Abstract
Alignment-free (AF) approaches have recently been highlighted as alternatives to methods based on multiple sequence alignment in phylogenetic inference. However, the sensitivity of AF methods to genome-scale evolutionary scenarios is little known. Here, using simulated microbial genome data we systematically assess the sensitivity of nine AF methods to three important evolutionary scenarios: sequence divergence, lateral genetic transfer (LGT) and genome rearrangement. Among these, AF methods are most sensitive to the extent of sequence divergence, less sensitive to low and moderate frequencies of LGT and most robust against genome rearrangement. We describe the application of AF methods to three well-studied empirical genome datasets and introduce a new application of the jackknife to assess node support. Our results demonstrate that AF phylogenomics is computationally scalable to multi-genome data and can generate biologically meaningful phylogenies and insights into microbial evolution.
Similar content being viewed by others
Introduction
Phylogenomics is the study of evolutionary relationships via comparative analysis of genome-scale data, intersecting the fields of evolution and genomics. Phylogenomic methods are used to assess diverse biological hypotheses, e.g. the emergence and spread of antibiotic resistance among bacterial pathogens1, or the tree of life2. Evolutionary relationships are inferred based on sequence homology in a comparative gene-by-gene3, concatenated multi-gene4 or whole-genome approach5. Classical phylogenomic approaches consist of three main steps: clustering of homologous sequences, multiple sequence alignment (MSA) of each cluster and inference of a phylogenetic tree based on the alignment, e.g. using maximum likelihood (ML) or Bayesian methods. MSA is a crucial step in these approaches. In carrying out MSA we implicitly assume that by inserting gaps and sliding blocks of sequence relative to each other, we can generate a position-wise hypothesis of homology across the entire length of the sequences. In reality, this may be unrealistic because genes and genomes are subject to recombination, inversion, rearrangement and lateral genetic transfer6.
These processes are intensified in most microbial genomes, where LGT, inversions and rearrangement are rampant5,7,8. These events violate models of nucleotide substitution across sequence positions and lineages, commonly specified in MSA-based approaches and thus bias subsequent phylogenetic inference9,10. In addition, increasing numbers of microbial genomes are now available both from a broad taxonomic breadth11,12 and within individual taxa13. MSA does not scale well with genome-scale data, such that classical approaches will soon be impractical for large-scale comparative genome analysis.
Alternatively, alignment-free (AF) methods, based on comparison of sub-sequences of defined length (known variously as k-mers or n-grams) instead of full-length sequences, can be used14,15,16. AF methods utilise subsequences (k-mers) as the basis for calculating an overall pairwise distance between one sequence and another. These methods (Fig. 1) fall into two broad categories15: those based on the number or frequency of k-mers shared between two sequences and those based on the length of matching k-mers. Furthermore the k-mers may or may not be required to match exactly. In general, match-length methods perform well in the comparison of highly similar sequences, due to the large proportion of exact matches. Algorithmically, most AF methods show linear-time complexity, but word-count methods are usually faster and, depending on the implementation, can require less memory than match-length methods.
Alignment-free methods classification.
Classification of alignment-free methods, modified following Haubold15. LZ: Lempel-Ziv.
Recent studies demonstrate the potential of AF approaches in the accurate inference of phylogenetic relationships15,17, with several methods more robust than MSA-based approaches against gene-level evolutionary scenarios including among-site rate heterogeneity, compositional biases, genetic recombination and insertions/deletions. The AF methods assessed were more sensitive to recent sequence divergence and sequence truncation17. Across diverse empirical gene-family datasets, AF approaches perform well for sequences sharing low divergence, at greater computation speed. However, the sensitivity of AF approaches to evolutionary scenarios at whole-genome level including genome rearrangements and LGT and their scalability to multi-genome data, remain to be systematically investigated.
Classical phylogenetic methods employ heuristics and iterative sampling strategies have been adopted to assess node support, among which the best-known are the non-parametric bootstrap18 in ML and the posterior probability of tree bipartitions in Bayesian inference19,20. In comparison, AF approaches provide exact solutions with no iterative processes, with the consequence that phylogenetic trees generated using AF approaches lack estimates of node support. Bootstrap and subsampling techniques have been proposed in recent studies21,22,23, but most studies of AF approaches focused only on topologies, with no or little emphasis on node support.
Here, using both simulated and empirical data we systematically assess the sensitivity of nine existing AF methods to genome-scale evolutionary scenarios involving sequence divergence, LGT and rearrangement. We introduce a new application of the jackknife24 technique to provide node-support values to trees inferred by AF approaches and demonstrate the scalability and potential of AF approaches in inferring phylogenetic trees quickly and accurately from genome-scale data.
Results
We investigated nine AF phylogenetic methods:  25, co-phylog26, cvt27, ffp28,29,30 and spaced31,32 based on word count and acs33, gram34, kmacs31,35 and kr36 based on match length (see Supplementary Information). The distance matrix generated by each of these methods was used to infer phylogenetic relationships using neighbour-joining. As a measure of accuracy we used the Robinson-Foulds distance37, which evaluates the topological congruence between an inferred tree and a reference tree. We designate RF as the normalised Robinson-Foulds distance (see Methods): RF = 0 indicates that the test-tree topology is completely congruent with that of the reference, while RF = 1 indicates that the two trees have no bipartitions in common.
25, co-phylog26, cvt27, ffp28,29,30 and spaced31,32 based on word count and acs33, gram34, kmacs31,35 and kr36 based on match length (see Supplementary Information). The distance matrix generated by each of these methods was used to infer phylogenetic relationships using neighbour-joining. As a measure of accuracy we used the Robinson-Foulds distance37, which evaluates the topological congruence between an inferred tree and a reference tree. We designate RF as the normalised Robinson-Foulds distance (see Methods): RF = 0 indicates that the test-tree topology is completely congruent with that of the reference, while RF = 1 indicates that the two trees have no bipartitions in common.
Simulated data
Using simulated data, we independently assessed the sensitivity of each AF approach to evolutionary scenarios of genome divergence, LGT and genome rearrangement. Because the underlying tree on which each dataset has been simulated is known, we used that as the reference. Here we report, for each scenario, results generated with each AF approach using the parameter setting that yields the smallest mean RF globally across all rates; these settings are optimal in this context. The parameters of interest are k (k-mer length) for cvt,  and ffp; K (half-context length) for co-phylog; n (number of patterns) for spaced; and mm (number of mismatches) for kmacs. We used default parameters for acs, gram and kr.
and ffp; K (half-context length) for co-phylog; n (number of patterns) for spaced; and mm (number of mismatches) for kmacs. We used default parameters for acs, gram and kr.
Genome divergence
We simulated sets of genomes (size N from 25 to 35) using a birth-death model across different levels of genome divergence as determined by the mutation rate m = 0.1, 0.5 and 0.9; m is the number of nucleotide substitutions per iteration. At maximum rate m = 1, almost all nucleotides are substituted per iteration38. Figure 2 shows the mean RF for each approach (50 replicates) across these values of m. Optimal settings for each approach were determined based on a wide range of parameters (see Supplementary Information and Supplementary Figure S1 for details). For all the word-count methods except spaced, RF is minimum at m = 0.5 (Fig. 2a), i.e. at an intermediate level of sequence divergence. On the other hand, for the match-length methods (except kmacs) RF increases with greater sequence divergence (Fig. 2b), suggesting that their accuracy in inferring the correct genome phylogeny decreases as the sequences become more dissimilar from one another.
Interestingly, the pattern of RF values observed for kmacs (RF = 0.06, 0.05 and 0.08 at m = 0.1, 0.5 and 0.9) follows that of most word-count methods, while the pattern of RF values we find for spaced (RF = 0.10, 0.31 and 0.48 at m = 0.1, 0.5 and 0.9) increases with greater sequence divergence, as with most of the match-length methods.
Lateral genetic transfer
We simulated genome sets at different extents of LGT as determined by the mean number of LGT events per iteration l = 0, 5, 25, 125, 250, 500; l is the mean number of LGT events attempted in the set at each iteration (i = 5000) during the simulation. Thus at l = 5, the simulator attempted five LGT events per iteration across a genome set (see Methods). LGT events were simulated to occur at random, i.e. anywhere along a genome sequence and between any pair of genomes in a set.
Figure 3 shows the mean RF for each AF method (50 replicates) across different values of l. Optimal settings for each method were determined by tuning a range of parameters (Supplementary Information and Supplementary Figure S2). In general, word-count methods (Fig. 3a) achieved lower RF values than did match-length methods (Fig. 3b) against the complication of LGT. Not surprisingly, for most AF methods the accuracy of inference falls off with increasing extent of LGT. Several methods were nonetheless able to reconstruct the reference tree with little incongruence at all but the two highest frequencies of LGT simulated, notably co-phylog, cvt and kmacs (RF < 0.10 at l ≤ 125; Fig. 3).
The word-count method spaced (Fig. 3a) and the match-length methods gram and kr (Fig. 3b) were relatively inaccurate (RF > 0.2 across l) by this measure. Although spaced and gram are robust against variation of l, the inaccuracy of kr decreased with increasing l while remaining unacceptably high. As mean RF values approaching 1.0 indicate that many of the inferred topological features are scarcely distinguishable from those of a random tree39, it appears that our simulation scenario was too extreme for kr.
We also varied the maximum evolutionary distance within which LGT can occur. At fixed l = 5 we simulated LGT while setting the divergence factor d = 200, 1000, 3000 or 5000, where d is the maximum number of iterations (generations) that can separate two genomes from their common ancestor for a proposed LGT event to be accepted. This simulates biological situations in which genetic material can be successfully transferred only among related organisms due to e.g. limited plasmid host range, or a threshold of local sequence identity below which homologous recombination is not successful. Figure 4 shows the mean RF for each AF method (50 replicates) across d at otherwise optimal parameter settings (Supplementary Information and Supplementary Figure S3). For both word-count (Fig. 4a) and match-length (Fig. 4b) methods, RF varies little with d. Thus the effectiveness of these AF methods to reconstruct the reference tree in the presence of LGT is little affected by relatedness of the sequence donor.
Genome rearrangement
We simulated genome sets as above (see Methods for details) at different frequencies of genome rearrangement r = 0.00, 0.01, 0.10 or 1.00 (each in 50 replicates). A rearrangement event was simulated as an inverted translocation occurring at any position of a genome. The latter three frequencies correspond to rearrangement of approximately 0.20%, 2.0% and 20% of each genome sequence (by length). As above, we report results generated using the parameter settings optimal for each AF method (see Supplementary Information and Supplementary Figures S4 and S5).
Figure 5 shows the mean RF for each method across these values of r, at optimal parameters. Three of these AF methods, co-phylog,  and kr, were affected by these simulated rearrangements much less (RF < 0.03 across r) than were the others (RF > 0.05). No trend with r was apparent. These results extend an earlier study17 in which AF methods were likewise found to be robust against genetic recombination at the level of individual genes.
and kr, were affected by these simulated rearrangements much less (RF < 0.03 across r) than were the others (RF > 0.05). No trend with r was apparent. These results extend an earlier study17 in which AF methods were likewise found to be robust against genetic recombination at the level of individual genes.
Empirical data
To assess the performance of the AF methods in application to empirical data, we used three sets of microbial genome sequences: (a) 143 bacterial and archaeal genomes8 previously used to infer the extent of LGT across divergent taxa, (b) 27 genomes of Escherichia coli and Shigella3 used to infer LGT among more-closely related taxa and (c) eight Yersinia genomes that are too similar in sequence for classical phylogenetic inference, but share patterns of genome rearrangement5 (see Methods and Supplementary Information for details). As references we used the MRP supertree40 summarising the well-resolved subtrees inferred by a standard MSA-based approach from single-copy protein sets for the former two studies3,8 and the consensus phylogenetic network based on genomic inversion events inferred from a whole-genome alignment for the Yersinia dataset5. For each AF tree we computed the jackknife (JK) support for each node across 100 pseudo-replicates (see Methods).
143 bacterial and archaeal genomes
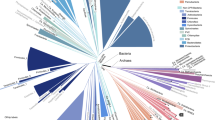
Figure 6 shows the 143-taxon tree inferred using an AF approach ( at k = 24), with the JK support for each node. Among the trees we inferred using other AF methods and parameter settings (see Supplementary Information and Supplementary Figure S6), this tree shows the greatest topological similarity (RF = 0.42) to the reference supertree8. Although 42% of bipartitions are incongruent with the reference, we recover 13 of the 15 phylum-level “backbone” nodes identified by Beiko et al.8 (Fig. 6), some with very strong JK support (e.g. Bacteroidetes 100%, Chlamydiales 99%, high G + C Firmicutes 98%). An inclusive grouping of proteobacteria is poorly supported (18%), but monophyletic epsilon- (99%) and alpha-proteobacterial (71%) clades are recovered, while the beta- and gamma-Proteobacteria form a single modestly supported (62%) clade within which many gamma-Proteobacteria constitute a solid (100%) sub-clade. By contrast, unconvincing JK values are recovered for several nodes along the subtree that joins the beta- and remaining gamma-Proteobacteria. There are two major discrepancies between our
at k = 24), with the JK support for each node. Among the trees we inferred using other AF methods and parameter settings (see Supplementary Information and Supplementary Figure S6), this tree shows the greatest topological similarity (RF = 0.42) to the reference supertree8. Although 42% of bipartitions are incongruent with the reference, we recover 13 of the 15 phylum-level “backbone” nodes identified by Beiko et al.8 (Fig. 6), some with very strong JK support (e.g. Bacteroidetes 100%, Chlamydiales 99%, high G + C Firmicutes 98%). An inclusive grouping of proteobacteria is poorly supported (18%), but monophyletic epsilon- (99%) and alpha-proteobacterial (71%) clades are recovered, while the beta- and gamma-Proteobacteria form a single modestly supported (62%) clade within which many gamma-Proteobacteria constitute a solid (100%) sub-clade. By contrast, unconvincing JK values are recovered for several nodes along the subtree that joins the beta- and remaining gamma-Proteobacteria. There are two major discrepancies between our  tree and the MRP reference: the Spirochaetales no longer form a monophyletic clade (Treponema pallidum is attracted to the problematic8 Aquifex and Thermotoga genomes), while our Crenarchaeota becomes non-exclusive (30%) with the inclusion of Nanoarchaeum. Overall, basal nodes exhibit lower JK support than do the more-terminal and leaf nodes, as is often the case for MSA-based trees of similar phyletic depth. As has been seen before41, most of the topological differences between the AF tree (Fig. 6) and the reference supertree arise from how bipartitions are resolved on short branches within major groups, not from disagreement about the membership of these groups.
tree and the MRP reference: the Spirochaetales no longer form a monophyletic clade (Treponema pallidum is attracted to the problematic8 Aquifex and Thermotoga genomes), while our Crenarchaeota becomes non-exclusive (30%) with the inclusion of Nanoarchaeum. Overall, basal nodes exhibit lower JK support than do the more-terminal and leaf nodes, as is often the case for MSA-based trees of similar phyletic depth. As has been seen before41, most of the topological differences between the AF tree (Fig. 6) and the reference supertree arise from how bipartitions are resolved on short branches within major groups, not from disagreement about the membership of these groups.
AF phylogeny of 143 prokaryote genomes.
Phylogenetic tree of 143 prokaryote genomes using  at k = 24, supported by JK values. The 15 phylum-level backbone nodes of Beiko et al.8 are marked with solid circles.
at k = 24, supported by JK values. The 15 phylum-level backbone nodes of Beiko et al.8 are marked with solid circles.
27 Escherichia coli and Shigella genomes
Figure 7 shows the phylogenetic tree of 27 E. coli and Shigella genomes generated using co-phylog (K = 8; Fig. 7a) or  (k = 26; Fig. 7b), as well as the reference MRP supertree that summarises 5282 single-copy protein trees3 (Fig. 7c). These 27 taxa have been assigned to six distinct groups (the E. coli reference, or ECOR, strains)3,42 based on allelic diversity of 11 genes43. Each ECOR group is monophyletic, except B2 in both AF trees and A in the
(k = 26; Fig. 7b), as well as the reference MRP supertree that summarises 5282 single-copy protein trees3 (Fig. 7c). These 27 taxa have been assigned to six distinct groups (the E. coli reference, or ECOR, strains)3,42 based on allelic diversity of 11 genes43. Each ECOR group is monophyletic, except B2 in both AF trees and A in the  tree; the relationships among the ECOR groups in the co-phylog and MRP trees are identical. The taxa labeled with an asterisk in each AF tree (Fig. 7a,b) are positioned differently in comparison to the reference. In comparison to the reference (Fig. 7c), the same relationship among the six phyletic groups is recovered in the tree generated using co-phylog (Fig. 7a) at strong JK support (≥84% in all but two nodes), with minor topological difference within Groups B1 and D (RF = 0.083): E. coli 55989 (instead of E24377A) is placed as basal lineage in Group B1, while E. coli UTI89 (instead of E. coli S88) is placed as sister to E. coli APEC O1 in Group D. On the other hand, the tree generated using
tree; the relationships among the ECOR groups in the co-phylog and MRP trees are identical. The taxa labeled with an asterisk in each AF tree (Fig. 7a,b) are positioned differently in comparison to the reference. In comparison to the reference (Fig. 7c), the same relationship among the six phyletic groups is recovered in the tree generated using co-phylog (Fig. 7a) at strong JK support (≥84% in all but two nodes), with minor topological difference within Groups B1 and D (RF = 0.083): E. coli 55989 (instead of E24377A) is placed as basal lineage in Group B1, while E. coli UTI89 (instead of E. coli S88) is placed as sister to E. coli APEC O1 in Group D. On the other hand, the tree generated using  (Fig. 7b; all nodes JK ≥ 90%) is less congruent with the reference (RF = 0.45); all Shigella isolates (noted as Group S) form a strongly supported clade (JK = 100%) with E. coli ATCC 8739, while in the reference tree Shigella dysenteriae is placed externally to the other Shigella isolates, as sister to Group E, the pathogenic E. coli O157:H7 isolates. As shown in Fig. 7b, groups A and B1 are sister groups, supporting previous studies42,44 (Fig. 7b) and group E is placed within a strongly supported clade (JK 100%) together with isolates of Group B2 and D and not with Groups A, B1 and S as shown in the reference (Fig. 7c).
(Fig. 7b; all nodes JK ≥ 90%) is less congruent with the reference (RF = 0.45); all Shigella isolates (noted as Group S) form a strongly supported clade (JK = 100%) with E. coli ATCC 8739, while in the reference tree Shigella dysenteriae is placed externally to the other Shigella isolates, as sister to Group E, the pathogenic E. coli O157:H7 isolates. As shown in Fig. 7b, groups A and B1 are sister groups, supporting previous studies42,44 (Fig. 7b) and group E is placed within a strongly supported clade (JK 100%) together with isolates of Group B2 and D and not with Groups A, B1 and S as shown in the reference (Fig. 7c).
Phylogenetic trees of 27 E. coli and Shigella species.
(a) Tree generated using co-phylog at K = 8, supported by JK values. (b) Tree generated using  at k = 26, supported by JK values. (c) MRP tree constructed from 5282 Bayesian protein trees. Taxa labeled with an asterisk in each AF tree (a,b) are positioned differently in comparison to the reference (c). ECOR groups and Shigella (S) are indicated.
at k = 26, supported by JK values. (c) MRP tree constructed from 5282 Bayesian protein trees. Taxa labeled with an asterisk in each AF tree (a,b) are positioned differently in comparison to the reference (c). ECOR groups and Shigella (S) are indicated.
Eight Yersinia genomes
Figure 8 shows the phylogenetic trees based on  at k = 7 (Fig. 8a) and k = 9 (Fig. 8b). These topologies are well-supported, with all nodes showing JK ≥ 74% and ≥97% respectively. Topologically the k = 7 tree is more congruent with the reference, which was based on inversion events5, than is the k = 9 tree. The two Y. pseudotuberculosis isolates are sisters (81%) in the k = 7 tree, as in the reference5. At k = 9, on the other hand, Y. pseudotuberculosis IP31758 groups solidly (100%) with Y. pestis KIM relative to the others. In the whole-genome alignment (generated using progressiveMauve in the previous study5; Supplementary Figure S7) the region of the Y. pseudotuberculosis IP31758 genome between about 1.3–3.3 Mbp shares more-similar configuration of locally collinear blocks5 with the genome of Y. pestis KIM than with the genome of Y. pseudotuberculosis IP32953 (e.g. the corresponding region is largely in reverse complement). The two Y. pseudotuberculosis genomes are the most dissimilar in this set, sharing only 40.5% of all 12-mers present in these two genomes, whereas Y. pseudotuberculosis IP31758 and Y. pestis Pestoides F 15–70 are the most similar (72.4% of shared 12-mers; Supplementary Table S1). Thus depending on the value of k,
at k = 7 (Fig. 8a) and k = 9 (Fig. 8b). These topologies are well-supported, with all nodes showing JK ≥ 74% and ≥97% respectively. Topologically the k = 7 tree is more congruent with the reference, which was based on inversion events5, than is the k = 9 tree. The two Y. pseudotuberculosis isolates are sisters (81%) in the k = 7 tree, as in the reference5. At k = 9, on the other hand, Y. pseudotuberculosis IP31758 groups solidly (100%) with Y. pestis KIM relative to the others. In the whole-genome alignment (generated using progressiveMauve in the previous study5; Supplementary Figure S7) the region of the Y. pseudotuberculosis IP31758 genome between about 1.3–3.3 Mbp shares more-similar configuration of locally collinear blocks5 with the genome of Y. pestis KIM than with the genome of Y. pseudotuberculosis IP32953 (e.g. the corresponding region is largely in reverse complement). The two Y. pseudotuberculosis genomes are the most dissimilar in this set, sharing only 40.5% of all 12-mers present in these two genomes, whereas Y. pseudotuberculosis IP31758 and Y. pestis Pestoides F 15–70 are the most similar (72.4% of shared 12-mers; Supplementary Table S1). Thus depending on the value of k,  can draw out phylogenetic signal that supports either the species classification (Fig. 8a) or the major rearrangement events apparent in the whole-genome alignment (Fig. 8b).
can draw out phylogenetic signal that supports either the species classification (Fig. 8a) or the major rearrangement events apparent in the whole-genome alignment (Fig. 8b).
Computational efficiency and scalability
Table 1 shows the computation time (t) taken by each AF method to generate pairwise distance matrices for each of the three empirical datasets described above, using a single CPU and only one thread for the multi-threaded methods (e.g.  and spaced). For each method, t increases with N. The approach using ffp is the fastest, requiring t = 0.11, 0.65 and 2.22 minutes at N = 8, 27 and 143, while the spaced and acs methods are the most computationally intensive among these nine methods. The requirement for memory varies over two orders of magnitude, from less than 500 Mb for kmacs to more than 60 Gb for acs and
and spaced). For each method, t increases with N. The approach using ffp is the fastest, requiring t = 0.11, 0.65 and 2.22 minutes at N = 8, 27 and 143, while the spaced and acs methods are the most computationally intensive among these nine methods. The requirement for memory varies over two orders of magnitude, from less than 500 Mb for kmacs to more than 60 Gb for acs and  at N = 143.
at N = 143.
Discussion
In this study we demonstrate that AF phylogenetic approaches can be used to quickly and accurately infer phylogenomic relationships of microbes using whole-genome data. We also introduce for the first time a method, based on the jackknife, to provide node-support values in phylogenetic trees constructed using AF approaches.
We examined two types of AF methods in this study. In general, the methods based on word count, particularly co-phylog and  , outperformed match-length methods. All these methods performed well on highly similar sequence data, but the methods based on word count (except spaced) are more robust as the input data become more divergent. All these AF methods proved robust against LGT at frequencies below l = 250 (at which every gene is likely to have affected by LGT at least once)38, antiquity of LGT and genome-scale rearrangement, extending our earlier results based on analysis of gene-scale sequence data17. Parameter values in this study were optimised for these data, based on our results and (for other AF methods) the original authors’ recommendations. These parameters are most sensitive to sequence length and genome divergence. AF methods have the advantage of being computationally fast, so we recommend that users compare a range of parameters on this basis.
, outperformed match-length methods. All these methods performed well on highly similar sequence data, but the methods based on word count (except spaced) are more robust as the input data become more divergent. All these AF methods proved robust against LGT at frequencies below l = 250 (at which every gene is likely to have affected by LGT at least once)38, antiquity of LGT and genome-scale rearrangement, extending our earlier results based on analysis of gene-scale sequence data17. Parameter values in this study were optimised for these data, based on our results and (for other AF methods) the original authors’ recommendations. These parameters are most sensitive to sequence length and genome divergence. AF methods have the advantage of being computationally fast, so we recommend that users compare a range of parameters on this basis.
The jackknife approach was simple to apply without prior sequence alignment. Here we deleted 40% of the data because 40% was previously shown to provide a reasonable balance between the generation of useful replicates without totally losing phylogenetic signal in sequence data45,46. The resulting JK support values appear to be biologically meaningful, as recognised taxa were often strongly supported (see also Supplementary Information and Supplementary Table S2). Particularly in the 143-genome dataset, JK support tended to decrease with increasing sequence divergence within-group, as is also seen with bootstrap support and Bayesian posterior probability in MSA-based studies. Problematic clades tend to be more weakly supported; for example, the beta- and gamma-Proteobacteria (JK 62% in Fig. 6) were the most-frequent LGT partners in the MSA-based study of this dataset8. Similarly, the appearance of Pseudomonas genomes within a clade of beta-Proteobacteria (JK 62%) can be explained by more than 150 LGT events8. Our AF tree also supports the hypothesis that Archaea is monophyletic (41%) and distinct from Bacteria47 (JK support 90% excluding the single representative (Rhodopirellula) in this dataset of the problematic Planctomycetes)48.
Likewise, AF trees based on the 27-genome E. coli and Shigella dataset recover the ECOR reference groups3. We observed two distinct trees: one congruent with the MRP supertree and another that supports a monophyletic Shigella and the sister-group relationship between ECOR groups A and B1 as previously expected44 but not recovered in the supertree. As described above, relationships among the Yersinia strains cannot be resolved using a standard sequence-based approach: the 16 S ribosomal RNA sequence is identical for seven of these isolates, while the eighth (Y. pseudotuberculosis IP31758) differs in only five positions. All our AF-based methods (Fig. 8 and Supplementary Information) recovered only two types of topology, one of which reflects the extent of genome rearrangement among these strains. Given the intricate evolution of these taxa and the fact that we cannot recreate history the true phylogeny in these instances remains an open question.
Our use of an MRP supertree as reference is not entirely unproblematic in this context. A supertree summarises the well-supported topological features in a set of input trees; in these two cases3,8 and many others, each input tree arises via a workflow of orthogroup identification, MSA and Bayesian phylogenetic inference. Even in microbial genomes, where protein-coding genes tend to be tightly packed, many regions are excluded from contributing to the reference topology including non-protein-coding, intergenic, rearranged, mis-assembled, unalignable or low-complexity regions, paralogs, pseudogenes and repetitive elements. By contrast, AF approaches make use of the entire genome sequence and are less dependent on pre-defined evolutionary models; assumptions implicit in these models are known to be highly simplified and may be unrealistic9.
The computational complexity of all the AF methods can be found in previous studies25,26,27,30,31,34,36 and ranges from O(n) for ffp, to O(k × n × z) for kmacs, with n the number of sequences, k the number of mismatches and z the average number of matches between two sequences. An advantage that AF methods have over the standard MSA-based approaches is their scalability15,17. Among the AF methods we tested, methods based on word counts can be orders of magnitude faster than those based on match lengths, but tend to be more memory-intensive and more sensitive to parameter settings. It has been proposed49 that filtering out non-informative k-mers can be a useful approach to reducing memory requirements and one can imagine a systematic, adaptive approach to parameterisation of k based on the nature of the sequence data (e.g. extent of divergence, presence of repetitive elements) as learned from simulated sequences. Biologically motivated approaches could likewise be explored, e.g. the adaptive use or weighting of k-mer patterns common in genomes (as learned from a comprehensive genome k-mer database) and/or relevant to biological processes or molecular function.
In cases where assessment of aligned positions across conserved regions is necessary, e.g. to infer structural features of proteins, MSA remains indispensable. However, these and other results show that AF approaches present exciting alternatives in phylogenetic inference for large sets of microbial-sized genomes at different phyletic breadth, even in the presence of genomic rearrangement and LGT. Indeed it might be asked whether AF approaches might be modified to detect genomic regions of lateral origin; we intend to address this in the very near future.
Methods
Simulated genome data
For all programs mentioned below, default settings were used unless otherwise specified. We simulated sets of genomes using two different programs depending on their functionality: Evolsimulator38 to simulate scenarios relevant to lateral genetic transfer (LGT) and ALF50 to simulate genome rearrangement. Evolsimulator allows the user to specify rates independently for speciation, extinction, gene content, extent of gene loss and/or duplication, nucleotide substitution and LGT. Moreover, LGT events can be allowed to succeed automatically, or can be evaluated under different criteria (e.g. controlled by gene complement, divergence, G + C similarity and/or host habitat); the receptivity of each genome to LGT can also be specified. We used the Generalised Time-Reversible (GTR)51 substitution model (rate parameters a = 0.987, b = 0.11, c = 0.218, d = 0.243, e = 0.395) in all simulations. We detail our simulation strategy for each evolutionary scenario below.
Sequence divergence and lateral genetic transfer
Each set of genomes was simulated under a birth-and-death model at speciation rate = extinction rate = 0.5. The number of genomes in each set was allowed to vary from 25 to 35, with each containing 2000–3000 genes of length 240–1500 nucleotides. LGT receptivity was at set at minimum 0.2, mean 0.5 and maximum 0.8, mutation rate m = 0.4–0.6 and number of generations i = 5000. Sequence change was simulated under a discrete approximation of the gamma distribution (shape parameter  = 1.0, 8 categories). To study the effect of LGT we simulated LGT between randomly selected genomes, varying the mean number of LGT events attempted per iteration l = 5, 25, 125, 250 or 500 (each scenario in 50 replicates). We also simulated datasets in which genomes were not selected at random (but instead were restricted by number of generations from their common ancestor) by varying the divergence factor d: 200, 1000, 3000 or 5000 (with l fixed at 5, m 0.4–0.6); larger d allows more-dissimilar sequences to be transferred. To assess the effect of genome divergence we set l = 5 and simulated datasets at three rates of mutation m: 0.01–0.2, 0.4–0.6 and 0.8–0.99. All other parameters in this simulation follow Beiko et al.52.
= 1.0, 8 categories). To study the effect of LGT we simulated LGT between randomly selected genomes, varying the mean number of LGT events attempted per iteration l = 5, 25, 125, 250 or 500 (each scenario in 50 replicates). We also simulated datasets in which genomes were not selected at random (but instead were restricted by number of generations from their common ancestor) by varying the divergence factor d: 200, 1000, 3000 or 5000 (with l fixed at 5, m 0.4–0.6); larger d allows more-dissimilar sequences to be transferred. To assess the effect of genome divergence we set l = 5 and simulated datasets at three rates of mutation m: 0.01–0.2, 0.4–0.6 and 0.8–0.99. All other parameters in this simulation follow Beiko et al.52.
Genome rearrangement
We simulated different extents of rearrangement (random events of inverted translocation) within genome sequence sets using ALF, setting the rate r = 0.00, 0.01, 0.10 or 1.00 (each in 50 replicates). These values correspond to zero and approximately 0.2%, 2.0% and 20% of the length of each genome undergoing rearrangement. Other settings were number of genomes in each set N = 30 (each with 2500 genes of length 240–2000 nt), speciation rate = 0.5, extinction rate = 0.1 and mutation rate = 0.5. We allowed up to 20 genes to be involved in each inverted translocation event, following a uniform distribution: i.e. there was a 5% chance for one gene to be involved, a 5% chance for two genes to be involved and so on.
Empirical genome data
All genome datasets were obtained from NCBI (ncbi.nlm.nih.gov) and are based on published studies. The dataset of 143 diverse bacterial and archaeal genomes is a subset of the 144 genomes used in a previous study8 to detect highways of gene sharing; two genomes in that study, Agrobacterium tumefaciens Cereon and C58, have since been merged into a single record in the NCBI database (as Agrobacterium fabrum str. C58). The 27 Escherichia coli and Shigella genomes and the eight Yersinia genomes, were taken respectively from Skippington and Ragan3 and Darling et al.5.
Phylogenetic trees
For each genome set, we used nine AF methods to compute a phylogenetic tree. These methods all into two groups: word-count ( , cvt, ffp, co-phylog, spaced) and match-length methods (gram, acs, kr, kmacs) (see Supplementary information). Six methods require a key parameter value to be set: k-mer length k for
, cvt, ffp, co-phylog, spaced) and match-length methods (gram, acs, kr, kmacs) (see Supplementary information). Six methods require a key parameter value to be set: k-mer length k for  , ffp and cvt; half-context length K for co-phylog; number of patterns n for spaced; and number of mismatches mm for kmacs. We tested wide range of values for these parameters (Supplementary Information) but for reasons of space report only the results obtained using the optimal parameter value, defined as that yielding the topology most congruent to the reference tree (see text). The distance matrix generated by each AF method was input into the neighbour-joining algorithm (neighbor in PHYLIP v3.69: evolution.genetics.washington.edu/phylip) to generate the corresponding tree.
, ffp and cvt; half-context length K for co-phylog; number of patterns n for spaced; and number of mismatches mm for kmacs. We tested wide range of values for these parameters (Supplementary Information) but for reasons of space report only the results obtained using the optimal parameter value, defined as that yielding the topology most congruent to the reference tree (see text). The distance matrix generated by each AF method was input into the neighbour-joining algorithm (neighbor in PHYLIP v3.69: evolution.genetics.washington.edu/phylip) to generate the corresponding tree.
Jackknifing technique
For the jackknifing procedure we adapted the approach used by Shi et al.46 and used a jackknife rate of 40% as suggested by the authors and our tests on different jackknife rates (see Supplementary information and Supplementary Figure S8). From each genome set we generated 100 pseudo-replicates: for each pseudo-replicate a randomly selected 100-nt fragment was deleted from each genome and this was iterated to a total of N times where N = (genome length × 0.4)/100, resulting in the deletion of (a different) 40% of each genome in each set. Then we generated a phylogenetic tree for each replicate using an AF method and calculated node support by comparing the pseudo-replicate trees and the test tree.
Assessment of accuracy
To assess the accuracy of each AF method we computed the Robinson-Foulds distance between a tree computed using that method (the “test tree”) and the corresponding reference tree, using treedist in PHYLIP v3.69. This distance represents the number of bipartitions that are present in only one of the two trees. To facilitate comparison of our results sequence sets (hence trees) of different sizes N, following Kupczok et al.53 we normalise this distance according to the maximum possible distance between two unrooted trees, 2(N − 3). We denote this normalised Robinson-Foulds distance as RF, with a value from 0 to 1 that can be interpreted as the proportion of false or missing bipartitions in the test-tree topology compared to the reference topology. When RF = 0 the test and reference topologies are identical, implying high accuracy for the method. Conversely, at RF = 1 no bipartition in the reference is recovered. A pair of randomly generated tree topologies of N taxa has a Robinson-Foulds distance that approaches the denominator for normalisation, 2(N − 3)54, i.e. when RF = 1. For the simulated data we used as reference the known tree (according to which the sequences were simulated) provided by Evolsimulator or ALF. For the E. coli - Shigella and 143-genome empirical datasets we used as reference the MRP supertrees from Skippington and Ragan3 and Beiko et al.8 respectively; in these cases RF does not inform directly on accuracy (as the true tree is unknown), but instead reflects the extent to which the AF method recovers the published topology. For the Yersinia dataset we used the consensus phylogenetic network5 as reference.
Computational scalability and runtime
Assessment of computational scalability was carried out using a high-performance distributed-memory computing cluster based on Intel Xeon ‘Haswell’ Cores 3.1 GHz. Comparative runtime analysis of alignment-free methods was done on Intel Xeon ‘Haswell’ Cores (E5-2667 v3) @ 3.1 GHz/boost 3.5 GHz (using a single processor and one thread).
Additional Information
How to cite this article: Bernard, G. et al. Alignment-free microbial phylogenomics under scenarios of sequence divergence, genome rearrangement and lateral genetic transfer. Sci. Rep. 6, 28970; doi: 10.1038/srep28970 (2016).
References
Tong, S. Y. et al. Genome sequencing defines phylogeny and spread of methicillin-resistant Staphylococcus aureus in a high transmission setting. Genome Res 25, 111–118, 10.1101/gr.174730.114 (2015).
Dunn, C. W. et al. Broad phylogenomic sampling improves resolution of the animal tree of life. Nature 452, 745–749, 10.1038/nature06614 (2008).
Skippington, E. & Ragan, M. A. Within-species lateral genetic transfer and the evolution of transcriptional regulation in Escherichia coli and Shigella. BMC Genomics 12, 532, 10.1186/1471-2164-12-532 (2011).
Jarvis, E. D. et al. Whole-genome analyses resolve early branches in the tree of life of modern birds. Science 346, 1320–1331, 10.1126/science.1253451 (2014).
Darling, A. E., Miklós, I. & Ragan, M. A. Dynamics of genome rearrangement in bacterial populations. PLoS Genet 4, e1000128, 10.1371/journal.pgen.1000128 (2008).
Chan, C. X. & Ragan, M. A. Next-generation phylogenomics. Biol Direct 8, 3, 10.1186/1745-6150-8-3 (2013).
Puigbò, P., Wolf, Y. I. & Koonin, E. V. The tree and net components of prokaryote evolution. Genome Biol Evol 2, 745–756, 10.1093/gbe/evq062 (2010).
Beiko, R. G., Harlow, T. J. & Ragan, M. A. Highways of gene sharing in prokaryotes. Proc Natl Acad Sci USA 102, 14332–14337, 10.1073/pnas.0504068102 (2005).
Stiller, J. W. Experimental design and statistical rigor in phylogenomics of horizontal and endosymbiotic gene transfer. BMC Evol Biol 11, 259, 10.1186/1471-2148-11-259 (2011).
Wong, K. M., Suchard, M. A. & Huelsenbeck, J. P. Alignment uncertainty and genomic analysis. Science 319, 473–476, 10.1126/science.1151532 (2008).
Shih, P. M. et al. Improving the coverage of the cyanobacterial phylum using diversity-driven genome sequencing. Proc Natl Acad Sci USA 110, 1053–1058, 10.1073/pnas.1217107110 (2013).
Wu, D. et al. A phylogeny-driven genomic encyclopaedia of Bacteria and Archaea. Nature 462, 1056–1060, 10.1038/nature08656 (2009).
Eyre, D. W. et al. A pilot study of rapid benchtop sequencing of Staphylococcus aureus and Clostridium difficile for outbreak detection and surveillance. BMJ Open 2, e001124, 10.1136/bmjopen-2012-001124 (2012).
Bonham-Carter, O., Steele, J. & Bastola, D. Alignment-free genetic sequence comparisons: a review of recent approaches by word analysis. Brief Bioinform 15, 890–905, 10.1093/bib/bbt052 (2014).
Haubold, B. Alignment-free phylogenetics and population genetics. Brief Bioinform 15, 407–418, 10.1093/bib/bbt083 (2014).
Vinga, S. & Almeida, J. Alignment-free sequence comparison-a review. Bioinformatics 19, 513–523 (2003).
Chan, C. X., Bernard, G., Poirion, O., Hogan, J. M. & Ragan, M. A. Inferring phylogenies of evolving sequences without multiple sequence alignment. Sci Rep 4, 6504, 10.1038/srep06504 (2014).
Felsenstein, J. Evolutionary trees from DNA sequences: a maximum likelihood approach. J Mol Evol 17, 368–376 (1981).
Huelsenbeck, J. P., Ronquist, F., Nielsen, R. & Bollback, J. P. Bayesian inference of phylogeny and its impact on evolutionary biology. Science 294, 2310–2314, 10.1126/science.1065889 (2001).
Huelsenbeck, J. P. & Ronquist, F. MRBAYES: Bayesian inference of phylogenetic trees. Bioinformatics 17, 754–755 (2001).
Fan, H., Ives, A. R., Surget-Groba, Y. & Cannon, C. H. An assembly and alignment-free method of phylogeny reconstruction from next-generation sequencing data. BMC Genomics 16, 522, 10.1186/s12864-015-1647-5 (2015).
Ren, J. et al. Inference of Markovian properties of molecular sequences from NGS data and applications to comparative genomics. Bioinformatics 32, 993–1000, 10.1093/bioinformatics/btv395 (2016).
Song, K. et al. Alignment-free sequence comparison based on next-generation sequencing reads. J Comput Biol 20, 64–79, 10.1089/cmb.2012.0228 (2013).
Miller, R. G. Jackknife - Review. Biometrika 61, 1–15 (1974).
Wan, L., Reinert, G., Sun, F. & Waterman, M. S. Alignment-free sequence comparison (II): theoretical power of comparison statistics. J Comput Biol 17, 1467–1490, 10.1089/cmb.2010.0056 (2010).
Yi, H. & Jin, L. Co-phylog: an assembly-free phylogenomic approach for closely related organisms. Nucleic Acids Res 41, e75, 10.1093/nar/gkt003 (2013).
Wang, H., Xu, Z., Gao, L. & Hao, B. A fungal phylogeny based on 82 complete genomes using the composition vector method. BMC Evol Biol 9, 195, 10.1186/1471-2148-9-195 (2009).
Jun, S. R., Sims, G. E., Wu, G. A. & Kim, S. H. Whole-proteome phylogeny of prokaryotes by feature frequency profiles: An alignment-free method with optimal feature resolution. Proc Natl Acad Sci USA 107, 133–138, 10.1073/pnas.0913033107 (2010).
Sims, G. E., Jun, S. R., Wu, G. A. & Kim, S. H. Alignment-free genome comparison with feature frequency profiles (FFP) and optimal resolutions. Proc Natl Acad Sci USA 106, 2677–2682, 10.1073/pnas.0813249106 (2009).
Sims, G. E. & Kim, S. H. Whole-genome phylogeny of Escherichia coli/Shigella group by feature frequency profiles (FFPs). Proc Natl Acad Sci USA 108, 8329–8334, 10.1073/pnas.1105168108 (2011).
Horwege, S. et al. Spaced words and kmacs: fast alignment-free sequence comparison based on inexact word matches. Nucleic Acids Res 42, W7–11, 10.1093/nar/gku398 (2014).
Leimeister, C. A., Boden, M., Horwege, S., Lindner, S. & Morgenstern, B. Fast alignment-free sequence comparison using spaced-word frequencies. Bioinformatics 30, 1991–1999, 10.1093/bioinformatics/btu177 (2014).
Ulitsky, I., Burstein, D., Tuller, T. & Chor, B. The average common substring approach to phylogenomic reconstruction. J Comput Biol 13, 336–350, 10.1089/cmb.2006.13.336 (2006).
Russell, D. J., Way, S. F., Benson, A. K. & Sayood, K. A grammar-based distance metric enables fast and accurate clustering of large sets of 16S sequences. BMC Bioinformatics 11, 601, 10.1186/1471-2105-11-601 (2010).
Leimeister, C. A. & Morgenstern, B. Kmacs: the k-mismatch average common substring approach to alignment-free sequence comparison. Bioinformatics 30, 2000–2008, 10.1093/bioinformatics/btu331 (2014).
Haubold, B., Pfaffelhuber, P., Domazet-Lošo, M. & Wiehe, T. Estimating mutation distances from unaligned genomes. J Comput Biol 16, 1487–1500, 10.1089/cmb.2009.0106 (2009).
Robinson, D. F. & Foulds, L. R. Comparison of phylogenetic trees. Math Biosci 53, 131–147, 10.1016/0025-5564(81)90043-2 (1981).
Beiko, R. G. & Charlebois, R. L. A simulation test bed for hypotheses of genome evolution. Bioinformatics 23, 825–831, 10.1093/bioinformatics/btm024 (2007).
Bryant, D. & Steel, M. Computing the distribution of a tree metric. IEEE/ACM Trans Comput Biol Bioinform 6, 420–426, 10.1109/TCBB.2009.32 (2009).
Ragan, M. A. Phylogenetic inference based on matrix representation of trees. Mol Phylogenet Evol 1, 53–58, 10.1016/1055-7903(92)90035-F (1992).
Ragan, M. A., Bernard, G. & Chan, C. X. Molecular phylogenetics before sequences: oligonucleotide catalogs as k-mer spectra. RNA Biol 11, 176–185, 10.4161/rna.27505 (2014).
Gordon, D. M., Clermont, O., Tolley, H. & Denamur, E. Assigning Escherichia coli strains to phylogenetic groups: multi-locus sequence typing versus the PCR triplex method. Environ Microbiol 10, 2484–2496, 10.1111/j.1462-2920.2008.01669.x (2008).
Ochman, H. & Selander, R. K. Standard reference strains of Escherichia coli from natural populations. J Bacteriol 157, 690–693, 0021-9193/84/020690-04$02.00/0 (1984).
Lecointre, G., Rachdi, L., Darlu, P. & Denamur, E. Escherichia coli molecular phylogeny using the incongruence length difference test. Mol Biol Evol 15, 1685–1695 (1998).
Farris, J. S., Albert, V. A., Källersjö, M., Lipscomb, D. & Kluge, A. G. Parsimony jackknifing outperforms neighbor-joining. Cladistics 12, 99–124 (1996).
Shi, J., Zhang, Y., Luo, H. & Tang, J. Using jackknife to assess the quality of gene order phylogenies. BMC Bioinformatics 11, 168, 10.1186/1471-2105-11-168 (2010).
Woese, C. R. & Fox, G. E. Phylogenetic structure of the prokaryotic domain: the primary kingdoms. Proc Natl Acad Sci USA 74, 5088–5090, 10.1073/pnas.74.11.5088 (1977).
Fuerst, J. A. & Sagulenko, E. Beyond the bacterium: planctomycetes challenge our concepts of microbial structure and function. Nat Rev Microbiol 9, 403–413, 10.1038/nrmicro2578 (2011).
Gunasinghe, U., Alahakoon, D. & Bedingfield, S. Extraction of high quality k-words for alignment-free sequence comparison. J Theor Biol 358, 31–51, 10.1016/j.jtbi.2014.05.016 (2014).
Dalquen, D. A., Anisimova, M., Gonnet, G. H. & Dessimoz, C. ALF–a simulation framework for genome evolution. Mol Biol Evol 29, 1115–1123, 10.1093/molbev/msr268 (2012).
Tavaré, S. Some probabilistic and statistical problems in the analysis of DNA sequences. Lectures on Mathematics in the Life Sciences 17, 57–86 (1986).
Beiko, R. G., Doolittle, W. F. & Charlebois, R. L. The impact of reticulate evolution on genome phylogeny. Syst Biol 57, 844–856, 10.1080/10635150802559265 (2008).
Kupczok, A., Schmidt, H. A. & von Haeseler, A. Accuracy of phylogeny reconstruction methods combining overlapping gene data sets. Algorithms Mol Biol 5, 37, 10.1186/1748-7188-5-37 (2010).
Bryant, D. & Steel, M. Computing the distribution of a tree metric. IEEE/ACM Trans. Comput. Biol. Bioinform. 6, 420–426, 10.1109/TCBB.2009.32 (2009).
Acknowledgements
We thank The University of Queensland and the James S. McDonnell Foundation for financial support. This work was supported by computational resources of the National Computational Infrastructure (NCI) National Facility systems through the NCI Merit Allocation Scheme (Project d85).
Author information
Authors and Affiliations
Contributions
G.B. implemented the analysis workflow and conducted the experiments, prepared all figures and tables and the first draft of the manuscript. G.B., C.X.C. and M.A.R. conceived the study, designed the experiments, analysed and interpreted the results. All authors prepared, wrote, reviewed, commented on and approved the final manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Bernard, G., Chan, C. & Ragan, M. Alignment-free microbial phylogenomics under scenarios of sequence divergence, genome rearrangement and lateral genetic transfer. Sci Rep 6, 28970 (2016). https://doi.org/10.1038/srep28970
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep28970
- Springer Nature Limited
This article is cited by
-
Exploring short k-mer profiles in cells and mobile elements from Archaea highlights the major influence of both the ecological niche and evolutionary history
BMC Genomics (2021)
-
Benchmarking of alignment-free sequence comparison methods
Genome Biology (2019)
-
Alignment-free sequence comparison: benefits, applications, and tools
Genome Biology (2017)












 at k = 7, supported by JK values. (b) Tree generated using
at k = 7, supported by JK values. (b) Tree generated using  at k = 9, supported by JK values.
at k = 9, supported by JK values.