Abstract
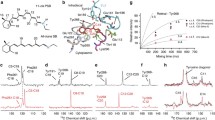
A series of vitamin-A aldehydes (retinals) with modified alkyl group substituents (9-demethyl-, 9-ethyl-, 9-isopropyl-, 10-methyl, 10-methyl-13-demethyl-, and 13-demethyl retinal) was synthesized and their 11-cis isomers were used as chromophores to reconstitute the visual pigment rhodopsin. Structural changes were selectively introduced around the photoisomerizing C11?C12 bond. The effect of these structural changes on rhodopsin formation and bleaching was determined. Global fit of assembly kinetics yielded lifetimes and spectral features of the assembly intermediates. Rhodopsin formation proceeds stepwise with prolonged lifetimes especially for 9-demethyl retinal (longest lifetime t3 = 7500 s, cf., 3500 s for retinal), and for 10-methyl retinal (t3 = 7850 s). These slowed-down processes are interpreted as either a loss of fixation (9dm) or an increased steric hindrance (10me) during the conformational adjustment within the protein. Combined quantum mechanics and molecular mechanics (QM/MM) simulations provided structural insight into the retinal analogues-assembled, full-length rhodopsins. Extinction coefficients, quantum yields and kinetics of the bleaching process (µs-to-ms time range) were determined. Global fit analysis yielded lifetimes and spectral features of bleaching intermediates, revealing remarkably altered kinetics: whereas the slowest process of wild-type rhodopsin and of bleached and 11-cis retinal assembled rhodopsin takes place with lifetimes of 7 and 3.8 s, respectively, this process for 10-methyl-13-demethyl retinal was nearly 10 h (34670 s), coming to completion only after ca. 50 h. The structural changes in retinal derivatives clearly identify the precise interactions between chromophore and protein during the light-induced changes that yield the outstanding efficiency of rhodopsin.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
I. Szundi, A. R. de Lera, Y. Pazos, R. Alvarez, M. Oliana, M. Sheves, J. W. Lewis, D. S. Kliger, Biochemistry, 2002, 41, 2028–2035.
U. M. Ganter, W. Gärtner, F. Siebert, Eur. Biophys. J., 1990, 18, 295–299.
K. Palczewski, Vertebrate Phototransduction and the Visual Cycle, Part A, Elsevier BV, San Diego, CA, 2000.
O. P. Ernst, D. T. Lodowski, M. Elstner, P. Hegemann, L. S. Brown, H. Kandori, Chem. Rev., 2014, 114, 126–163.
K. Palczewski, Vertebrate Phototransduction and the Visual Cycle, Part B, Elsevier BV, San Diego, CA, 2000.
K. P. Hofmann, P. Scheerer, P. W. Hildebrand, H.-W. Choe, J. H. Park, M. Heck, O. P. Ernst, Trends Biochem. Sci., 2009, 34, 540–552.
C. K. Meyer, M. Böhme, A. Ockenfels, W. Gärtner, K. P. Hofmann, O. P. Ernst, J. Biol. Chem., 2000, 275, 19713–19718.
M. Han, M. Groesbeek, S. O. Smith, T. P. Sakmar, Biochemistry, 1998, 37, 538–545.
K. Nakanishi, N. Berova, N. Fishkin, N. Fujioka, J. Chin. Chem. Soc., 2002, 49, 443–451.
F. DeLange, P. H. M. Bovee-Geurts, J. VanOostrum, M. D. Portier, P. J. E. Verdegem, J. Lugtenburg, W. J. DeGrip, Biochemistry, 1998, 37, 1411–1420.
M. A. Verhoeven, P. H. M. Bovee-Geurts, H. J. M. de Groot, J. Lugtenburg, W. J. DeGrip, J. Mol. Biol., 2006, 363, 98–113.
L. U. Colmenares, R. S. H. Liu, Tetrahedron, 1996, 52, 109–118.
J. Ni, J. Liu, L. U. Colmenares, R. S. H. Liu, Tetrahedron Lett., 2001, 42, 1643–1644.
P. H. M. Bovee-Geurts, I. Fernández Fernández, R. S. H. Liu, R. A. Mathies, J. Lugtenburg, W. J. DeGrip, J. Am. Chem. Soc., 2009, 131, 17933–17942.
J. W. Lewis, G.-B. Fan, M. Sheves, I. Szundi, D. S. Kliger, J. Am. Chem. Soc., 2001, 123, 10024–10029.
R. Vogel, G. B. Fan, S. Ludeke, F. Siebert, M. Sheves, J. Biol. Chem., 2002, 277, 40222–40228.
G. Fan, F. Siebert, M. Sheves, R. Vogel, J. Biol. Chem., 2002, 277, 40229–40234.
Y. Wang, P. H. M. Bovee-Geurts, J. Lugtenburg, W. J. DeGrip, Biochemistry, 2004, 43, 14802–14810.
R. S. H. Liu, F. Crescitelli, M. Denny, H. Matsumoto, A. E. Asato, Biochemistry, 1986, 25, 7026–7030.
W. J. de Grip, P. H. M. Bovee-Geurts, Y. Wang, M. A. Verhoeven, J. Lugtenburg, J. Nat. Prod., 2011, 74, 383–390.
R. Vogel, S. Lüdeke, F. Siebert, T. P. Sakmar, A. Hirshfeld, M. Sheves, Biochemistry, 2006, 45, 1640–1652.
D. Koch, W. Gärtner, Photochem. Photobiol., 1997, 65, 181–186.
W. J. de Grip, F. J. M. Daemen and S. L. Bonting, in Methods in Enzymology, Vitamins and Coenzymes Part F, ed. D. B. McCormick and L. D. Wright, Academic Press, 1980, vol. 67, pp. 301–320.
H. J. A. Dartnall, C. F. Goodeve, R. J. Lythgoe, Proc. R. Soc. London, A, 1936, 156, 158–170.
W. Gärtner, D. Ullrich, K. Vogt, Photochem. Photobiol., 1991, 54, 1047–1055.
J. M. Strassburger, W. Gärtner, S. E. Braslavsky, Biophys. J., 1997, 72, 2294–2303.
P. Schmidt, T. Gensch, A. Remberg, W. Gärtner, S. E. Braslavsky, K. Schaffner, Photochem. Photobiol., 1998, 68, 754.
A. Warshel, M. Levitt, J. Mol. Biol., 1976, 103, 227–249.
H. M. Senn, W. Thiel, Angew. Chem., Int. Ed., 2009, 48, 1198–1229.
A. Warshel, Angew. Chem., Int. Ed., 2014, 53, 10020–10031.
V. Hornak, R. Abel, A. Okur, B. Strockbine, A. Roitberg, C. Simmerling, Proteins: Struct., Funct., Bioinf., 2006, 65, 712–725.
T. Vreven, K. Morokuma, Ö. Farkas, H. B. Schlegel, M. J. Frisch, J. Comput. Chem., 2003, 24, 760–769.
N. Ferré, J. G. Ángyán, Chem. Phys. Lett., 2002, 356, 331–339.
F. Aquilante, J. Autschbach, R. K. Carlson, L. F. Chibotaru, M. G. Delcey, L. De Vico, I. F. Galván, N. Ferré, L. M. Frutos, L. Gagliardi, M. Garavelli, A. Giussani, C. E. Hoyer, G. Li Manni, H. Lischka, D. Ma, P. Å. Malmqvist, T. Müller, A. Nenov, M. Olivucci, T. B. Pedersen, D. Peng, F. Plasser, B. Pritchard, M. Reiher, I. Rivalta, I. Schapiro, J. Segarra-Martí, M. Stenrup, D. G. Truhlar, L. Ungur, A. Valentini, S. Vancoillie, V. Veryazov, V. P. Vysotskiy, O. Weingart, F. Zapata, R. Lindh, J. Comput. Chem., 2015. 10.1002/jcc.24221
J. W. Ponder, F. M. Richards, J. Comput. Chem., 1987, 8, 1016–1024.
T. Okada, M. Sugihara, A.-N. Bondar, M. Elstner, P. Entel, V. Buss, J. Mol. Biol., 2004, 342, 571–583.
F. Melaccio, M. Olivucci, R. Lindh, N. Ferré, Int. J. Quantum Chem., 2011, 111, 3339–3346.
A. Strambi, P. B. Coto, L. M. Frutos, N. Ferré, M. Olivucci, J. Am. Chem. Soc., 2008, 130, 3382–3388.
S. Sekharan, V. Buss, J. Am. Chem. Soc., 2008, 130, 17220–17221.
J. S. Frähmcke, M. Wanko, P. Phatak, M. A. Mroginski, M. Elstner, J. Phys. Chem. B, 2010, 114, 11338–11352.
G. Tomasello, G. Olaso-González, P. Altoè, M. Stenta, L. Serrano-Andrés, M. Merchán, G. Orlandi, A. Bottoni, M. Garavelli, J. Am. Chem. Soc., 2009, 131, 5172–5186.
T. Andruniów, M. Olivucci, J. Chem. Theory Comput., 2009, 5, 3096–3104.
N. Ferré, M. Olivucci, J. Am. Chem. Soc., 2003, 125, 6868–6869.
E. N. Laricheva, S. Gozem, S. Rinaldi, F. Melaccio, A. Valentini, M. Olivucci, J. Chem. Theory Comput., 2012, 8, 2559–2563.
T. Andruniów, N. Ferré, M. Olivucci, Proc. Natl. Acad. Sci. U. S. A., 2004, 101, 17908–17913.
S. Sekharan, M. Sugihara, V. Buss, Angew. Chem., Int. Ed., 2007, 46, 269–271.
S. Sekharan, M. Sugihara, O. Weingart, T. Okada, V. Buss, J. Am. Chem. Soc., 2007, 129, 1052–1054.
S. Sekharan, Photochem. Photobiol., 2009, 85, 517–520.
S. Gozem, M. Huntress, I. Schapiro, R. Lindh, A. A. Granovsky, C. Angeli, M. Olivucci, J. Chem. Theory Comput., 2012, 8, 4069–4080.
S. Gozem, F. Melaccio, R. Lindh, A. I. Krylov, A. A. Granovsky, C. Angeli, M. Olivucci, J. Chem. Theory Comput., 2013, 9, 4495–4506.
E. Walczak, T. Andruniów, Phys. Chem. Chem. Phys., 2015, 17, 17169–17181.
P. J. v. d. Tempel, H. O. Huisman, Tetrahedron, 1966, 22, 293–299.
R. N. Gedye, P. C. Arora, K. Deck, Can. J. Chem., 1971, 49, 1764–1766.
J. W. Lewis, I. Pinkas, M. Sheves, M. Ottolenghi, D. S. Kliger, J. Am. Chem. Soc., 1995, 117, 918–923.
M. Groesbeek, R. van der Steen, J. C. van Vliet, L. B. J. Vertegaal, J. Lugtenburg, Recl. Trav. Chim. Pays-Bas, 1989, 108, 427–436.
F. J. H. M. Jansen, M. Kwestro, D. Schmitt, J. Lugtenburg, Recl. Trav. Chim. Pays-Bas, 1994, 113, 552–562.
W. Gaertner, P. Towner, H. Hopf, D. Oesterhelt, Biochemistry, 1983, 22, 2637–2644.
E. Tajkhorshid, B. Paizs, S. Suhai, J. Phys. Chem. B, 1999, 103, 4518–4527.
B. Honig, U. Dinur, K. Nakanishi, V. Balogh-Nair, M. A. Gawinowicz, M. Arnaboldi, M. G. Motto, J. Am. Chem. Soc., 1979, 101, 7084–7086.
M. Schreiber, M. R. Silva-Junior, S. P. A. Sauer, W. Thiel, J. Chem. Phys., 2008, 128, 134110.
M. Sugihara, V. Buss, Biochemistry, 2008, 47, 13733–13735.
G. G. Kochendoerfer, P. J. E. Verdegem, I. van der Hoef, J. Lugtenburg, R. A. Mathies, Biochemistry, 1996, 35, 16230–16240.
H. J. A. Dartnall, Vision Res., 1968, 8, 339–358.
M. Heck, S. A. Schadel, D. Maretzki, F. J. Bartl, E. Ritter, K. Palczewski, K. P. Hofmann, J. Biol. Chem., 2002, 278, 3162–3169.
R. Vogel, F. Siebert, G. Mathias, P. Tavan, G. Fan, M. Sheves, Biochemistry, 2003, 42, 9863–9874.
B. Knierim, K. P. Hofmann, O. P. Ernst, W. L. Hubbell, Proc. Natl. Acad. Sci. U. S. A., 2007, 104, 20290–20295.
E. Hessel, M. Heck, P. Muller, A. Herrmann, K. P. Hofmann, J. Biol. Chem., 2003, 278, 22853–22860.
S. Jäger, K. Palczewski, K. P. Hofmann, Biochemistry, 1996, 35, 2901–2908.
P. B. S. Dawadi, J. Lugtenburg, Molecules, 2010, 15, 1825–1872.
W. Gärtner, S. Ternieden, J. Photochem. Photobiol., B, 1996, 33, 83–86.
Q. Tan, K. Nakanishi, R. K. Crouch, J. Am. Chem. Soc., 1998, 120, 12357–12358.
M. E. Estevez, A. V. Kolesnikov, P. Ala-Laurila, R. K. Crouch, V. I. Govardovskii, M. C. Cornwall, J. Gen. Physiol., 2009, 134, 137–150.
M. Han, S. W. Lin, S. O. Smith, T. P. Sakmar, J. Biol. Chem., 1996, 271, 32330–32336.
M. Han, M. Groesbeek, T. P. Sakmar, S. O. Smith, Proc. Natl. Acad. Sci. U. S. A., 1997, 94, 13442–13447.
R. Nelson, J. K. deRiel, A. Kropf, Proc. Natl. Acad. Sci. U. S. A., 1970, 66, 531–538.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Electronic supplementary information (ESI) available. See DOI: 10.1039/c5pp00322a
Rights and permissions
About this article
Cite this article
Ockenfels, A., Schapiro, I. & Gärtner, W. Rhodopsins carrying modified chromophores — the ‘making of’, structural modelling and their light-induced reactivity. Photochem Photobiol Sci 15, 297–308 (2016). https://doi.org/10.1039/c5pp00322a
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c5pp00322a