Abstract
A large number of human diseases are caused by chronic tissue injury with fibrosis potentially leading to organ failure. There is a need for more effective anti-fibrotic therapies. Congenital muscular dystrophy type 1A (MDC1A) is a devastating form of muscular dystrophy caused by laminin α2 chain-deficiency. It is characterized with early inflammation and build-up of fibrotic lesions, both in patients and MDC1A mouse models (e.g. dy3K/dy3K). Despite the enormous impact of inflammation on tissue remodelling in disease, the inflammatory response in MDC1A has been poorly described. Consequently, a comprehensive understanding of secondary mechanisms (impaired regeneration, enhanced fibrosis) leading to deterioration of muscle phenotype in MDC1A is missing. We have monitored inflammatory processes in dy3K/dy3K muscle and created mice deficient in laminin α2 chain and osteopontin or galectin-3, two pro-inflammatory and pro-fibrotic molecules drastically increased in dystrophic muscle. Surprisingly, deletion of osteopontin worsened the phenotype of dy3K/dy3K mice and loss of galectin-3 did not reduce muscle pathology. Our results indicate that osteopontin could even be a beneficial immunomodulator in MDC1A. This knowledge is essential for the design of future therapeutic interventions for muscular dystrophies that aim at targeting inflammation, especially that osteopontin inhibition has been suggested for Duchenne muscular dystrophy therapy.
Similar content being viewed by others
Introduction
Inflammation is a powerful regulator of both physiological and pathological processes in tissues. Inflammation and fibrosis trigger loss of muscle function in various types of muscular dystrophy. Congenital muscular dystrophy caused by mutations in the laminin α2 chain gene (MDC1A) is one of the most devastating forms of muscular dystrophy (both in humans and mice)1. Clinical symptoms include severe muscle wasting, progressive muscle weakness, joint contractures, respiratory and feeding difficulties and numerous complications. Most patients lose ambulation in childhood, suffer enormous discomfort and have dramatically decreased life-span1. The pathology of laminin α2 chain-deficient muscle is presented with muscle fibre degeneration/regeneration, apoptosis, acute inflammation and subsequent infiltration of connective tissue2,3,4,5,6,7,8,9,10,11,12,13. Despite the tremendous impact of inflammation on tissue remodelling in disease, the inflammatory response in MDC1A has been poorly characterized. Consequently, full understanding of secondary mechanisms (e.g. impaired regeneration, fibrosis) leading to deterioration of muscle phenotype in MDC1A is missing. Several mouse models for the disease exist, among which dy3K/dy3K mice display complete deficiency of laminin α2 chain and adequately mirror the severe phenotype of MDC1A patients2.
Osteopontin is a multifunctional protein, expressed by a variety of cell types in multiple tissues14,15,16,17,18. It plays a major role in several fibrotic disorders19,20,21,22. Importantly, in recent studies the molecule has been attributed the status of a pro-inflammatory cytokine, as it powerfully regulates immune cell activity and fate23,24,25,26,27,28,29. Although osteopontin levels in normal skeletal muscle are very low30,31, pleiotropic roles of the cytokine in injured or diseased muscle have recently become evident. In injured muscle inflammatory cells and myoblasts produce osteopontin32 and its upregulation contributes to both muscle repair and fibrosis30,31,32,33,34,35. The complexity of osteopontin interactions is illustrated by its multidirectional influence on cells that contribute to muscle repair and/or muscle deterioration: the molecule is associated with intricate regulation of inflammation that prompts myogenic cell (myoblast) proliferation and differentiation as well as fibrogenic cell (myofibroblast) differentiation22,32,33,35. Yet, the mechanisms of osteopontin-steered inflammatory events that impact muscle phenotype have not been fully understood. Notably, the protein has been shown to be upregulated in muscles from Duchenne muscular dystrophy patients and in dystrophin-deficient mdx mice31,34, and has been suggested to mediate the progression of dystrophin-deficiency31,36. Consequently, the deletion of osteopontin in mdx mice resulted in reduced fibrosis and improvement of muscle strength, possibly through skewing the macrophage population towards a pro-regenerative phenotype, demonstrating osteopontin’s powerful properties to control macrophage polarization in the dystrophic muscle37. Collectively, these data suggest that inflammation is the link between myogenesis and fibrosis and osteopontin could be the immunomodulator of muscle diseases. It has even been proposed that osteopontin may be a promising therapeutic target for reducing inflammation and fibrosis in Duchenne muscular dystrophy individuals34. Yet, its impact on disease progress in MDC1A has not been demonstrated, although there is a dramatic increase of osteopontin expression in muscle from patients and dyw/dyw mice (animals that express low amounts of truncated laminin α2 chain)31,38.
Galectin-3, a multifunctional β-galactoside-binding animal lectin, is also an important modulator of both acute and chronic inflammation39,40,41. The precise inflammatory role of galectin-3 seems to depend on the type of stimulus and organ damage. However, a majority of studies suggest galectin-3 to be pro-inflammatory during acute tissue injury42 whereas chronic tissue damage and inflammation lead to a shift of galectin-3 function towards wound healing, promoting formation of fibrotic tissue43. Galectin-3 is increased in a number of different fibrotic conditions including muscular dystrophy44. Galectin-3 inhibitors protect against fibrotic disorders45,46 and are currently tested for the treatment of idiopathic pulmonary fibrosis47. The role of galectin-3 in MDC1A has never been investigated.
Hence, in the current study we address in detail the role of pro-inflammatory molecules osteopontin and galectin-3 in MDC1A by generating laminin α2 chain-osteopontin and laminin α2 chain-galectin-3 double knockout mice (dy3K/OPN and dy3K/GAL).
Results
Cytokine profile in dy3K/dy3K dystrophic muscle over the course of the disease
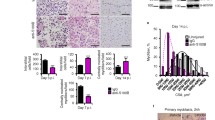
Since inflammation could potentially dictate the muscle disease outcome, it is urgent to characterize the immunological milieu in laminin α2 chain-deficient muscle. To our knowledge, the cytokine expression profile has not been examined in dy3K/dy3K mice over a time course of the disease. Consequently, we aimed to assess cytokine levels in early pathology (1-week-old), intermediate disease stage (2-week-old) and late pathology (3-week-old) of dy3K/dy3K mice in comparison to age-matched wild-type animals. Cytokine analysis revealed common pattern of robust cytokine peak in 2-week-old dystrophic muscle and subsequent drop of cytokine levels, both pro- and anti-inflammatory, at 3 weeks of age (Fig. 1a), suggesting a time-dependent association of the muscle inflammation. Such mechanism of cumulated, intensified inflammatory response with overlap of pro- and anti-inflammatory signals is different from inflammation characteristics in healing normal muscle and resembles the inflammatory status in muscle with impaired regeneration48. We did not observe this general pattern when analysing the same cytokines in circulation. Rather, serum levels of interleukin-1β, interleukin-4, interleukin-6, TNFα, interleukin-10 and IFNγ were unaffected by laminin α2-deficiency and only circulating levels of interleukin-2, interleukin-5 and interleukin-12 were increased (Fig. 1b).
(a) Levels of IFNγ, interleukin-1β, interleukin-4, interleukin-6, interleukin-10, TNFα and CXCL1 are increased in 2-week-old and/or 3-week-old dy3K/dy3K muscle. Concentrations are presented as pg cytokine/mg protein. The same number of mice as indicated in the first graph was included in all analyses. (b) In serum, only interleukin-2, interleukin-5 and interleukin-12 levels are increased in dy3K/dy3K mice (top row, depicted in red). Non-altered cytokines are presented in blue (middle and bottom rows). Concentrations are presented as pg cytokine/ml serum. The number of mice included at each time point is indicated in the graphs. Stars indicate significant difference between dystrophic and age-matched wild-type mice (Kruskal-Wallis test followed by Dunn’s test).
Both osteopontin and galectin-3 have been shown to influence and/or to be influenced by the numerous cytokines studied above. For example, IFNγ, interleukin-1β, TNFα and interleukin-10 are involved in osteopontin and galectin-3 regulation in different tissues49,50,51,52,53,54,55,56. Furthermore, these cytokines contribute either to muscle repair and/or to the pathology of muscular dystrophy11,57,58,59,60,61,62. Additionally, augmented expression of osteopontin has been shown to promote disease progression and fibrosis in the mdx mouse by stimulation of pro-fibrotic macrophage population34,37. Finally, osteopontin and galectin-3 are massively upregulated in skeletal muscles from MDC1A patients31 as well as in the dyW/dyW mouse model for the disease38,44. In the light of these results published by others and in the context of our mesoscale data (Fig. 1), we postulated that osteopontin and galectin-3 could also be disease modulators in MDC1A. In order to determine if osteopontin and galectin-3 deletion prevent muscle wasting in mice with MDC1A, we generated laminin α2 chain-deficient animals that lack osteopontin (dy3K/OPN) or galectin-3 (dy3K/GAL). Immunostaining with antibodies against osteopontin and galectin-3 confirmed dramatically increased levels in dy3K/dy3K muscle (Fig. 2).
(a) Longitudinal hind-limb paraffin sections from 13-day-old (intermediate stage of the disease) and 21-day-old (late pathology) dy3K/dy3K mice (P13 and P21, respectively); 21-day-old wild-type mice (WT, P21), osteopontin knockout mice (OPN ko, P21) and double knockout mice (dy3K/OPN, P21) were probed with an anti-osteopontin antibody. Osteopontin is absent from wild-type muscle, but the cytokine expression is highly increased throughout the disease course in dy3K/dy3K muscle. Multiple muscles from the whole limb were analysed. As expected, osteopontin is not present in osteopontin-deficient muscles (OPN ko and dy3K/OPN). (b) Transverse cryosections of quadriceps muscle from 3-week-old WT, galectin-3 knockout (GAL ko), dy3K/dy3K, and double knockout (dy3K/GAL) mice stained for galectin-3. Galectin-3 is only expressed in dy3K/dy3K muscle. Bars: 50 μm.
Deletion of osteopontin or galectin-3 does not improve the overall health of dy3K/dy3K mice
We have analysed the phenotype of all animal genotypes at the age of 1, 2 and 3-weeks, with particular emphasis on week 3 (corresponding to late pathology), when the readout of a mouse condition is the most complete. Neither osteopontin nor galectin-3 single knockout mice (OPN ko and GAL ko, respectively) display any overt phenotype (Fig. 3 and Supplementary material, Video S1)15,63. Initial observations of 2-3-week-old dy3K/dy3Kand both double knockout mice strains revealed muscular dystrophy symptoms, such as: decreased size, waggling gait, reduced eagerness to move, tremor and hind-limb lameness, muscle wasting and gradual worsening of general health (Fig. 3a,e and Supplementary material, Video S2 and S3). Detailed analysis of the outward phenotype of 3-week-old animals confirmed no improvement of the overall condition of dy3K/dy3K mice upon osteopontin or galectin-3 deletion. Both dy3K/OPN and dy3K/GAL animals showed greatly reduced weight compared with healthy controls, but there was no significant difference in body mass between dy3K/dy3Kand dy3K/OPN or dy3K/GAL mice, respectively (Fig. 3c and f). What is more, dy3K/OPN mice had a significantly shorter life span than single knockout dy3K/dy3K animals (Fig. 3b) (median survival 21 and 24 days, respectively). In conclusion, deletion of osteopontin or galectin-3 was not at all beneficial for the general condition of dy3K/dy3Kmice.
(a) Outward phenotype of 3-week-old single and double knockout mice. Decreased size, emaciation and severe muscle wasting is evident in dy3K/dy3K and dy3K/OPN animals. OPN ko littermates are normal. (b) Dy3K/dy3K and dy3K/OPN survival curves show decreased life span of animals that lack osteopontin (p = 0.0141, log-rank Cox-Mantel test). N = 21 and n = 23, respectively. (c) Body weight analysis of 3-week-old males reveals no significant differences between dy3K/dy3K and dy3K/OPN mice (p > 0.9999). Both are significantly smaller than wild-type and osteopontin knockout littermates (p = 0.0003, p = 0.0009; p = 0.0002, p = 0.0006; respectively, Kruskal-Wallis, followed by Dunn’s test). (d) Activity of dy3K/dy3K and dy3K/OPN mice is equally compromised as shown by number of stand ups (left) and walking time measurement (right). Dy3K/dy3K and dy3K/OPN animals were significantly less active than normal littermates, both in the walking test (p = 0.0037 and p = 0.0292, respectively; one-way ANOVA followed by Sidak’s test) and in stand-up test (p < 0.0001, one-way ANOVA followed by Sidak’s test). Both dystrophic mouse strains were equally inactive (p > 0.9999 and p = 0.9193, number of stand ups and walking time, respectively; one-way ANOVA followed by Sidak’s test). (e) Double knockout (dy3K/GAL) mice are presented with the same outward muscular dystrophy features as single knockout dy3K/dy3K animals. (f) Body weight of wild-type (WT), galectin-3 knockout (GAL ko), dy3K/dy3K, and double knockout (dy3K/GAL) mice were recorded at 3 weeks of age. Both dy3K/dy3K and dy3K/GAL mice weigh significantly less compared with WT (p = 0.0021 and p = 0.0188, respectively; Kruskal-Wallis test followed by Dunn’s test). (g) Removal of galectin-3 does not increase muscle strength in dy3K/dy3K mice, as demonstrated by grip strength test (forelimbs) (p = 0.3429, Mann-Whitney).
Muscle function is not enhanced upon osteopontin or galectin-3 deletion in dy3K/dy3K mice
To compare muscle function in dy3K/dy3K single knockout and dy3K/OPN double knockout mice, we carried out two activity tests on 3-week-old animals (including wild-type and osteopontin-null mice): vertical activity test (number of stand ups during 5 minutes) and horizontal activity test (5 minutes locomotion test when exploring a new cage) (Fig. 3d). We found that dy3K/dy3K and dy3K/OPN animals were significantly less active than wild-type mice (Fig. 3d, left and right panel, respectively). Moreover, double knockout mice did not show any improvement of motor activity compared to dy3K/dy3K individuals (Fig. 3d). Even if both dy3K/dy3K and dy3K/OPN animals were to some extent able to explore the new cage (amount of time spent moving), the quality of movement clearly pointed towards impaired muscle function: mice were moving slowly, waddling or wobbling and often tripping (Supplementary Video 2 and 3). To assess muscle strength in dy3K/dy3K and dy3K/GAL mice we used a grip strength meter. Calculation of normalized strength did not reveal any significant difference between dy3K/dy3K and dy3K/GAL mice (Fig. 3g). Thus, muscle function is not improved upon osteopontin or galectin-3 deletion in laminin α2 chain-deficient mice.
Muscle phenotype is exacerbated upon osteopontin deletion in 2-week-old dy3K/dy3K mice
Severe muscle wasting and loss of muscle function in laminin α2 chain-deficiency is a consequence of muscle fibre damage, muscle cell death, robust inflammatory response and subsequent build-up of fibrotic tissue2. Muscle degeneration, followed by inflammation and tissue regeneration (manifested by a presence of small centrally nucleated muscle fibres) are the disease features that occur earliest and are prominent at week 1 and even more so at week 2 in dy3K/dy3K mice. Thus, we have analysed muscle morphology in single and double knockout mice at these ages. Severe muscle damage and infiltration of mononuclear cells were detected in 1-week-old muscles from both dy3K/dy3K and dy3K/OPN mice (Fig. 4a). Interestingly, muscles from 2-week-old dy3K/OPN mice seemed to be more affected than muscles from dy3K/dy3K single knockouts (especially triceps) (Fig. 4b). They displayed larger areas of deteriorating fibres that were extensively invaded by inflammatory cells (more detailed inflammation analysis from 2-week-old mice is described a few paragraphs below). These observations pointed out that osteopontin deletion could have a negative influence on muscle condition in laminin α2 chain-deficiency. Yet, the weight of 2-week-old double knockout mice was not decreased compared to single knockout dy3K/dy3K mice (Fig. 4c).
(a) Hematoxylin and eosin staining of 1-week-old dy3K/dy3K and dy3K/OPN transverse cryosections shows extensive infiltrates of mononuclear cells and disruption of muscle fascicle integrity. (b) Muscles from 2-week-old double knockout mice tend to be more affected than muscles from age-matched dy3K/dy3K mice. Muscle damage and inflammation is intensified in both genotypes at this stage. Osteopontin knockout mice show normal muscles. Five mice from each genotype where analysed. (c) There is no difference in body weight between 2-week-old dy3K/dy3K and dy3K/OPN mice (p = 0.9954, Mann-Whitney). Bars: 40 μm.
Muscles from 3-week-old double knockout animals show similar dystrophic features as muscles from dy3K/dy3K single knockout animals
All muscular dystrophy hallmarks (degeneration/regeneration cycles, inflammatory response, fibrosis) were clearly visible in various 3-week-old dy3K/dy3K, dy3K/OPN and dy3K/GAL muscles, but not in osteopontin or galectin-3 knockout mice (Fig. 5a and b). However, no obvious difference between dy3K/dy3K and double knockout dystrophic strains was detected at week 3, despite observations that muscle morphology was aggravated in 2-week-old dy3K/OPN mice.
(a) Hematoxylin and eosin staining of quadriceps, triceps and diaphragm cryosections from 3-week-old wild-type (WT), osteopontin knockout (OPN ko), dy3K/dy3K and dy3K/OPN animals. Osteopontin-null muscles display normal morphology, whereas dy3K/dy3K and dy3K/OPN muscles share dystrophic features of MDC1A: disrupted integrity of muscle fascicles, muscle fibre degeneration/regeneration, infiltration of mononuclear cells, and presence of fibrotic lesions. (b) Transverse paraffin sections of quadriceps muscle from 3-week-old wild-type (WT), galectin-3 knockout (GAL ko), dy3K/dy3K, and double knockout (dy3K/GAL) mice stained with hematoxylin and eosin. Deletion of galectin-3 in dy3K/dy3K mice does not reduce muscle pathology. Galectin-3 knockout muscle does not display any muscle defects. Bars: 50 μm. (c) Osteopontin absence negatively impacts muscle regeneration in laminin α2 chain-deficiency (left panel). The number of regenerating fibres was significantly decreased in triceps from 3-week-old dy3K/OPN animals compared with dy3K/dy3K mice (p = 0.0205, Mann-Whitney). Removal of galectin-3 does not impact muscle regeneration in laminin α2 chain-deficiency (right panel) (p = 0.6286, Mann-Whitney). (d) Embryonic myosin heavy chain staining (green) reveals low numbers of newly regenerating fibres in 3-week-old dy3K/dy3K and dy3K/OPN triceps. Collagen IV immunostaining (red) and DAPI labelling (blue) depict muscle fibres and nuclei, respectively. At least five mice from each group were analysed. Bar: 50 μm.
Notably, despite its role in muscle pathology progress34,37, osteopontin has been shown to promote muscle repair30,33. Consequently, we assessed the impact of osteopontin deletion on muscle regeneration in 3-week-old double knockout animals. Quantification of centrally nucleated fibres (represents overall regeneration) revealed a decrease in regeneration capacity in double knockout muscle compared with dy3K/dy3K muscle (Fig. 5c). We also analysed whether dy3K/dy3K and dy3K/OPN animals were able to form newly regenerating fibres at 3 weeks of age. Embryonic myosin heavy chain immunostaining revealed very few newly regenerating fibres at the terminal stage of the disease in both dystrophic mouse models, especially in dy3K/OPN mice, which showed only spread single positive fibres, whereas dy3K/dy3K mice showed occasional clusters of regenerating fibres (Fig. 5d). These results further confirm a role for osteopontin in muscle regeneration.
Conversely, deletion of galectin-3 in dy3K/dy3K mice did not affect myofibre regeneration (Fig. 5c, right graph).
Fibrotic lesions are not reduced upon osteopontin or galectin-3 deletion from laminin α2 chain-deficient muscle
Osteopontin promotes formation of collagen fibrils in injured tissue15. Fibrosis inhibition upon osteopontin deletion in mdx mice was the major consequence of this genetic manipulation34. We investigated whether osteopontin ablation prevents the build-up of connective tissue in dy3K/dy3K mouse model despite its negative impact on muscle repair. Sirius red staining (that visualizes collagens) and immunostaining towards collagen III and fibronectin showed abundant fibrotic lesions in dy3K/dy3K and dy3K/OPN muscles (Fig. 6a). What is more, quantification of hydroxyproline in quadriceps and triceps showed increased collagen content in dy3K/OPN limb muscles compared with dy3K/dy3K muscles (Fig. 6b). However, such upregulation of collagen was not evident in dy3K/OPN diaphragm (Fig. 6b). This data indicates that slightly impaired regeneration in dy3K/OPN limb muscle (Fig. 5c) together with increased inflammatory response in 2-week-old mice (Fig. 4b, see also below) possibly result in augmented fibrosis.
(a) Sirius red staining (visualizes collagens) of 3-week-old quadriceps and immunolabelling for collagen III (green) and fibronectin (red) in 3-week-old triceps reveal no fibrosis prevention upon osteopontin deletion in laminin α2 chain-deficient muscular dystrophy. Collagen III and fibronectin are clearly upregulated in dy3K/dy3K and dy3K/OPN muscles compared with wild-type and osteopontin-null muscles. Bars: 50 μm. (b) Quantification of hydroxyproline content in quadriceps, triceps and diaphragm muscle from dy3K/dy3K and dy3K/OPN animals shows significant increase of collagen deposition in double knockout limb muscles (p = 0.0175 and p = 0.0262, respectively), but not in diaphragm (p = 0.7104, Mann-Whitney). (c) qPCR analysis of normal (wild-type, osteopontin knockout) and dystrophic (dy3K/dy3K and dy3K/OPN) quadriceps revealed no impact on TGF-β transcript levels upon osteopontin deletion in laminin α2 chain-deficient muscle (p > 0.9999, Kruskal-Wallis, followed by Dunn’s test). (d) Fibrosis is equally severe in dy3K/dy3K and dy3K/GAL muscles. Quantification of hydroxyproline content in quadriceps muscle shows no significant difference between single and double knockout mice (p = 0.5476, Mann-Whitney).
Since fibrosis reduction in mdx/OPN muscle was associated with decreased TGF-β gene expression, we analysed whether fibrosis enhancement upon osteopontin deletion in laminin α2 chain-deficiency is a consequence of a TGF-β-related mechanism. TGF-β has been shown to be upregulated in MDC1A13,64,65. The analysis of TGF-β gene expression revealed no reduction of TGF-β transcript levels upon osteopontin deletion in dy3K/dy3K muscle (Fig. 6c). Taken together, loss of osteopontin has an opposite effect on fibrotic tissue build-up in dy3K/dy3K mice compared to mdx mice.
Mounting evidence demonstrates that galectin-3 plays a pro-fibrotic role in organ fibrogenesis and galectin-3 inhibitors can protect against fibrotic disorders46,66. However, deletion of galectin-3 did not reduce fibrosis in dy3K/GAL muscle, as revealed by Sirius red staining (Supplementary Fig. S1) and hydroxyproline assay (Fig. 6d).
Inflammatory response in dy3K/dy3K dystrophic muscle is moderately influenced by osteopontin ablation
Both osteopontin and galectin-3 are potent regulators of inflammatory cells. In particular, they modulate the activity of macrophages and neutrophils23,26,37,67,68,69,70, major inflammatory cell types in mdx muscle34. In MDC1A, macrophages have been suggested to constitute a major population of inflammatory cells10,11,71. We investigated the contribution of neutrophils to inflammation in dy3K/dy3K muscle. Ly6G staining (depicting neutrophils) was clearly less pronounced than CD11b staining (labelling both macrophages and neutrophils), suggesting that neutrophils are not as numerous as macrophages in 3-week-old dy3K/dy3K muscle (Supplementary Fig. S2). Since neutrophils were shown to be reduced in mdx/OPN mice34, we inspected the impact of osteopontin deletion on neutrophils in laminin α2 chain-deficiency. In 1-week-old dy3K/dy3K and dy3K/OPN mice, at the onset of inflammation, muscles were mildly invaded by neutrophils (Fig. 7a, top panel). However, occasional large clusters of neutrophils were present in some (but not all) analysed muscles among both dy3K/dy3K and dy3K/OPN individuals (Fig. 7a, bottom panel). Consistent with data shown in Supplementary Fig. S2, some neutrophils were observed in dy3K/dy3K muscle as the disease progressed (3-week-old mice) (Fig. 7b). Almost no neutrophils were detected in 3-week-old dy3K/OPN muscles (Fig. 7b), suggesting that osteopontin deletion influences neutrophil population in dy3K/dy3K muscle. This data is in line with findings in mdx mice deficient in osteopontin34.
Osteopontin deletion reduces neutrophil numbers in double knockout muscle. (a) One-week-old muscles display the same pattern of neutrophil distribution in dy3K/dy3K and dy3K/OPN mice (Ly6G, red staining). Triceps and quadriceps (cryosections) were analysed and both muscles showed similar characteristics of neutrophil infiltration. Either none or very few neutrophils are present (top panel, triple staining, triceps is shown), or they are occasionally more pronounced focally (bottom panel, single labelling, quadriceps is shown). No neutrophils were detected in wild-type and osteopontin-null muscles. (b) Labelling against Ly6G (red) in 3-week-old muscles (triceps and quadriceps) showed almost none or very few neutrophils in double knockout mice. A unique area with relatively many neutrophils in dy3K/OPN triceps is presented (top panel), which is still less affected than an average dy3K/dy3K site. A representative picture of dy3K/OPN quadriceps with very few positive cells is shown (bottom panel). Five mice from each group were analysed in (a) and (b). Sections were co-stained with collagen IV antibody (green) and DAPI (blue) to depict muscle fibres and nuclei, respectively. Ly6G immunolabelling in spleen and mdx muscle is shown as positive control. Bars: 50 μm.
We next analysed the degree of general inflammatory response in muscle (staining against CD11b: marker for monocytes, macrophages, granulocytes and natural killer cells) from 1, 2 and 3-week-old mice (Supplementary Fig. S3, Fig. 8 and Supplementary Fig. S4). Inflammatory cells invade the laminin α2 chain-deficient muscle already at the age of 1 week10 (Supplementary Fig. S3a), increase in numbers at 2 weeks of age (Fig. 8a) and remain pronounced at the terminal stage of the disease (week 3) (Fig. 8b). Infiltrates of CD11b positive inflammatory cells were also clearly evident in 1-week-old, 2-week-old and 3-week-old dy3K/OPN muscles, as well as in 3-week-old dy3K/GAL muscles (Supplementary Fig. S3a, Fig. 8 and Supplementary Fig. S4). We subsequently quantified the area covered by inflammatory cell infiltrates at those stages of the disease in dy3K/dy3K and dy3K/OPN muscles. No significant difference between single and double knockout animals was found for either 1-week-old triceps (Supplementary Fig. S3a’) or 3-week-old quadriceps and triceps (Fig. 8b’). However, as concluded from muscle histology analysis (Fig. 4), we detected a transient increase of inflammatory response in 2-week-old dy3K/OPN muscles (Fig. 8a’). Similar to osteopontin removal, deletion of galectin-3 in dy3K/dy3K muscle did not seem to influence the general inflammatory response in 3-week-old animals (Supplementary Fig. S4).
(a) Muscles from 2-week-old dy3K/OPN mice are more inflamed (especially triceps, bottom panel), as demonstrated by immunostaining against CD11b. (a’) Quantification of CD11b-stained muscle area in quadriceps and triceps confirms a strong trend for increased inflammation in limb muscles from double knockout mice (p = 0,0952 and p = 0.0079, for quadriceps and triceps, respectively; Mann-Whitney). (b) Inflammation is sustained in 3-week-old dy3K/dy3K and dy3K/OPN mice, to the same degree in both dystrophic groups. (b’) Quantification of CD11b immunolabelling in both quadriceps and triceps reveals no significant difference between 3-week-old dy3K/dy3K single knockout and double knockout mice (p = 0.0952 and p = 0.9004, respectively; Mann-Whitney). For both (a) and (b) a representative area of CD11b-stained quadriceps (red staining, top panel) and a whole CD11b-labelled triceps are shown (white staining, bottom panel). No inflammation was detected in wild-type and osteopontin-null muscles. Five mice from each group were analysed. Bars: 50 μm and 100 μm.
Although the numbers of macrophages were not affected upon osteopontin deletion in the mdx mouse model34, macrophage population was shifted toward pro-regenerative phenotype37. Thus, we have studied whether osteopontin removal has the same impact on macrophages in MDC1A (in different inflammatory milieu). We have in fact detected increased numbers of monocytes/macrophages in 2-week-old dy3K/OPN muscle compared to dy3K/dy3K muscle (CD68 immunostaining) (Fig. 9a and a’). In agreement with CD11b staining, this difference in macrophage numbers was not maintained in 3-week-old double knockout mice (Fig. 9b and b’, left graph).
(a) CD68 immunostaining of 2-week-old dy3K/dy3K and dy3K/OPN triceps muscle reveals increased infiltration of macrophages/monocytes in double knockout muscle. Representative areas of triceps and whole triceps are shown. (a’) Quantification of CD68-stained muscle area confirms significant escalation of macrophage numbers in 2-week-old double knockout mice compared to dy3K/dy3K single knockout mice (p = 0.0317, Mann Whitney) (b) Increase in macrophage numbers in younger double knockout mice is only transient. Three-week-old triceps muscle from both dy3K/dy3K and dy3K/OPN mice show the same appearance when stained against CD68 (top panel, green staining). M1 macrophages are present in both dy3K/dy3K and dy3K/OPN triceps, as demonstrated by immunostaining against iNOS (red, middle panel) and CD68 (merged staining, bottom panel). iNOS is expressed at low levels in vessels in wild-type and osteopontin-deficient muscle. (b’) Quantification of CD68 and iNOS labelling confirms no change of M1 macrophage-mediated immune response upon osteopontin deletion in 3-week-old mice (p = 0.2222 and p = 0.6905, respectively; Mann-Whitney). Five mice from each group were analysed in (a) and (b). Bars: 50 and 100 μm.
Since M1 macrophages were reduced in mdx/OPN mice, we further analysed whether osteopontin deletion influenced the M1 macrophage population. For that purpose we stained 3-week-old muscle sections for iNOS, which is highly produced by M1 pro-inflammatory macrophages in response to an inflammatory stimulus. Since iNOS can also be expressed by endothelial cells and vascular smooth muscle cells, we used CD68 antibody to depict macrophages that produce iNOS. CD68/iNOS positive cells were abundant both in dy3K/dy3K and dy3K/OPN muscles (Fig. 9b), showing no depletion of M1 macrophages in double knockout muscles. This was further confirmed by quantification of iNOS staining (Fig. 9b’, right graph).
These data indicate that deletion of galectin-3 or osteopontin from laminin α2 chain-deficient muscle does not, or only mildly influence the general character of inflammatory response to tissue damage. Additionally, inflammation analyses from dy3K/dy3K muscle lacking osteopontin only partially match the results obtained from mdx mice deficient in osteopontin34,37.
Osteopontin deletion does not improve the phenotype of milder mouse model for laminin α2 chain-deficiency (dy2J/dy2J)
We also investigated the condition of dy2J/OPN mice (dy2J/dy2Janimals express low levels of truncated laminin α2 chain) to exclude the possibility that the general severity of the dy3K/dy3K phenotype is the cause of no health improvement upon osteopontin ablation. Analysis of muscle histology and immunostainings against collagen III and CD11b (macrophages) revealed no differences between dy2J/dy2J and dy2J/OPN muscle phenotype (Supplementary Fig. S5). Additionally, dy2J/OPN mice showed equal impairment of force generation as dy2J/dy2J littermates in grip strength testing (Supplementary Fig. S5, graph).
Discussion
The scientific community still struggles to find cure for muscular dystrophy, although great strides towards understanding of the disease pathogenesis have been made. Since genetic interventions that aim at restoration of a mutated gene have encountered numerous obstacles, prevention of secondary defects that accompany a protein loss has become an attractive target for therapy of muscular dystrophy. Development of anti-apoptotic, anti-fibrotic, anti-atrophic, and pro-regenerative treatments has been extensively explored2,72,73,74,75,76. Additionally, anti-inflammatory approaches have also yielded attention34,61,75,76,77, as inflammation could be a potent modulator of the disease outcome. Yet, we only begin to understand molecular interactions in inflammatory milieu of dystrophic muscle.
In the present study, we examined the role of two pro-inflammatory and pro-fibrotic molecules, osteopontin and galectin-3, in MDC1A. Osteopontin or galectin-3 deletion in dy3K/dy3K mouse model for MDC1A did not positively influence the mouse phenotype at any time point of the disease course. Quite in contrary, removal of osteopontin even worsened the condition of dy3K/dy3K animals. These results differ from data obtained from mdx mice that lack osteopontin, although similar level of osteopontin up-regulation was detected in both dystrophin- and laminin α2 chain-deficient mouse models34,38. There might be a few reasons why dy3K/dy3K mice responded to this genetic manipulation in a diverse manner. First, inflammatory milieu in various types of muscular dystrophy is unique: different subset of inflammatory cells and molecules involved may create a distinctive molecular signature that steers the disease progress. In muscle from Duchenne muscular dystrophy patients and from mdx mice a chronic inflammatory state is present78,79,80. The nature of inflammation in MDC1A muscles is somewhat different. The inflammatory response both in patients and in dy3K/dy3K mice takes place at the early stage of disease, being one of the first features of muscle pathology1,9,10,11,81. However, the inflammatory response in patients is only transient1. Yet, detailed characterization of inflammation in MDC1A is still missing. In particular, extending our knowledge about macrophage populations and behaviour in MDC1A could be vital, since they appear to be significant for the condition of dystrophic muscle (at least in dystrophin-deficiency)37,58. Our study shows that osteopontin deletion in laminin α2 chain-deficiency did not reduce the population of M1 macrophages and thus did not contribute to change in proportions between proinflammatory/proregenerative macrophages - a shift that improved the phenotype of the mdx/OPN mouse37. This could explain why ablation of the osteopontin gene in dy3K/dy3K mice did not result in a similar amelioration of the phenotype. The observed decrease of neutrophil numbers upon osteopontin deletion was clearly not sufficient to reverse the pathology of dy3K/dy3K mice. Additionally, a temporary acute increase of inflammatory response in 2-week-old dy3K/OPN mice could definitely have a tremendous impact on phenotype worsening in older mice (week 3).
Therefore, it is possible that osteopontin in MDC1A inflammatory milieu is in fact beneficial for muscle condition as well as for overall health, as its absence caused further decline in survival and deterioration of already severe muscle phenotype in dy3K/dy3K mice. Two crucial osteopontin functions in MDC1A are unravelled by our study: 1) osteopontin is involved in muscle regeneration 2) osteopontin does not play a key role as a pro-fibrotic molecule in laminin α2 chain-deficient skeletal muscle. Contrary, it could have an anti-fibrotic effect in MDC1A. Yet, the mechanism of its action is not entirely clear, although it does not seem to involve TGF-β signalling. Osteopontin plays a major role in several fibrotic diseases in different tissues19,20,21. It promotes formation of collagen fibrils15, but most likely other molecules compensated for osteopontin absence in dy3K/dy3K mice and stimulated collagen production in limb muscles to even higher degree. It might also be that increased inflammation in dy3K/OPN muscle at the early stage of the disease contributes to compromised tissue repair and augmented fibrosis. Otherwise, impaired regeneration due to osteopontin absence could be the primary cause of inflammation boost and subsequent build-up of fibrotic lesions. Considering that osteopontin has a multidirectional impact on myoblasts, myofibroblasts and inflammatory cells, a link between osteopontin, these cells and muscle regeneration and fibrosis needs to be established. So far an association between regulatory T cells and muscle repair has been clearly demonstrated82 and osteopontin seems to influence regulatory T cell numbers34.
Alternatively, we postulate that the severity of laminin α2-chain-deficiency could lie behind the failure of disease amelioration upon osteopontin deletion in two mouse models (dy3K/dy3K mouse in particular). It is clearly in contrast to the mdx animals, which show a mild phenotype (milder than moderately affected dy2J/dy2J mice) with a lifespan that is close to normal83,84. In fact, a large number of the therapeutic approaches in mdx mice have been successful, ranging from genetic manipulations that aim at dystrophin restoration, through pharmacological approaches targeting secondary defects of the disease, to even protein therapy73,76,85,86. When similar therapeutic approaches are tested in the dy3K/dy3K mouse or in other mouse models for laminin-deficiency, they commonly fail or result in only a modest improvement of the phenotype2,87,88. Nevertheless, our current results and a wide range of previous studies, strongly suggest that for convincing preclinical analysis the choice of animal models that adequately mirror human condition is vital. Another issue to consider is that we developed constitutive double knockout models. We cannot be entirely certain whether gene inactivation of osteopontin and galectin-3 at specific time points would modulate the dy3K/dy3K phenotype differently.
In summary, our results emphasize that osteopontin may exert different biological functions in different types of muscular dystrophy. This knowledge needs to be considered when designing new therapeutic interventions for muscular dystrophies that aim at modifying inflammatory molecules. Our data question whether targeting osteopontin should be considered as a valid therapeutic approach for MDC1A.
The role of galectin-3 in muscular dystrophy has never been investigated previously. Similar to osteopontin, galectin-3 is a modulator of inflammation and capable of promoting fibrogenesis in different organs and currently galectin-3 inhibition is tested clinically. We now demonstrate that deletion of galectin-3 does not reduce fibrotic scarring in dy3K/dy3K muscle, suggesting that during chronic muscle damage, galectin-3 may not be a major pro-fibrotic component. Clearly, the function of galectin-3 is dependent on type of injury and the context of organ damage and it cannot be ruled out that modulating galectin-3 expression in other types of muscular dystrophy could be beneficial. Also, it should be noted that galectin-1 protein therapy in mdx mice resulted in reduced muscle damage, partly by stabilizing the muscle cell membrane via upregulation of integrin α7β1 and utrophin89. This approach remains to be tested in muscular dystrophy caused by laminin α2 chain-deficiency.
Since the molecular signature of inflammation is evidently complex, modulating inflammation in different types of muscular dystrophy should be carefully designed and probably optimized individually for each patient. Further studies will be essential to verify whether therapies aiming at inhibition of inflammation are beneficial for muscular dystrophy patients. We also possibly need to answer a broader question: how efficient is it to target the secondary defects of muscle disease?
Materials and Methods
Animal models
Laminin α2 chain-deficient dy3K/dy3K, dy2J/dy2J 90,91, osteopontin-null (OPN ko) and galectin-3 knockout mice (GAL ko) were previously described4,63,92. Double knockout animals (dy3K/OPN or dy3K/GAL) and respective control mice (OPN ko, GAL ko and wild-type (WT) were generated by breeding dy3K/+; OPN/+ or dy3K/+; GAL/+ males and females. All experimental procedures involving animals were approved by the Malmö/Lund (Sweden) Ethical Committee for Animal Research (the ethical permit number: M15-12 and M152-14) in accordance with the guidelines issued by the Swedish Board of Agriculture.
Activity test
Locomotion features of 3-week-old mice were assessed by measuring the walking time and number of stand-ups within 5 first minutes of presence in a new cage93. Animals were tested one by one, each in a new empty cage without food. Time was measured when the mouse was active and timer was paused each time the mouse rested. The number of full stand ups was counted at the same time. Mice had not been trained prior to the test.
Grip strength
Grip strength measurements (forelimbs) were performed on 3-week-old mice dy3K strain) or 8-9-week-old animals (dy2J strain) as described before94. Briefly, each mouse was allowed to grasp a horizontal metal bar attached to a grip strength meter (Columbus Instruments, Columbus, OH). Upon a good grip (symmetric with both paws) the mouse was gently pulled away until its grasp broke. The test was repeated 5 times for each mouse and the mean value of each session was calculated for analysis. Body weight was recorded in parallel. Animals were not subjected to any training prior to the experiment. Results are presented as normalized strength (force (g)/body weight (g)).
Animal handling and tissue collection
One-week-old, 2-week-old and 3-week-old control mice, single knockouts and double knockouts were euthanized by cervical dislocation. Dy2J/dy2J and dy2J/OPN mice were sacrificed at week 8. Quadriceps, triceps and diaphragm muscles were dissected for hydroxyproline assay, RNA extraction or immunohistochemistry (embedded in O.C.T compound or in paraffin). Cross sections (7 μm thick for frozen tissues or 5 μm for paraffin embedded tissues) were cut using cryostat (Microm HM 560, Cellab Nordia AB) and microtome (Microm H355, Cellab), respectively.
Histology and morphometric analysis
Muscle sections were stained with hematoxylin and eosin10 or picrosirius red/fast green6. Stained cross-sections were scanned using Aperio’s Scanscope CS2. Centrally nucleated muscle fibres and peripherally nucleated normal muscle cells were counted using ImageJ software version 143u. A whole area of each muscle cross section was considered.
Immunofluorescence
Cryosections were subjected to immunofluorescence labelling with antibodies against: embryonic myosin heavy chain (mouse monoclonal F1.652, 1:10, Developmental Studies Hybridoma Bank, University of Iowa), CD11b (rat monoclonal M1/70, 1:250, BD Pharmingen), Ly6G (rat monoclonal 1A8, 1:200, BD Pharmingen), CD68 (rat monoclonal FA-11, 1:100, AbD Serotec), iNOS (rabbit polyclonal, 1:100, Abcam), collagen III (goat polyclonal, 1:100, Southern Biotech), fibronectin (rabbit polyclonal, 1:1000, Abcam), collagen IV (rabbit polyclonal, 1:200, Millipore) and galectin-3 (rat monoclonal, 1:100).
Paraffin sections were used for staining with osteopontin antibody (rabbit polyclonal AB10910, 1:50, Millipore). Samples were deparaffinized and treated with protease XXIV (20 μg/ml in PBS, Subtilisin Carlsberg, Sigma) for 20 min at RT prior to blocking and staining.
Primary antibodies were detected with relevant secondary antibodies (Molecular Probes). Images were taken using Openlab 4 software. The area corresponding to CD11b, CD68 and iNOS labelling was quantified in relation to the entire area of muscle cross-sections. Pictures covering the whole muscle section were stitched using Photoshop software. In order to avoid false positive signal, artefacts and non-muscle tissue (if present) were removed from the images for the quantification process. Otherwise no modifications were applied to the images. Photos were captured at the same exposure times. At least 5 mice were analysed for each immunostaining. ImageJ software version 143u was used for quantification.
Mesoscale
Whole blood (from heart puncture) and quadriceps muscles were collected from 1-week, 2-week and 3-week-old wild-type and dy3K/dy3K mice. Blood samples were processed according to the manufacturer’s specifications (Microvette CB 300, Sarstedt). Muscles were homogenized in Tris Lysis Buffer with proteinase inhibitor tablet (Pierce) using Tissue Lyser (Qiagen). Protein concentration in muscle samples was assessed using Bradford assay. Cytokine and chemokine concentrations were determined by multiplex technology in quadriceps samples and serum (25 μl/sample) using a MSD Mouse Pro-inflammatory V-Plex Plus Kit (IFNγ, interleukin-1β, interleukin-2, interleukin-4, interleukin-5, interleukin-6, interleukin-10, interleukin-12p70, CXCL1, TNFα; K15012C, Mesoscale Discovery) and a SECTOR Imager 6000 (Mesoscale Discovery, Rockville, MD) plate reader according to the manufacturer’s instructions. Data were analysed using MSD Discovery Workbench software. For the muscle homogenate samples the concentrations were normalized to the different protein concentrations measured in the Bradford assay of each homogenized sample95.
Hydroxyproline assay
Quadriceps, triceps and diaphragm muscles from 3-week-old animals were weighed and incubated overnight in concentrated HCl (6 M) at 95 °C (10 mg tissue per 100 μl HCl). For muscle weights ≤20 mg, 200 μl HCl was used. Hydroxyproline standards were treated in the same way as muscle samples. Twenty-five microliters of hydrolysate was neutralized with 25 μl NaOH (6 M) and incubated with 450 μl Chloramine-T reagent (0.056 M) at RT for 25 minutes, followed by incubation with 500 μl Ehrlich’s reagent at 65 °C for 40 minutes. Samples and standards were transferred to a 96-well plate and absorbance was read at 560 nm6. All absorbance values were subtracted with blank (0 μg/ml hydroxyproline). Absorbance (A560) of standards was plotted against amount of hydroxyproline (microgram) and a linear regression was performed to determine slope and intercept. Hydroxyproline content in measured samples was calculated by equation: x (μg) =(A560 − Yaxis intercept)/slope, followed by muscle weight and acid volume adjustment in order to obtain the hydroxyproline content in muscle wet weight (μg/mg).
RNA isolation and quantitative RT-PCR analysis
RNA was isolated from 3-week-old muscles using Qiagen RNeasy Plus Universal Kit (Qiagen), following the manufacturer’s specifications. The quality and concentration of RNA were assessed using Agilent 2100 Bioanalyzer (Agilent RNA 6000 Nano Kit). One microgram of muscle RNA was used to synthesize cDNA with High Capacity cDNA Reverse Transcription Kit (Applied Biosystems) according to manufacturer’s protocol. The amplification was performed in a LightCycler 480 Real-Time PCR System (Roche), including no-RT control reactions. TaqMan probes detecting mouse TGF-β and β-actin (reference gene) were used (Applied Biosystems). Comparative CT method was used for relative quantitation.
Statistical analysis
All statistical analyses were performed with GraphPad Prism software version 6. Averaged data were reported as means ± SEM. When comparing more than two groups, D’Agostino-Pearson normality test was applied prior to choosing between one-way ANOVA or Kruskal-Wallis test. To determine significance between two particular groups, Sidak’s or Dunn’s multiple comparison post-hoc tests were applied post one-way ANOVA and Kruskal-Wallis test, respectively. Adjusted p-value was used for assessing statistical significance. For comparisons between two groups Mann-Whitney test was used and statistical significance was accepted for p < 0.05. For survival analysis log-rank Mantel-Cox test was used.
Additional Information
How to cite this article: Gawlik, K. I. et al. Potent pro-inflammatory and pro-fibrotic molecules, osteopontin and galectin-3, are not major disease modulators of laminin α2 chain-deficient muscular dystrophy. Sci. Rep. 7, 44059; doi: 10.1038/srep44059 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Voit, T. & Tomé, F. S. In Myology: The congenital muscular dystrophies. Vol. 2 1203–1238 (McGraw-Hill, New York, US, 2004).
Gawlik, K. I. & Durbeej, M. Skeletal muscle laminin and MDC1A: pathogenesis and treatment strategies. Skeletal muscle 1, 9, doi: 10.1186/2044-5040-1-9 (2011).
Holmberg, J. & Durbeej, M. Laminin-211 in skeletal muscle function. Cell adhesion & migration 7, 111–121, doi: 10.4161/cam.22618 (2013).
Miyagoe, Y. et al. Laminin alpha2 chain-null mutant mice by targeted disruption of the Lama2 gene: a new model of merosin (laminin 2)-deficient congenital muscular dystrophy. FEBS letters 415, 33–39 (1997).
Kuang, W., Xu, H., Vilquin, J. T. & Engvall, E. Activation of the lama2 gene in muscle regeneration: abortive regeneration in laminin alpha2-deficiency. Laboratory investigation; a journal of technical methods and pathology 79, 1601–1613 (1999).
Holmberg, J., Alajbegovic, A., Gawlik, K. I., Elowsson, L. & Durbeej, M. Laminin alpha2 Chain-Deficiency is Associated with microRNA Deregulation in Skeletal Muscle and Plasma. Frontiers in aging neuroscience 6, 155, doi: 10.3389/fnagi.2014.00155 (2014).
Vachon, P. H., Loechel, F., Xu, H., Wewer, U. M. & Engvall, E. Merosin and laminin in myogenesis; specific requirement for merosin in myotube stability and survival. The Journal of cell biology 134, 1483–1497 (1996).
Girgenrath, M., Dominov, J. A., Kostek, C. A. & Miller, J. B. Inhibition of apoptosis improves outcome in a model of congenital muscular dystrophy. The Journal of clinical investigation 114, 1635–1639, doi: 10.1172/JCI22928 (2004).
Pegoraro, E. et al. Congenital muscular dystrophy with primary laminin alpha2 (merosin) deficiency presenting as inflammatory myopathy. Annals of neurology 40, 782–791, doi: 10.1002/ana.410400515 (1996).
Gawlik, K. I., Holmberg, J. & Durbeej, M. Loss of Dystrophin and beta-Sarcoglycan, Respectively, Significantly Exacerbates the Phenotype of Laminin alpha2 Chain-Deficient Animals. The American journal of pathology, doi: 10.1016/j.ajpath.2013.11.017 (2014).
Wardrop, K. E. & Dominov, J. A. Proinflammatory signals and the loss of lymphatic vessel hyaluronan receptor-1 (LYVE-1) in the early pathogenesis of laminin alpha2-deficient skeletal muscle. J Histochem Cytochem 59, 167–179, doi: 10.1369/jhc.2010.956672 (2011).
Accorsi, A., Mehuron, T., Kumar, A., Rhee, Y. & Girgenrath, M. Integrin dysregulation as a possible driver of matrix remodeling in Laminin-deficient congenital muscular dystrophy (MDC1A). Journal of Neuromuscular Diseases 2, 51–61 (2015).
Elbaz, M. et al. Losartan, a therapeutic candidate in congenital muscular dystrophy: studies in the dy(2J)/dy(2J) mouse. Annals of neurology 71, 699–708, doi: 10.1002/ana.22694 (2012).
Denhardt, D. T., Noda, M., O’Regan, A. W., Pavlin, D. & Berman, J. S. Osteopontin as a means to cope with environmental insults: regulation of inflammation, tissue remodeling, and cell survival. The Journal of clinical investigation 107, 1055–1061, doi: 10.1172/JCI12980 (2001).
Liaw, L. et al. Altered wound healing in mice lacking a functional osteopontin gene (spp1). The Journal of clinical investigation 101, 1468–1478, doi: 10.1172/JCI2131 (1998).
Rodan, G. A. Osteopontin overview. Annals of the New York Academy of Sciences 760, 1–5 (1995).
Denhardt, D. T. & Guo, X. Osteopontin: a protein with diverse functions. FASEB journal: official publication of the Federation of American Societies for Experimental Biology 7, 1475–1482 (1993).
Asou, Y. et al. Osteopontin facilitates angiogenesis, accumulation of osteoclasts, and resorption in ectopic bone. Endocrinology 142, 1325–1332, doi: 10.1210/endo.142.3.8006 (2001).
Leung, T. M., Wang, X., Kitamura, N., Fiel, M. I. & Nieto, N. Osteopontin delays resolution of liver fibrosis. Laboratory investigation; a journal of technical methods and pathology 93, 1082–1089, doi: 10.1038/labinvest.2013.104 (2013).
Zahradka, P. Novel role for osteopontin in cardiac fibrosis. Circulation research 102, 270–272, doi: 10.1161/CIRCRESAHA.107.170555 (2008).
Pardo, A. et al. Up-regulation and profibrotic role of osteopontin in human idiopathic pulmonary fibrosis. PLoS Med 2, e251, doi: 10.1371/journal.pmed.0020251 (2005).
Lenga, Y. et al. Osteopontin expression is required for myofibroblast differentiation. Circulation research 102, 319–327, doi: 10.1161/CIRCRESAHA.107.160408 (2008).
Lund, S. A., Giachelli, C. M. & Scatena, M. The role of osteopontin in inflammatory processes. J Cell Commun Signal 3, 311–322, doi: 10.1007/s12079-009-0068-0 (2009).
Koh, A. et al. Role of osteopontin in neutrophil function. Immunology 122, 466–475, doi: 10.1111/j.1365-2567.2007.02682.x (2007).
Zhu, B. et al. Osteopontin modulates CD44-dependent chemotaxis of peritoneal macrophages through G-protein-coupled receptors: evidence of a role for an intracellular form of osteopontin. Journal of cellular physiology 198, 155–167, doi: 10.1002/jcp.10394 (2004).
Rittling, S. R. Osteopontin in macrophage function. Expert Rev Mol Med 13, e15, doi: 10.1017/S1462399411001839 (2011).
Lund, S. A. et al. Osteopontin mediates macrophage chemotaxis via alpha4 and alpha9 integrins and survival via the alpha4 integrin. Journal of cellular biochemistry 114, 1194–1202, doi: 10.1002/jcb.24462 (2013).
O’Regan, A. W. et al. Osteopontin is associated with T cells in sarcoid granulomas and has T cell adhesive and cytokine-like properties in vitro. J Immunol 162, 1024–1031 (1999).
Cantor, H. & Shinohara, M. L. Regulation of T-helper-cell lineage development by osteopontin: the inside story. Nat Rev Immunol 9, 137–141, doi: 10.1038/nri2460 (2009).
Hirata, A. et al. Expression profiling of cytokines and related genes in regenerating skeletal muscle after cardiotoxin injection: a role for osteopontin. The American journal of pathology 163, 203–215, doi: 10.1016/S0002-9440(10)63644-9 (2003).
Zanotti, S. et al. Osteopontin is highly expressed in severely dystrophic muscle and seems to play a role in muscle regeneration and fibrosis. Histopathology 59, 1215–1228, doi: 10.1111/j.1365-2559.2011.04051.x (2011).
Uaesoontrachoon, K. et al. Osteopontin and skeletal muscle myoblasts: association with muscle regeneration and regulation of myoblast function in vitro. Int J Biochem Cell Biol 40, 2303–2314, doi: 10.1016/j.biocel.2008.03.020 (2008).
Uaesoontrachoon, K., Wasgewatte Wijesinghe, D. K., Mackie, E. J. & Pagel, C. N. Osteopontin deficiency delays inflammatory infiltration and the onset of muscle regeneration in a mouse model of muscle injury. Dis Model Mech 6, 197–205, doi: 10.1242/dmm.009993 (2013).
Vetrone, S. A. et al. Osteopontin promotes fibrosis in dystrophic mouse muscle by modulating immune cell subsets and intramuscular TGF-beta. The Journal of clinical investigation 119, 1583–1594, doi: 10.1172/JCI37662 (2009).
Pagel, C. N., Wasgewatte Wijesinghe, D. K., Taghavi Esfandouni, N. & Mackie, E. J. Osteopontin, inflammation and myogenesis: influencing regeneration, fibrosis and size of skeletal muscle. J Cell Commun Signal 8, 95–103, doi: 10.1007/s12079-013-0217-3 (2014).
Pegoraro, E. et al. SPP1 genotype is a determinant of disease severity in Duchenne muscular dystrophy. Neurology 76, 219–226, doi: 10.1212/WNL.0b013e318207afeb (2011).
Capote, J. et al. Osteopontin ablation ameliorates muscular dystrophy by shifting macrophages to a pro-regenerative phenotype. The Journal of cell biology 213, 275–288, doi: 10.1083/jcb.201510086 (2016).
Mehuron, T. et al. Dysregulation of matricellular proteins is an early signature of pathology in laminin-deficient muscular dystrophy. Skeletal muscle 4, 14, doi: 10.1186/2044-5040-4-14 (2014).
Ochieng, J., Leite-Browning, M. L. & Warfield, P. Regulation of cellular adhesion to extracellular matrix proteins by galectin-3. Biochemical and biophysical research communications 246, 788–791, doi: 10.1006/bbrc.1998.8708 (1998).
Ochieng, J., Warfield, P., Green-Jarvis, B. & Fentie, I. Galectin-3 regulates the adhesive interaction between breast carcinoma cells and elastin. Journal of cellular biochemistry 75, 505–514 (1999).
Matarrese, P. et al. Galectin-3 overexpression protects from apoptosis by improving cell adhesion properties. Int J Cancer 85, 545–554 (2000).
Burguillos, M. A. et al. Microglia-Secreted Galectin-3 Acts as a Toll-like Receptor 4 Ligand and Contributes to Microglial Activation. Cell Rep, doi: 10.1016/j.celrep.2015.02.012 (2015).
Li, L. C., Li, J. & Gao, J. Functions of galectin-3 and its role in fibrotic diseases. The Journal of pharmacology and experimental therapeutics 351, 336–343, doi: 10.1124/jpet.114.218370 (2014).
Doe, J. A. et al. Transgenic overexpression of the alpha7 integrin reduces muscle pathology and improves viability in the dy(W) mouse model of merosin-deficient congenital muscular dystrophy type 1A. Journal of cell science 124, 2287–2297, doi: 10.1242/jcs.083311 (2011).
Henderson, N. C. et al. Galectin-3 expression and secretion links macrophages to the promotion of renal fibrosis. The American journal of pathology 172, 288–298, doi: 10.2353/ajpath.2008.070726 (2008).
Mackinnon, A. C. et al. Regulation of transforming growth factor-beta1-driven lung fibrosis by galectin-3. American journal of respiratory and critical care medicine 185, 537–546, doi: 10.1164/rccm.201106-0965OC (2012).
Ford, P. et al. TD139, A Novel Inhaled Galectin-3 Inhibitor For The Treatment Of Idiopathic Pulmonary Fibrosis (IPF). Results From The First In Human Study In Healthy Volunteers. American journal of respiratory and critical care medicine 191, A6441 (2015).
Perdiguero, E., Kharraz, Y., Serrano, A. L. & Munoz-Canoves, P. MKP-1 coordinates ordered macrophage-phenotype transitions essential for stem cell-dependent tissue repair. Cell Cycle 11, 877–886, doi: 10.4161/cc.11.5.19374 (2012).
Li, X., O’Regan, A. W. & Berman, J. S. IFN-gamma induction of osteopontin expression in human monocytoid cells. J Interferon Cytokine Res 23, 259–265, doi: 10.1089/107999003321829971 (2003).
Jeon, S. B. et al. Galectin-3 exerts cytokine-like regulatory actions through the JAK-STAT pathway. J Immunol 185, 7037–7046, doi: 10.4049/jimmunol.1000154 (2010).
Serlin, D. M. et al. Interleukin-1beta induces osteopontin expression in pulmonary fibroblasts. Journal of cellular biochemistry 97, 519–529, doi: 10.1002/jcb.20661 (2006).
Boza-Serrano, A. et al. The role of Galectin-3 in alpha-synuclein-induced microglial activation. Acta Neuropathol Commun 2, 156, doi: 10.1186/s40478-014-0156-0 (2014).
Hashimoto, M., Sun, D., Rittling, S. R., Denhardt, D. T. & Young, W. Osteopontin-deficient mice exhibit less inflammation, greater tissue damage, and impaired locomotor recovery from spinal cord injury compared with wild-type controls. J Neurosci 27, 3603–3611, doi: 10.1523/JNEUROSCI.4805-06.2007 (2007).
Nishi, Y. et al. Role of galectin-3 in human pulmonary fibrosis. Allergol Int 56, 57–65, doi: 10.2332/allergolint.O-06-449 (2007).
Konno, S. et al. Interleukin-10 and Th2 cytokines differentially regulate osteopontin expression in human monocytes and dendritic cells. J Interferon Cytokine Res 26, 562–567, doi: 10.1089/jir.2006.26.562 (2006).
Chung, A. W. et al. Galectin-3 regulates the innate immune response of human monocytes. The Journal of infectious diseases 207, 947–956, doi: 10.1093/infdis/jis920 (2013).
Villalta, S. A., Deng, B., Rinaldi, C., Wehling-Henricks, M. & Tidball, J. G. IFN-gamma promotes muscle damage in the mdx mouse model of Duchenne muscular dystrophy by suppressing M2 macrophage activation and inhibiting muscle cell proliferation. J Immunol 187, 5419–5428, doi: 10.4049/jimmunol.1101267 (2011).
Villalta, S. A. et al. Interleukin-10 reduces the pathology of mdx muscular dystrophy by deactivating M1 macrophages and modulating macrophage phenotype. Human molecular genetics 20, 790–805, doi: 10.1093/hmg/ddq523 (2011).
Pelosi, L. et al. Increased levels of interleukin-6 exacerbate the dystrophic phenotype in mdx mice. Human molecular genetics 24, 6041–6053, doi: 10.1093/hmg/ddv323 (2015).
Cohen, T. V. et al. Upregulated IL-1beta in dysferlin-deficient muscle attenuates regeneration by blunting the response to pro-inflammatory macrophages. Skeletal muscle 5, 24, doi: 10.1186/s13395-015-0048-4 (2015).
Lemos, D. R. et al. Nilotinib reduces muscle fibrosis in chronic muscle injury by promoting TNF-mediated apoptosis of fibro/adipogenic progenitors. Nature medicine 21, 786–794, doi: 10.1038/nm.3869 (2015).
Gosselin, L. E. & Martinez, D. A. Impact of TNF-alpha blockade on TGF-beta1 and type I collagen mRNA expression in dystrophic muscle. Muscle & nerve 30, 244–246, doi: 10.1002/mus.20056 (2004).
Colnot, C., Fowlis, D., Ripoche, M. A., Bouchaert, I. & Poirier, F. Embryonic implantation in galectin 1/galectin 3 double mutant mice. Developmental dynamics: an official publication of the American Association of Anatomists 211, 306–313, doi: 10.1002/(SICI)1097-0177(199804)211:4<306::AID-AJA2>3.0.CO;2-L (1998).
Meinen, S., Lin, S. & Ruegg, M. A. Angiotensin II type 1 receptor antagonists alleviate muscle pathology in the mouse model for laminin-alpha2-deficient congenital muscular dystrophy (MDC1A). Skeletal muscle 2, 18, doi: 10.1186/2044-5040-2-18 (2012).
Ishitobi, M. et al. Elevated plasma levels of transforming growth factor beta1 in patients with muscular dystrophy. Neuroreport 11, 4033–4035 (2000).
Yu, L. et al. Genetic and pharmacological inhibition of galectin-3 prevents cardiac remodeling by interfering with myocardial fibrogenesis. Circ Heart Fail 6, 107–117, doi: 10.1161/CIRCHEARTFAILURE.112.971168 (2013).
Farnworth, S. L. et al. Galectin-3 reduces the severity of pneumococcal pneumonia by augmenting neutrophil function. The American journal of pathology 172, 395–405, doi: 10.2353/ajpath.2008.070870 (2008).
Dong, S. & Hughes, R. C. Macrophage surface glycoproteins binding to galectin-3 (Mac-2-antigen). Glycoconj J 14, 267–274 (1997).
Liu, F. T. et al. Expression and function of galectin-3, a beta-galactoside-binding lectin, in human monocytes and macrophages. The American journal of pathology 147, 1016–1028 (1995).
Chen, S. C. & Kuo, P. L. The Role of Galectin-3 in the Kidneys. Int J Mol Sci 17, doi: 10.3390/ijms17040565 (2016).
Connolly, A. M., Keeling, R. M., Streif, E. M., Pestronk, A. & Mehta, S. Complement 3 deficiency and oral prednisolone improve strength and prolong survival of laminin alpha2-deficient mice. J Neuroimmunol 127, 80–87 (2002).
Cohn, R. D. et al. Angiotensin II type 1 receptor blockade attenuates TGF-beta-induced failure of muscle regeneration in multiple myopathic states. Nature medicine 13, 204–210, doi: 10.1038/nm1536 (2007).
Bogdanovich, S. et al. Functional improvement of dystrophic muscle by myostatin blockade. Nature 420, 418–421, doi: 10.1038/nature01154 (2002).
Millay, D. P. et al. Genetic and pharmacologic inhibition of mitochondrial-dependent necrosis attenuates muscular dystrophy. Nature medicine 14, 442–447, doi: 10.1038/nm1736 (2008).
Serrano, A. L. & Munoz-Canoves, P. Regulation and dysregulation of fibrosis in skeletal muscle. Experimental cell research 316, 3050–3058, doi: 10.1016/j.yexcr.2010.05.035 (2010).
Kornegay, J. N. et al. Pharmacologic management of Duchenne muscular dystrophy: target identification and preclinical trials. ILAR J 55, 119–149, doi: 10.1093/ilar/ilu011 (2014).
Pelosi, L. et al. Functional and Morphological Improvement of Dystrophic Muscle by Interleukin 6 Receptor Blockade. EBioMedicine 2, 285–293, doi: 10.1016/j.ebiom.2015.02.014 (2015).
McDouall, R. M., Dunn, M. J. & Dubowitz, V. Nature of the mononuclear infiltrate and the mechanism of muscle damage in juvenile dermatomyositis and Duchenne muscular dystrophy. Journal of the neurological sciences 99, 199–217 (1990).
Spencer, M. J. & Tidball, J. G. Do immune cells promote the pathology of dystrophin-deficient myopathies? Neuromuscular disorders: NMD 11, 556–564 (2001).
Porter, J. D. et al. A chronic inflammatory response dominates the skeletal muscle molecular signature in dystrophin-deficient mdx mice. Human molecular genetics 11, 263–272 (2002).
Jeudy, S., Wardrop, K. E., Alessi, A. & Dominov, J. A. Bcl-2 inhibits the innate immune response during early pathogenesis of murine congenital muscular dystrophy. PloS one 6, e22369, doi: 10.1371/journal.pone.0022369 (2011).
Burzyn, D. et al. A special population of regulatory T cells potentiates muscle repair. Cell 155, 1282–1295, doi: 10.1016/j.cell.2013.10.054 (2013).
Durbeej, M. & Campbell, K. P. Muscular dystrophies involving the dystrophin-glycoprotein complex: an overview of current mouse models. Current opinion in genetics & development 12, 349–361 (2002).
Chamberlain, J. S., Metzger, J., Reyes, M., Townsend, D. & Faulkner, J. A. Dystrophin-deficient mdx mice display a reduced life span and are susceptible to spontaneous rhabdomyosarcoma. FASEB journal: official publication of the Federation of American Societies for Experimental Biology 21, 2195–2204, doi: 10.1096/fj.06-7353com (2007).
Rooney, J. E., Knapp, J. R., Hodges, B. L., Wuebbles, R. D. & Burkin, D. J. Laminin-111 protein therapy reduces muscle pathology and improves viability of a mouse model of merosin-deficient congenital muscular dystrophy. The American journal of pathology 180, 1593–1602, doi: 10.1016/j.ajpath.2011.12.019 (2012).
Wagner, K. R., McPherron, A. C., Winik, N. & Lee, S. J. Loss of myostatin attenuates severity of muscular dystrophy in mdx mice. Annals of neurology 52, 832–836, doi: 10.1002/ana.10385 (2002).
Li, Z. F., Shelton, G. D. & Engvall, E. Elimination of myostatin does not combat muscular dystrophy in dy mice but increases postnatal lethality. The American journal of pathology 166, 491–497, doi: 10.1016/S0002-9440(10)62271-7 (2005).
Van Ry, P. M., Minogue, P., Hodges, B. L. & Burkin, D. J. Laminin-111 improves muscle repair in a mouse model of merosin-deficient congenital muscular dystrophy. Human molecular genetics 23, 383–396, doi: 10.1093/hmg/ddt428 (2014).
Van Ry, P. M., Wuebbles, R. D., Key, M. & Burkin, D. J. Galectin-1 Protein Therapy Prevents Pathology and Improves Muscle Function in the mdx Mouse Model of Duchenne Muscular Dystrophy. Molecular therapy: the journal of the American Society of Gene Therapy 23, 1285–1297, doi: 10.1038/mt.2015.105 (2015).
Xu, H., Wu, X. R., Wewer, U. M. & Engvall, E. Murine muscular dystrophy caused by a mutation in the laminin alpha 2 (Lama2) gene. Nature genetics 8, 297–302, doi: 10.1038/ng1194-297 (1994).
Sunada, Y., Bernier, S. M., Utani, A., Yamada, Y. & Campbell, K. P. Identification of a novel mutant transcript of laminin alpha 2 chain gene responsible for muscular dystrophy and dysmyelination in dy2J mice. Human molecular genetics 4, 1055–1061 (1995).
Franzen, A., Hultenby, K., Reinholt, F. P., Onnerfjord, P. & Heinegard, D. Altered osteoclast development and function in osteopontin deficient mice. J Orthop Res 26, 721–728, doi: 10.1002/jor.20544 (2008).
Gawlik, K. I. & Durbeej, M. Deletion of integrin alpha7 subunit does not aggravate the phenotype of laminin alpha2 chain-deficient mice. Sci Rep 5, 13916, doi: 10.1038/srep13916 (2015).
Gawlik, K. I. & Durbeej, M. Transgenic overexpression of laminin alpha1 chain in laminin alpha2 chain-deficient mice rescues the disease throughout the lifespan. Muscle & nerve 42, 30–37, doi: 10.1002/mus.21616 (2010).
Svensson, M., Rosvall, P., Boza-Serrano, A., Andersson, E., Lexell J. & Deierborg, T. Forced treadmill exercise can induce stress and increase neuronal damage in a mouse model of global cerebral ischemia. Neurobiol Stress 5, 8–18, doi: 10.1016/j.ynstr.2016.09.002 (2016).
Acknowledgements
This work was supported by the Swedish Research Council, Association Française contre les Myopathies, Greta and Johan Kock Foundation, Linnea and Josef Carlsson Foundation, Fanny Ekdahl Foundation, Lars Hierta Foundation, The Royal Physiografic Society, Crafoord Foundation, Olle Engkvist Byggmästare Foundation, Thelma Zoéga Foundation and CNPq - National Council for Scientific and Technological Development, Brazil (Bernardo M.S. Oliveira). We thank Dr. Anna Hultgårdh-Nilsson for osteopontin knockout mice; Dr. Catharina Müller for paraffin sections of limb muscles; Dr. Uwe Rauch for discussion about M1 macrophages; Jonna Uddbäck, Katrina Kappy, and Dr. Anki Knutsson for technical assistance and Dr. Hakon Leffler for galectin-3 antibody.
Author information
Authors and Affiliations
Contributions
K.I.G. and J.H. contributed equally to this manuscript. K.I.G., J.H. and M.D. conceived and designed the study. K.I.G., J.H., M.S., M.E. and B.M.S.O. performed the experiments. K.I.G., J.H., M.S., M.E., B.M.S.O., T.D. and M.D. analysed the data. K.I.G. and J.H. generated figures. K.I.G. and J.H. wrote the manuscript. T.D. and M.D. supervised the study and revised the article content. All authors approved of the submitted version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Gawlik, K., Holmberg, J., Svensson, M. et al. Potent pro-inflammatory and pro-fibrotic molecules, osteopontin and galectin-3, are not major disease modulators of laminin α2 chain-deficient muscular dystrophy. Sci Rep 7, 44059 (2017). https://doi.org/10.1038/srep44059
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep44059
- Springer Nature Limited
This article is cited by
-
Early skeletal muscle pathology and disease progress in the dy3K/dy3K mouse model of congenital muscular dystrophy with laminin α2 chain-deficiency
Scientific Reports (2019)
-
Tissue-enhanced plasma proteomic analysis for disease stratification in amyotrophic lateral sclerosis
Molecular Neurodegeneration (2018)