J. Chang, Chem. Rev., 2008, 108, 1517.
C. Vulpe, B. Levinson, S. Whitney, S. Packman, and J. Gitschier, Nat. Genet., 1993, 3, 7.
D. J. Waggoner, T. B. Bartnikas, and J. D. Gitlin, Neurobiol. Dis., 1999, 6, 221.
J. S. Valentine, and P. J. Hart, Proc. Natl. Acad. Sci. U. S. A., 2003, 100, 3617.
D. R. Brown, and H. Kozlowski, Dalton Trans., 2004, 1907.
K. J. Barnham, C. L. Masters, and A. I. Bush, Nat. Rev. Drug Discovery, 2004, 3, 205.
B. E. Kim, T. Nevitt, and D. J. Thiele, Nat. Chem. Biol., 2008, 4, 176.
G. J. Brewer, Curr. Opin. Chem. Biol., 2003, 7, 207.
G. L. Millhauser, Acc. Chem. Res., 2004, 37, 79.
S. P. Leach, M. D. Salman, and D. Hamar, Anim. Health Res. Rev., 2006, 7, 97.
K. J. Barnham, and A. I. Bush, Curr. Opin. Chem. Biol., 2008, 12, 222.
R. R. Crichton, D. T. Dexter, and R. J. Ward, Coord. Chem. Rev., 2008, 252, 1189.
Metal Ions in Biological Systems: Properties of Copper, ed. M. Dekker and H. Sigel, New York, 1981, p. 12.
E. L. Que, D. W. Domaille, and C. J. Chang, Chem. Rev., 2008, 108, 1517.
E. Gaggelli, H. Kozlowski, D. Valensin, and G. Valensin, Chem. Rev., 2006, 106, 1995.
H. N. Kim, M. H. Lee, H. J. Kim, J. S. Kim, and J. Yoon, Chem. Soc. Rev., 2008, 37, 1465.
S. Seo, H. Y. Lee, M. Park, J. M. Lim, D. Kang, J. Yoon, and J. H. Jung, Eur. J. Inorg. Chem., 2010, 843.
H. H. Wang, L. Xue, Z. J. Fang, G. P. Li, and H. Jiang, New J. Chem., 2010, 34, 1239.
W. Wang, A. Fu, J. You, G. Gao, J. Lan, and L. Chen, Tetrahedron, 2010, 66, 3695.
J. P. Sumner, N. M. Westerberg, A. K. Stoddard, T. K. Hurst, M. Cramer, R. B. Thompson, C. A. Fierke, and R. Kopelman, Biosens. Bioelectron., 2006, 21, 1302.
M. H. Kim, H. H. Jang, S. Yi, S. Chang, and M. S. Han, Chem. Commun., 2009, 4838.
Z. Xu, J. Yoon, and D. R. Spring, Chem. Commun., 2010, 46, 2563.
J. Liu, and Y. Lu, J. Am. Chem. Soc., 2007, 129, 9838.
Y. Xiang, A. Tong, P. Jin, and Y. Ju, Org. Lett., 2006, 8, 2863.
A. P. S. Gonzales, M. A. Firmino, C. S. Nomura, F. R. P. Rocha, P. V. Oliveira, and I. Gaubeur, Anal. Chim. Acta, 2009, 636, 198.
J. S. Becker, A. Matusch, C. Depboylu, J. Dobrowolska, and M. V. Zoriy, Anal. Chem., 2007, 79, 3208.
Y. Liu, P. Liang, and L. Guo, Talanta, 2005, 68, 25.
P. Pathirathna, Y. Yang, K. Forzley, S. P. McElmurry, and P. Hashemi, Anal. Chem., 2012, 84, 6298.
Y.-Q. Weng, F. Yue, Y.-R. Zhong, and B.-H. Ye, Inorg. Chem., 2007, 46, 7749.
K. L. Ciesienski, L. M. Hyman, S. Derisavifard, and K. J. Franz, Inorg. Chem., 2010, 49, 6808.
S.-P. Wu, K.-J. Du, and Y.-M. Sung, Dalton Trans., 2010, 39, 4363.
S. H. Choi, K. Pang, K. Kim, and D. G. Churchill, Inorg. Chem., 2007, 46, 10564.
A. P. de Silva, H. Q. N. Gunaratne, T. Gunnlaugsson, A. J. M. Huxley, C. P. McCoy, J. T. Rademacher, and T. E. Rice, Chem. Rev., 1997, 97, 1515.
B. Valeur, and I. Leray, Coord. Chem. Rev., 2000, 205, 3.
H. Chen, Y. Wu, Y. Cheng, H. Yang, F. Li, and P. Yang, Inorg. Chem. Commun., 2007, 10, 1413.
C. Sousa, P. Gameiro, C. Freire, and B. Castro, Polyhedron, 2004, 23, 1401.
O. Oter, K. Ertekin, R. Kilincarslan, M. Ulusoy, and B. Çetinkaya, Dyes Pigm., 2007, 74, 730.
N. Aksuner, E. Henden, A. Çukurovali, and I. Yilmaz, Sens. Actuators, B, 2008, 134, 510.
SMART: Bruker Molecular Analysis Research Tools, version 5.618, Bruker Analytical X-ray Systems, Madison, WI, 2000.
G. M. Sheldrick, SAINT-NT, version 6.04, Bruker Analytical X-ray Systems, Madison, WI, 2001.
G. M. Sheldrick, SHELXTL-NT, version 6.10, Bruker Analytical X-ray Systems, Madison, WI, 2000.
B. Klaus, DIAMOND, version 2.1 c, University of Bonn, Bonn, Germany, 1999.
A. Kuwar, R. Patil, A. Singh, S. K. Sahoo, and N. Singh, J. Mater. Chem. C, 2015, 3, 453–460.
M. Yang, W. Meng, X. Liu, N. Su, J. Zhou, and B. Yang, RSC Adv., 2014, 4, 22288.
H. A. Benesi, and J. H. Hildebrand, J. Am. Chem. Soc., 1949, 71, 2703.
L. Gao, J. Q. Wang, L. Huang, X. X. Fan, J. H. Zhu, Y. Wang, and Z. G. Zou, Inorg. Chem., 2007, 46, 10287.
W. Lin, L. Yuan, X. Cao, B. Chen, and Y. Feng, Sens. Actuators, B, 2009, 138, 637.
M. Ganjali, M. Emami, and M. Salavati-Niasari, Bull. Korean Chem. Soc., 2002, 23, 1394.
P. Job, Ann. Chim., 1928, 9, 113–203.
M. J. Frisch, {etet al.}, Gaussian 09, G09W®, Gaussian Inc., Wallingford, USA, 2009.
Acknowledgments
AK acknowledges Department of Science and Technology Government of India for funding and School of Chemical Sciences, North Maharashtra University, Jalgaon, India for providing infrastructure for the accomplishment of this project.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Torawane, P., Keshav, K., Kumawat, M.K. et al. A novel terephthalaldehyde based turn-on fluorescent chemosensor for Cu2+ and its application in imaging of living cells. Photochem Photobiol Sci 16, 1464–1470 (2017). https://doi.org/10.1039/c7pp00182g
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c7pp00182g