Abstract
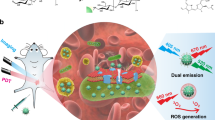
Targeted imaging and antimicrobial photodynamic inactivation (PDI) are emerging methods for detecting and eradicating pathogenic microorganisms. This study describes two structurally related optical probes that are conjugates of a zinc(ii)-dipicolylamine targeting unit and a BODIPY chromophore. One probe is a microbial targeted fluorescent imaging agent, mSeek, and the other is an oxygen photosensitizing analogue, mDestroy. The conjugates exhibited high fluorescence quantum yield and singlet oxygen production, respectively. Fluorescence imaging and detection studies examined four bacterial strains: E. coli, S. aureus, K. pneumonia, and B. thuringiensis vegetative cells and purified spores. The fluorescent probe, mSeek, is not phototoxic and enabled detection of all tested bacteria at concentrations of ≈100 CFU mL-1 for B. thuringiensis spores, ≈1000 CFU mL−1 for S. aureus and ≈10 000 CFU mL−1 for E. coli. The photosensitizer analogue, mDestroy, inactivated 992–99.99% of bacterial samples and selectively killed bacterial cells in the presence of mammalian cells. However, mDestroy was ineffective against B. thuringiensis spores. Together, the results demonstrate a new two-probe strategy to optimize PDI of bacterial infection/contamination.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- PDI:
-
Photodynamic inactivation
- PS:
-
Photosensitizer
- 1O2:
-
Singlet oxygen
- ZnDPA:
-
Zinc(ii)-dipicolylamine
- DPB:
-
1,3-diphenylisobenzofuran
References
B. Spellberg, R. Guidos, D. Gilbert, J. Bradley, H. W. Boucher, W. M. Scheld, J. G. Bartlett, J. Edwards, Jr., The epidemic of antibiotic-resistant infections: a call to action for the medical community from the Infectious Diseases Society of America, Clin. lnfect. Dis., 2008, 46, 155–164.
E. R. Sydnor, T. M. Perl, Hospital epidemiology and infection control in acute-care settings, Clin. Microbiol. Rev., 2011, 24, 141–173.
F. F. Sperandio, Y. Y. Huang, M. R. Hamblin, Antimicrobial photodynamic therapy to kill Gram-negative bacteria, Recent Pat. Anti-infect. Drug Discovery, 2013, 8, 108–120.
J. L. Wardlaw, T. J. Sullivan, C. N. Lux, F. W. Austin, Photodynamic therapy against common bacteria causing wound and skin infections, Vet. J., 2012, 192, 374–377.
K. Konopka, T. Goslinski, Photodynamic therapy in dentistry, J. Dent. Res., 2007, 86, 694–707.
S. Noimark, C. W. Dunnill, I. P. Parkin, Shining light on materials-a self-sterilising revolution, Adv. Drug Delivery Rev., 2013, 65, 570–580.
T. Maisch, S. Hackbarth, J. Regensburger, A. Felgentrager, W. Baumler, M. Landthaler, B. Roder, Photodynamic inactivation of multi-resistant bacteria (PIB) - a new approach to treat superficial infections in the 21st century, J. Dtsch. Dermatol. Ges., 2011, 9, 360–366.
F. M. Lauro, P. Pretto, L. Covolo, G. Jori, G. Bertoloni, Photoinactivation of bacterial strains involved in periodontal diseases sensitized by porphycene-polylysine conjugates, Photochem. Photobiol. Sci., 2002, 1, 468–470.
N. Komerik, M. Wilson, S. Poole, The effect of photodynamic action on two virulence factors of gram-negative bacteria, Photochem. Photobiol., 2000, 72, 676–680.
T. Dai, Y. Y. Huang, M. R. Hamblin, Photodynamic therapy for localized infections–state of the art, Photodiagn. Photodyn. Ther., 2009, 6, 170–188.
Z. Lim, J. L. Cheng, T. W. Lim, E. G. Teo, J. Wong, S. George, A. Kishen, Light activated disinfection: an alternative endodontic disinfection strategy, Aust. Dent. J., 2009, 54, 108–114.
G. A. Johnson, N. Muthukrishnan, J. P. Pellois, Photoinactivation of Gram positive and Gram negative bacteria with the antimicrobial peptide (KLAKLAK)(2) conjugated to the hydrophilic photosensitizer eosin Y, Bioconjugate Chem., 2013, 24, 114–123.
F. Liu, A. Soh Yan Ni, Y. Lim, H. Mohanram, S. Bhattacharjya, B. Xing, Lipopolysaccharide neutralizing peptide-porphyrin conjugates for effective photoinactivation and intracellular imaging of Gram-negative bacteria strains, Bioconjugate Chem., 2012, 23, 1639–1647.
M. Bhatti, A. MacRobert, B. Henderson, P. Shepherd, J. Cridland, M. Wilson, Antibody-targeted lethal photosensitization of Porphyromonas gingivalis, Antimicrob. Agents Chemother., 2000, 44, 2615–2618.
M. L. Embleton, S. P. Nair, B. D. Cookson, M. Wilson, Selective lethal photosensitization of methicillin-resistant Staphylococcus aureus using an IgG-tin(IV) chlorin e6 conjugate, J. Antimicrob. Chemother., 2002, 50, 857–864.
M. L. Embleton, S. P. Nair, B. D. Cookson, M. Wilson, Antibody-directed photodynamic therapy of methicillin resistant Staphylococcus aureus, Microb. Drug Resist., 2004, 10, 92–97.
M. L. Embleton, S. P. Nair, W. Heywood, D. C. Menon, B. D. Cookson, M. Wilson, Development of a novel targeting system for lethal photosensitization of antibiotic-resistant strains of Staphylococcus aureus, Antimicrob. Agents Chemother., 2005, 49, 3690–3696.
N. S. Soukos, L. A. Ximenez-Fyvie, M. R. Hamblin, S. S. Socransky, T. Hasan, Targeted antimicrobial photochemotherapy, Antimicrob. Agents Chemother., 1998, 42, 2595–2601.
G. P. Tegos, M. Anbe, C. Yang, T. N. Demidova, M. Satti, P. Mroz, S. Janjua, F. Gad, M. R. Hamblin, Protease-stable polycationic photosensitizer conjugates between polyethyleneimine and chlorin(e6) for broad-spectrum antimicrobial photoinactivation, Antimicrob. Agents Chemother., 2006, 50, 1402–1410.
C. L. Zhu, Q. O. Yang, L. B. Liu, F. T. Lv, S. Y. Li, G. Q. Yang, S. Wang, Multifunctional cationic poly(p-phenylene vinylene) polyelectrolytes for selective recognition, imaging, and killing of bacteria over mammalian cells, Adv. Mater., 2011, 23, 4805–4810.
E. J. O’Neil, B. D. Smith, Anion recognition using dimetallic coordination complexes, Coord. Chem. Rev., 2006, 250, 3068–3080.
R. E. Hancock, Alterations in outer membrane permeability, Annu. Rev. Microbiol., 1984, 38, 237–264.
W. M. Leevy, J. R. Johnson, C. Lakshmi, J. Morris, M. Marquez, B. D. Smith, Selective recognition of bacterial membranes by zinc(II)-coordination complexes, Chem. Commun., 2006, 1595–1597.
D. R. Rice, A. J. Plaunt, S. Turkyilmaz, M. Smith, Y. Wang, M. Rusckowski, B. D. Smith, Evaluation of 111[In]-labeled zinc-dipicolylamine tracers for SPECT imaging of bacterial infection, Mol. Imaging Biol., 2014, 1–10.
A. G. White, N. Fu, W. M. Leevy, J. J. Lee, M. A. Blasco, B. D. Smith, Optical imaging of bacterial infection in living mice using deep-red fluorescent squaraine rotaxane probes, Bioconjugate Chem., 2010, 21, 1297–1304.
A. G. White, B. D. Gray, K. Y. Pak, B. D. Smith, Deep-red fluorescent imaging probe for bacteria, Bioorg. Med. Chem. Lett., 2012, 22, 2833–2836.
K. M. DiVittorio, W. M. Leevy, E. J. O’Neil, J. R. Johnson, S. Vakulenko, J. D. Morris, K. D. Rosek, N. Serazin, S. Hilkert, S. Hurley, M. Marquez, B. D. Smith, Zinc(II) coordination complexes as membrane-active fluorescent probes and antibiotics, ChemBioChem, 2008, 9, 286–293.
T. Yogo, Y. Urano, Y. Ishitsuka, F. Maniwa, T. Nagano, Highly efficient and photostable photosensitizer based on BODIPY chromophore, J. Am. Chem. Soc., 2005, 127, 12162–12163.
T. E. Wood, A. Thompson, Advances in the chemistry of dipyrrins and their complexes, Chem. Rev., 2007, 107, 1831–1861.
H. G. Jang, M. Park, J. S. Wishnok, S. R. Tannenbaum, G. N. Wogan, Hydroxyl-specific fluorescence labeling of ABP-deoxyguanosine, PhIP-deoxyguanosine, and AFB1-formamidopyrimidine with BODIPY-FL, Anal. Biochem., 2006, 359, 151–160.
L. Bonardi, H. Kanaan, F. Camerel, P. Jolinat, P. Retailleau, R. Ziessel, Fine-tuning of yellow or red photo- and electroluminescence of functional difluoro-boradiazaindacene films, Adv. Funct. Mater., 2008, 18, 401–413.
S. G. Awuah, J. Polreis, V. Biradar, Y. You, Singlet oxygen generation by novel NIR BODIPY dyes, Org. Lett., 2011, 13, 3884–3887.
E. Diez-Barra, J. C. Garcia-Martinez, S. Merino, R. del Rey, J. Rodriguez-Lopez, P. Sanchez-Verdu, J. Tejeda, Synthesis, characterization, and optical response of dipolar and non-dipolar poly(phenylenevinylene) dendrimers, J. Org. Chem., 2001, 66, 5664–5670.
S. Yamaguchi, I. Yoshimura, T. Kohira, S. Tamaru, I. Hamachi, Cooperation between artificial receptors and supramolecular hydrogels for sensing and discriminating phosphate derivatives, J. Am. Chem. Soc., 2005, 127, 11835–11841.
S. Ozlem, E. U. Akkaya, Thinking outside the silicon box: molecular and logic as an additional layer of selectivity in singlet oxygen generation for photodynamic therapy, J. Am. Chem. Soc., 2009, 131, 48–49.
A. B. Nepomnyashchii, A. J. Pistner, A. J. Bard, J. Rosenthal, Synthesis, photophysics, electrochemistry and electrogenerated chemiluminescence of PEG-modified BODIPY dyes in organic and aqueous solutions, J. Phys. Chem. C, 2013, 117, 5599–5609.
T. L. Buhr, D. C. McPherson, B. W. Gutting, Analysis of broth-cultured Bacillus atrophaeus and Bacillus cereus spores, J. Appl. Microbiol., 2008, 105, 1604–1613.
E. M. Peck, C. G. Collins, B. D. Smith, Thiosquaraine rotaxanes: synthesis, dynamic structure, and oxygen photosensitization, Org. Lett., 2013, 15, 2762–2765.
Y. Cakmak, S. Kolemen, S. Duman, Y. Dede, Y. Dolen, B. Kilic, Z. Kostereli, L. T. Yildirim, A. L. Dogan, D. Guc, E. U. Akkaya, Designing excited states: theory-guided access to efficient photosensitizers for photodynamic action, Angew. Chem., Int. Ed., 2011, 50, 11937–11941.
D. Dulin, A. Le Gall, K. Perronet, N. Soler, D. Fourmy, S. Yoshizawa, P. Bouyer, N. Westbrook, Reduced photobleaching of BODIPY-FL, Phys. Proc., 2010, 3, 1563–1567.
I. Swiecicka, D. K. Bideshi, B. A. Federici, Novel isolate of Bacillus thuringiensis subsp. thuringiensis that produces a quasicuboidal crystal of Cry1Ab21 toxic to larvae of Trichoplusia ni, Appl. Environ. Microbiol., 2008, 74, 923–930.
A. Magge, B. Setlow, A. E. Cowan, P. Setlow, Analysis of dye binding by and membrane potential in spores of Bacillus species, J. Appl. Microbiol., 2009, 106, 814–824.
J. Moan, Q. Peng, An outline of the hundred-year history of PDT, Anticancer Res., 2003, 23, 3591–3600.
M. R. Hamblin, T. Hasan, Photodynamic therapy: a new antimicrobial approach to infectious disease?, Photochem. Photobiol. Sci., 2004, 3, 436–450.
D. P. Valenzeno, J. P. Pooler, Cell-membrane photo-mondification - relative effects of halogenated fluorosciens for photohemolysis, Photochem. Photobiol., 1982, 35, 343–350.
J. P. Pooler, Photooxidation of cell-membranes using eosin derivatives that locate in lipid or protein to study the role of diffusible intermediates, Photochem. Photobiol., 1989, 50, 55–68.
M. N. Usacheva, M. C. Teichert, M. A. Biel, Comparison of the methylene blue and toluidine blue photobactericidal efficacy against Gram-positive and Gram-negative microorganisms, Lasers Surg. Med., 2001, 29, 165–173.
T. Maisch, C. Bosl, R. M. Szeimies, N. Lehn, C. Abels, Photodynamic effects of novel XF porphyrin derivatives on prokaryotic and eukaryotic cells, Antimicrob. Agents Chemother., 2005, 49, 1542–1552.
Y. Nitzan, M. Gutterman, Z. Malik, B. Ehrenberg, Inactivation of Gram-negative bacteria by photosensitized porphyrins, Photochem. Photobiol., 1992, 55, 89–96.
Z. Q. Xu, M. T. Flavin, J. Flavin, Combating multidrug-resistant Gram-negative bacterial infections, Expert Opin. Invest. Drugs, 2014, 23, 163–182.
M. J. Leggett, G. McDonnell, S. P. Denyer, P. Setlow, J. Y. Maillard, Bacterial spore structures and their protective role in biocide resistance, J. Appl. Microbiol., 2012, 113, 485–498.
J. A. Tufts, M. W. Calfee, S. D. Lee, S. P. Ryan, Bacillus thuringiensis as a surrogate for Bacillus anthracis in aerosol research, World. J. Microbiol. Biotechnol., 2014, 30, 1453–1461.
S. Ghosal, T. J. Leighton, K. E. Wheeler, I. D. Hutcheon, P. K. Weber, Spatially resolved characterization of water and ion incorporation in Bacillus spores, Appl. Environ. Microbiol., 2010, 76, 3275–3282.
J. Errington, Bacillus subtilis sporulation: regulation of gene expression and control of morphogenesis, Microbiol. Rev., 1993, 57, 1–33.
B. Senthil Kumar, Z. Ralte, A. K. Passari, V. K. Mishra, B. M. Chutia, B. P. Singh, G. Guruswami, S. K. Nachimuthu, Characterization of Bacillus thuringiensis Cry1 class proteins in relation to their insecticidal action, Interdiscip. Sci., 2013, 5, 127–135.
G. Pesce, G. Rusciano, A. Sasso, R. Isticato, T. Sirec, E. Ricca, Surface charge and hydrodynamic coefficient measurements of Bacillus subtilis spore by optical tweezers, Colloids Surf., B, 2014, 116, 568–575.
A. Terada, A. Yuasa, T. Kushimoto, S. Tsuneda, A. Katakai, M. Tamada, Bacterial adhesion to and viability on positively charged polymer surfaces, Microbiology, 2006, 152, 3575–3583.
M. Schafer, C. Schmitz, R. Facius, G. Horneck, B. Milow, K. H. Funken, J. Ortner, Systematic study of parameters influencing the action of rose bengal with visible light on bacterial cells: comparison between the biological effect and singlet-oxygen production, Photochem. Photobiol., 2000, 71, 514–523.
T. N. Demidova, M. R. Hamblin, Photodynamic inactivation of Bacillus spores, mediated by phenothiazinium dyes, Appl. Environ. Microbiol., 2005, 71, 6918–6925.
A. Oliveira, A. Almeida, C. M. Carvalho, J. P. Tome, M. A. Faustino, M. G. Neves, A. C. Tome, J. A. Cavaleiro, A. Cunha, Porphyrin derivatives as photosensitizers for the inactivation of Bacillus cereus endospores, J. Appl. Microbiol., 2009, 106, 1986–1995.
P. Setlow, Spores of Bacillus subtilis: their resistance to and killing by radiation, heat and chemicals, J. Appl. Microbiol., 2006, 101, 514–525.
H. Chirakkal, M. O’Rourke, A. Atrih, S. J. Foster, A. Moir, Analysis of spore cortex lytic enzymes and related proteins in Bacillus subtilis endospore germination, Microbiology, 2002, 148, 2383–2392.
P. A. Pinzon-Arango, R. Nagarajan, T. A. Camesano, Effects of L-alanine and inosine germinants on the elasticity of Bacillus anthracis spores, Langmuir, 2010, 26, 6535–6541.
S. Moller-Tank, W. Maury, Phosphatidylserine receptors: enhancers of enveloped virus entry and infection, Virology, 2014, 468–470, 565–580.
J. L. Wanderley, P. E. Thorpe, M. A. Barcinski, L. Soong, Phosphatidylserine exposure on the surface of Leishmania amazonensis amastigotes modulates in vivo infection and dendritic cell function, Parasite Immunol., 2013, 35, 109–119.
D. Magde, R. Wong, P. G. Seybold, Fluorescence quantum yields and their relation to lifetimes of rhodamine 6G and fluorescein in nine solvents: improved absolute standards for quantum yields, Photochem. Photobiol., 2002, 75, 327–334.
Author information
Authors and Affiliations
Corresponding author
Additional information
Electronic supplementary information (ESI) available. See DOI: 10.1039/c5pp00100e
Rights and permissions
About this article
Cite this article
Rice, D.R., Gan, H. & Smith, B.D. Bacterial imaging and photodynamic inactivation using zinc(ii)-dipicolylamine BODIPY conjugates. Photochem Photobiol Sci 14, 1271–1281 (2015). https://doi.org/10.1039/c5pp00100e
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c5pp00100e