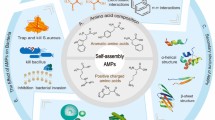
Abstract
Antimicrobial resistance is a major threat to human health, and this ‘invisible pandemic’ is a looming public health crisis. Accordingly, both broad-spectrum and selective antimicrobial agents that do not induce resistance are urgently required. Synthetic peptide-polymers and their mimics and in particular structurally nano-engineered antimicrobial peptide-polymers (SNAPPs) are antimicrobial materials with clinical potential as novel therapeutics to combat antimicrobial resistance due to their inherent biodegradability, biocompatibility and tuneable cytocompatibility. Macromolecular design in conjunction with rational monomer composition can direct their architecture, self-assembly and chemical behaviour, ultimately guiding the choice of appropriate application within the biomedical field. This Review focuses on several facets of antimicrobial peptide-polymers including their synthesis, diversity, physicochemical properties and bacteria-killing mechanisms. We discuss current strategies in the antimicrobial field that improve antibacterial activity in the context of their current and potential application to peptide-polymers. Further, different strategies to enhance the antibacterial activity of peptide-polymers are discussed, along with burgeoning developments in medical applications. The challenges of future applications of synthetic peptide branched polymers in biomedical engineering are highlighted.
Key points
-
Infectious diseases caused by antibiotic-resistant pathogens are one of the greatest challenges to global health care.
-
Synthetic peptide branched polymers produced by ring-opening polymerization of N-carboxyanhydride amino acids have exquisite antimicrobial efficacy while maintaining the peptide-polymer advantages of being highly biocompatible, biodegradable materials with highly potent antimicrobial activity.
-
Structurally nano-engineered antimicrobial peptide-polymers (SNAPPs) are an example of these synthetic peptide branched polymers.
-
SNAPPs, and other peptide-polymers, possess multiple mechanisms of action for killing bacteria, which reduces the ability of bacteria to develop resistance.
-
Peptide-polymers have unique synthesis advantages as their production is more cost-effective than antimicrobial peptides made by solid-phase peptide synthesis. They can also be made on a large scale and can incorporate a variety of post-modifications.
-
Synthetic peptide branched polymers might be used for drug delivery, gene therapy, tissue engineering, vaccine therapeutics and other biomedical applications.





Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.References
Cook, M. A. & Wright, G. D. The past, present, and future of antibiotics. Sci. Transl Med. 14, eabo7793 (2022).
Chremos, A. & Douglas, J. F. Influence of branching on the configurational and dynamical properties of entangled polymer melts. Polymers 11, 1045 (2019).
Zasloff, M. Antimicrobial peptides of multicellular organisms: my perspective. Adv. Exp. Med. Biol. 1117, 3–6 (2019).
Brogden, K. A. Antimicrobial peptides: pore formers or metabolic inhibitors in bacteria? Nat. Rev. Microbiol. 3, 238–250 (2005).
Steiner, H., Hultmark, D., Engström, A., Bennich, H. & Boman, H. G. Sequence and specificity of two antibacterial proteins involved in insect immunity. Nature 292, 246–248 (1981).
Zasloff, M. Magainins, a class of antimicrobial peptides from Xenopus skin: isolation, characterization of two active forms, and partial cDNA sequence of a precursor. Proc. Natl Acad. Sci. USA 84, 5449–5453 (1987).
Selsted, M. E. et al. Primary structures of six antimicrobial peptides of rabbit peritoneal neutrophils. J. Biol. Chem. 260, 4579–4584 (1985).
Li, W., Separovic, F., O’Brien-Simpson, N. M. & Wade, J. D. Chemically modified and conjugated antimicrobial peptides against superbugs. Chem. Soc. Rev. 50, 4932–4973 (2021).
Rasines Mazo, A. et al. Ring opening polymerization of α-amino acids: advances in synthesis, architecture and applications of polypeptides and their hybrids. Chem. Soc. Rev. 49, 4737–4834 (2020).
Shao, N. et al. Heterochiral β-peptide polymers combating multidrug-resistant cancers effectively without inducing drug resistance. J. Am. Chem. Soc. 144, 7283–7294 (2022).
Cao, J. et al. Interfacial ring-opening polymerization of amino-acid-derived N-thiocarboxyanhydrides toward well-defined polypeptides. ACS Macro Lett. 6, 836–840 (2017).
Tian, Z. Y., Zhang, Z., Wang, S. & Lu, H. A moisture-tolerant route to unprotected α/β-amino acid N-carboxyanhydrides and facile synthesis of hyperbranched polypeptides. Nat. Commun. 12, 5810 (2021).
Deming, T. J. Synthesis of side-chain modified polypeptides. Chem. Rev. 116, 786–808 (2016).
Jia, F. et al. d-amino acid substitution enhances the stability of antimicrobial peptide polybia-CP. Acta Biochim. Biophys. Sin. 49, 916–925 (2017).
Kirshenbaum, K. et al. Sequence-specific polypeptoids: a diverse family of heteropolymers with stable secondary structure. Proc. Natl Acad. Sci. USA 95, 4303–4308 (1998).
Song, Z. et al. Synthesis of polypeptides via bioinspired polymerization of in situ purified N-carboxyanhydrides. Proc. Natl Acad. Sci. USA 116, 10658–10663 (2019).
Grazon, C. et al. Aqueous ring‐opening polymerization‐induced self‐assembly (ROPISA) of N‐carboxyanhydrides. Angew. Chem. 132, 632–636 (2020).
Gradisar, S., Zagar, E. & Pahovnik, D. Ring-opening polymerization of N-carboxyanhydrides initiated by a hydroxyl group. ACS Macro Lett. 6, 637–640 (2017).
Obeid, R. et al. The behavior of poly (amino acids) containing l‐cysteine and their block copolymers with poly (ethylene glycol) on gold surfaces. J. Polym. Sci. A 52, 248–257 (2014).
Ji, S., Xu, L., Fu, X., Sun, J. & Li, Z. Light-and metal ion-induced self-assembly and reassembly based on block copolymers containing a photoresponsive polypeptide segment. Macromolecules 52, 4686–4693 (2019).
Liu, S.-T., Tuan-Mu, H.-Y., Hu, J.-J. & Jan, J.-S. Genipin cross-linked PEG-block-poly (l-lysine)/disulfide-based polymer complex micelles as fluorescent probes and pH-/redox-responsive drug vehicles. RSC Adv. 5, 87098–87107 (2015).
Lv, S. et al. Co-delivery of doxorubicin and paclitaxel by PEG-polypeptide nanovehicle for the treatment of non-small cell lung cancer. Biomaterials 35, 6118–6129 (2014).
Wang, X. et al. Facile synthesis of helical multiblock copolypeptides: minimal side reactions with accelerated polymerization of N-carboxyanhydrides. ACS Macro Lett. 8, 1517–1521 (2019).
Veronese, F. M. Peptide and protein PEGylation: a review of problems and solutions. Biomaterials 22, 405–417 (2001).
Webber, M. J. et al. Supramolecular PEGylation of biopharmaceuticals. Proc. Natl Acad. Sci. USA 113, 14189–14194 (2016).
Le Fer, G. et al. Design and self-assembly of PBLG-b-ELP hybrid diblock copolymers based on synthetic and elastin-like polypeptides. Org. Biomol. Chem. 15, 10095–10104 (2017).
Gradišar, Š., Žagar, E. & Pahovnik, D. Hybrid block copolymers of polyesters/polycarbonates and polypeptides synthesized via one-pot sequential ring-opening polymerization. Polym. Chem. 9, 4764–4771 (2018).
Xu, H. et al. Dual-responsive mPEG-PLGA-PGlu hybrid-core nanoparticles with a high drug loading to reverse the multidrug resistance of breast cancer: an in vitro and in vivo evaluation. Acta Biomater. 16, 156–168 (2015).
Arimura, H., Ohya, Y. & Ouchi, T. The formation of biodegradable polymeric micelles from newly synthesized poly (aspartic acid)‐block‐polylactide AB‐type diblock copolymers. Macromol. Rapid Commun. 25, 743–747 (2004).
Tinajero-Diaz, E., de Ilarduya, A. M. & Muñoz-Guerra, S. Synthesis and properties of diblock copolymers of ω-pentadecalactone and α-amino acids. Eur. Polym. J. 116, 169–179 (2019).
Jacobs, J. et al. Facile synthesis of fluorescent latex nanoparticles with selective binding properties using amphiphilic glycosylated polypeptide surfactants. Macromolecules 47, 7303–7310 (2014).
Wang, Y. & Ling, J. Synthetic protocols toward polypeptide conjugates via chain end functionalization after RAFT polymerization. RSC Adv. 5, 18546–18553 (2015).
Levit, M. et al. Synthesis and characterization of well-defined poly (2-deoxy-2-methacrylamido-d-glucose) and its biopotential block copolymers via RAFT and ROP polymerization. Eur. Polym. J. 105, 26–37 (2018).
Li, S.-L. et al. Phosphatase-triggered cell-selective release of a Pt (IV)-backboned prodrug-like polymer for an improved therapeutic index. Biomater. Sci. 5, 1558–1566 (2017).
Quadir, M. A. et al. PEG–polypeptide block copolymers as pH-responsive endosome-solubilizing drug nanocarriers. Mol. Pharmaceut. 11, 2420–2430 (2014).
Negri, G. E. & Deming, T. J. Protein complexation and pH dependent release using boronic acid containing PEG‐polypeptide copolymers. Macromol. Biosci. 17, 1600136 (2017).
Chan, N. J.-A. et al. Spider-silk inspired polymeric networks by harnessing the mechanical potential of β-sheets through network guided assembly. Nat. Commun. 11, 1630 (2020).
Shi, S. Y. et al. Polypeptide‐b‐poly (phenyl isocyanide) hybrid rod–rod copolymers: one‐pot synthesis, self‐assembly, and cell imaging. Macromol. Rapid Commun. 36, 1511–1520 (2015).
Schmid, T. E. et al. Aluminum‐catalyzed one‐pot synthesis of polyester‐b‐polypeptide block copolymers by ring‐opening polymerization. Macromol. Chem. Phys. 220, 1900040 (2019).
Tappertzhofen, K. et al. Poly‐l‐lysine–poly [HPMA] block copolymers obtained by RAFT polymerization as polyplex‐transfection reagents with minimal toxicity. Macromol. Biosci. 15, 1159–1173 (2015).
Hou, Y. et al. Therapeutic protein PEPylation: the helix of nonfouling synthetic polypeptides minimizes antidrug antibody generation. ACS Cent. Sci. 5, 229–236 (2019).
Hou, Y., Yuan, J., Zhou, Y., Yu, J. & Lu, H. A concise approach to site-specific topological protein–poly (amino acid) conjugates enabled by in situ-generated functionalities. J. Am. Chem. Soc. 138, 10995–11000 (2016).
Hou, Y. et al. Macrocyclization of interferon–poly (α-amino acid) conjugates significantly improves the tumor retention, penetration, and antitumor efficacy. J. Am. Chem. Soc. 140, 1170–1178 (2018).
Fu, L., Liu, L., Ruan, Z., Zhang, H. & Yan, L. Folic acid targeted pH-responsive amphiphilic polymer nanoparticles conjugated with near infrared fluorescence probe for imaging-guided drug delivery. RSC Adv. 6, 40312–40322 (2016).
Wang, H. et al. Enzyme-activatable interferon–poly (α-amino acid) conjugates for tumor microenvironment potentiation. Biomacromolecules 20, 3000–3008 (2019).
Agouridas, V. et al. Native chemical ligation and extended methods: mechanisms, catalysis, scope, and limitations. Chem. Rev. 119, 7328–7443 (2019).
Kulkarni, S. S., Sayers, J., Premdjee, B. & Payne, R. J. Rapid and efficient protein synthesis through expansion of the native chemical ligation concept. Nat. Rev. Chem. 2, 0122 (2018).
Conibear, A. C., Watson, E. E., Payne, R. J. & Becker, C. F. W. Native chemical ligation in protein synthesis and semi-synthesis. Chem. Soc. Rev. 47, 9046–9068 (2018).
Morell, M. & Puiggalí, J. Hybrid block copolymers constituted by peptides and synthetic polymers: an overview of synthetic approaches, supramolecular behavior and potential applications. Polymers 5, 188–224 (2013).
You, Y., Chen, Y., Hua, C. & Dong, C. M. Synthesis and thermoreversible gelation of dendron‐like polypeptide/linear poly (ε‐caprolactone)/dendron‐like polypeptide triblock copolymers. J. Polym. Sci. A 48, 709–718 (2010).
Lam, S. J. et al. Combating multidrug-resistant Gram-negative bacteria with structurally nanoengineered antimicrobial peptide polymers. Nat. Microbiol. 1, 16162 (2016).
Habraken, G. J., Peeters, M., Dietz, C. H., Koning, C. E. & Heise, A. How controlled and versatile is N-carboxy anhydride (NCA) polymerization at 0 C? Effect of temperature on homo-, block-and graft (co) polymerization. Polym. Chem. 1, 514–524 (2010).
Wibowo, S. H. et al. Assembly of free‐standing polypeptide films via the synergistic combination of hyperbranched macroinitiators, the grafting‐from approach, and cross‐chain termination. Adv. Mater. 25, 4619–4624 (2013).
Blencowe, A., Tan, J. F., Goh, T. K. & Qiao, G. G. Core cross-linked star polymers via controlled radical polymerisation. Polymer 50, 5–32 (2009).
Gao, H. & Matyjaszewski, K. Synthesis of functional polymers with controlled architecture by CRP of monomers in the presence of cross-linkers: from stars to gels. Prog. Polym. Sci. 34, 317–350 (2009).
Sulistio, A., Widjaya, A., Blencowe, A., Zhang, X. & Qiao, G. Star polymers composed entirely of amino acid building blocks: a route towards stereospecific, biodegradable and hierarchically functionalized stars. Chem. Commun. 47, 1151–1153 (2011).
Sulistio, A., Blencowe, A., Widjaya, A., Zhang, X. & Qiao, G. Development of functional amino acid-based star polymers. Polym. Chem. 3, 224–234 (2012).
Sulistio, A. et al. Folic acid conjugated amino acid-based star polymers for active targeting of cancer cells. Biomacromolecules 12, 3469–3477 (2011).
Ren, J. M. et al. Star polymers. Chem. Rev. 116, 6743–6836 (2016).
Mazo, A. R. et al. Ring opening polymerization of α-amino acids: advances in synthesis, architecture and applications of polypeptides and their hybrids. Chem. Soc. Rev. 49, 4737–4834 (2020).
Deming, T. J. Preparation and development of block copolypeptide vesicles and hydrogels for biological and medical applications. Wiley Interdisc. Rev. Nanomed. Nanobiotechnol. 6, 283–297 (2014).
Pranantyo, D., Xu, L. Q., Hou, Z., Kang, E.-T. & Chan-Park, M. B. Increasing bacterial affinity and cytocompatibility with four-arm star glycopolymers and antimicrobial α-polylysine. Polym. Chem. 8, 3364–3373 (2017).
Chang, X., Liu, L., Guan, Y. & Dong, C. M. Disulfide‐centered star‐shaped polypeptide‐PEO block copolymers for reduction‐triggered drug release. J. Polym. Sci. A 52, 2000–2010 (2014).
Pu, Y., Zhang, L., Zheng, H., He, B. & Gu, Z. Synthesis and drug release of star‐shaped poly (benzyl l‐aspartate)‐block‐poly (ethylene glycol) copolymers with POSS cores. Macromol. Biosci. 14, 289–297 (2014).
Dürr, C. J., Hlalele, L., Kaiser, A., Brandau, S. & Barner-Kowollik, C. Mild and efficient modular synthesis of poly (acrylonitrile-co-butadiene) block and miktoarm star copolymer architectures. Macromolecules 46, 49–62 (2013).
Müllner, M. Molecular polymer brushes in nanomedicine. Macromol. Chem. Phys. 217, 2209–2222 (2016).
Yu, H. et al. Poly (l-lysine)-graft-chitosan copolymers: synthesis, characterization, and gene transfection effect. Biomacromolecules 8, 1425–1435 (2007).
Chi, P., Wang, J. & Liu, C. Synthesis and characterization of polycationic chitosan-graft-poly (l-lysine). Mater. Lett. 62, 147–150 (2008).
Li, P. et al. Cationic peptidopolysaccharides show excellent broad‐spectrum antimicrobial activities and high selectivity. Adv. Mater. 24, 4130–4137 (2012).
Perdih, P. et al. Synthesis of chitosan-graft-poly (sodium-l-glutamate) for preparation of protein nanoparticles. Cellulose 21, 3469–3485 (2014).
Bielawski, C. W. & Grubbs, R. H. Highly efficient ring‐opening metathesis polymerization (ROMP) using new ruthenium catalysts containing N‐heterocyclic carbene ligands. Angew. Chem. Int. Edn Engl. 39, 2903–2906 (2000).
Lummiss, J. A., Ireland, B. J., Sommers, J. M. & Fogg, D. E. Amine‐mediated degradation in olefin metathesis reactions that employ the second‐generation Grubbs catalyst. ChemCatChem 6, 459–463 (2014).
Lu, H., Wang, J., Lin, Y. & Cheng, J. One-pot synthesis of brush-like polymers via integrated ring-opening metathesis polymerization and polymerization of amino acid N-carboxyanhydrides. J. Am. Chem. Soc. 131, 13582–13583 (2009).
Engler, A. C., Lee, H. I. & Hammond, P. T. Highly efficient “grafting onto” a polypeptide backbone using click chemistry. Angew. Chem. 121, 9498–9502 (2009).
Zhou, C. et al. Antibacterial polypeptide-grafted chitosan-based nanocapsules as an “armed” carrier of anticancer and antiepileptic drugs. ACS Macro Lett. 2, 1021–1025 (2013).
Fu, X., Ma, Y., Sun, J. & Li, Z. Biodegradable thermal-and redox-responsive poly (l-glutamate) with Y-shaped oligo (ethylene glycol) side-chain and tunable phase transition temperature. RSC Adv. 6, 70243–70250 (2016).
Mookherjee, N., Anderson, M. A., Haagsman, H. P. & Davidson, D. J. Antimicrobial host defence peptides: functions and clinical potential. Nat. Rev. Drug Discov. 19, 311–332 (2020).
Hancock, R. E. & Sahl, H.-G. Antimicrobial and host-defense peptides as new anti-infective therapeutic strategies. Nat. Biotechnol. 24, 1551–1557 (2006).
Cui, Z. et al. Molecular engineering of antimicrobial peptide (AMP)–polymer conjugates. Biomater. Sci. 9, 5069–5091 (2021).
Silva, A. R. P. et al. Recent advances in the design of antimicrobial peptide conjugates. J. Mater. Chem. B 10, https://doi.org/10.1039/D1TB02757C (2022).
Han, Y., Zhang, M., Lai, R. & Zhang, Z. Chemical modifications to increase the therapeutic potential of antimicrobial peptides. Peptides 146, 170666 (2021).
Hamley, I. W. PEG–peptide conjugates. Biomacromolecules 15, 1543–1559 (2014).
Sun, H. et al. Synthesis, self-assembly, and biomedical applications of antimicrobial peptide–polymer conjugates. Biomacromolecules 19, 1701–1720 (2018).
Morris, C. J. et al. Pegylation of antimicrobial peptides maintains the active peptide conformation, model membrane interactions, and antimicrobial activity while improving lung tissue biocompatibility following airway delivery. Antimicrob. Agents Chemother. 56, 3298–3308 (2012).
Yu, W. et al. PEGylation of the antimicrobial peptide PG-1: a link between propensity for nanostructuring and capacity of the antitrypsin hydrolytic ability. J. Med. Chem. 64, 10469–10481 (2021).
Pelegri-O’Day, E. M. et al. PEG analogs synthesized by ring-opening metathesis polymerization for reversible bioconjugation. Bioconjug. Chem. 29, 3739–3745 (2018).
Hou, Z. et al. Nanoparticles of short cationic peptidopolysaccharide self-assembled by hydrogen bonding with antibacterial effect against multidrug-resistant bacteria. ACS Appl. Mater. Interf. 9, 38288–38303 (2017).
Chen, Y. et al. Design and synthesis of biocompatible, hemocompatible, and highly selective antimicrobial cationic peptidopolysaccharides via click chemistry. Biomacromolecules 20, 2230–2240 (2019).
Pranantyo, D., Xu, L. Q., Kang, E.-T., Mya, M. K. & Chan-Park, M. B. Conjugation of polyphosphoester and antimicrobial peptide for enhanced bactericidal activity and biocompatibility. Biomacromolecules 17, 4037–4044 (2016).
Palermo, E. F. & Kuroda, K. Chemical structure of cationic groups in amphiphilic polymethacrylates modulates the antimicrobial and hemolytic activities. Biomacromolecules 10, 1416–1428 (2009).
Palermo, E. F., Lee, D.-K., Ramamoorthy, A. & Kuroda, K. Role of cationic group structure in membrane binding and disruption by amphiphilic copolymers. J. Phys. Chem. B 115, 366–375 (2011).
Wang, H. et al. Antibacterial activity of geminized amphiphilic cationic homopolymers. Langmuir 31, 13469–13477 (2015).
Ergene, C., Yasuhara, K. & Palermo, E. F. Biomimetic antimicrobial polymers: recent advances in molecular design. Polym. Chem. 9, 2407–2427 (2018).
Zhang, R., Zheng, N., Song, Z., Yin, L. & Cheng, J. The effect of side-chain functionality and hydrophobicity on the gene delivery capabilities of cationic helical polypeptides. Biomaterials 35, 3443–3454 (2014).
Rothbard, J. B., Jessop, T. C. & Wender, P. A. Adaptive translocation: the role of hydrogen bonding and membrane potential in the uptake of guanidinium-rich transporters into cells. Adv. Drug Delivery Rev. 57, 495–504 (2005).
Song, Z. et al. Polypeptides with quaternary phosphonium side chains: synthesis, characterization, and cell-penetrating properties. Biomacromolecules 15, 1491–1497 (2014).
Kuroda, K., Caputo, G. A. & DeGrado, W. F. The role of hydrophobicity in the antimicrobial and hemolytic activities of polymethacrylate derivatives. Chem. Eur. J. 15, 1123–1133 (2009).
Chen, Q. et al. Homo-catiomer integration into PEGylated polyplex micelle from block-catiomer for systemic anti-angiogenic gene therapy for fibrotic pancreatic tumors. Biomaterials 33, 4722–4730 (2012).
Ikeda, T., Hirayama, H., Yamaguchi, H., Tazuke, S. & Watanabe, M. Polycationic biocides with pendant active groups: molecular weight dependence of antibacterial activity. Antimicrob. Agents Chemother. 30, 132–136 (1986).
Kanazawa, A., Ikeda, T. & Endo, T. Polymeric phosphonium salts as a novel class of cationic biocides. II. Effects of counter anion and molecular weight on antibacterial activity of polymeric phosphonium salts. J. Polym. Sci. A 31, 1441–1447 (1993).
Lienkamp, K., Kumar, K.-N., Som, A., Nüsslein, K. & Tew, G. N. “Doubly selective” antimicrobial polymers: how do they differentiate between bacteria? Chem. Eur. J. 15, 11710–11714 (2009).
Jiang, Y., Zheng, W., Kuang, L., Ma, H. & Liang, H. Hydrophilic phage-mimicking membrane active antimicrobials reveal nanostructure-dependent activity and selectivity. ACS Infect. Dis. 3, 676–687 (2017).
Yin, L. et al. Reconfiguring the architectures of cationic helical polypeptides to control non-viral gene delivery. Biomaterials 34, 2340–2349 (2013).
Namivandi-Zangeneh, R. et al. The effects of polymer topology and chain length on the antimicrobial activity and hemocompatibility of amphiphilic ternary copolymers. Polym. Chem. 9, 1735–1744 (2018).
Priftis, D. et al. Self-assembly of α-helical polypeptides driven by complex coacervation. Angew. Chem. Int. Edn Engl. 54, 11128–11132 (2015).
Xiong, M. et al. Helical antimicrobial polypeptides with radial amphiphilicity. Proc. Natl Acad. Sci. USA 112, 13155–13160 (2015).
Lee, M. W. et al. Interactions between membranes and “metaphilic” polypeptide architectures with diverse side-chain populations. ACS Nano 11, 2858–2871 (2017).
Melo, M. N., Ferre, R. & Castanho, M. A. Antimicrobial peptides: linking partition, activity and high membrane-bound concentrations. Nat. Rev. Microbiol. 7, 245–250 (2009).
Lesiuk, M., Paduszyńska, M. & Greber, K. E. Synthetic antimicrobial immunomodulatory peptides: ongoing studies and clinical trials. Antibiotics 11, 1062 (2022).
Silva, T. & Gomes, M. S. Immuno-stimulatory peptides as a potential adjunct therapy against intra-macrophagic pathogens. Molecules 22, 1297 (2017).
Konai, M. M., Bhattacharjee, B., Ghosh, S. & Haldar, J. Recent progress in polymer research to tackle infections and antimicrobial resistance. Biomacromolecules 19, 1888–1917 (2018).
Mohamed, Y. F., Abou-Shleib, H. M., Khalil, A. M., El-Guink, N. M. & El-Nakeeb, M. A. Membrane permeabilization of colistin toward pan-drug resistant Gram-negative isolates. Braz. J. Microbiol. 47, 381–388 (2016).
Hasan, J., Crawford, R. J. & Ivanova, E. P. Antibacterial surfaces: the quest for a new generation of biomaterials. Trends Biotechnol. 31, 295–304 (2013).
Sharma, S., Sahoo, N. & Bhunia, A. Antimicrobial peptides and their pore/ion channel properties in neutralization of pathogenic microbes. Curr. Top. Med. Chem. 16, 46–53 (2016).
Roy, S. et al. Antimicrobial mechanisms of biomaterials: from macro to nano. Biomater. Sci. 10, 4392–4423 (2022).
Kohanski, M. A., Dwyer, D. J. & Collins, J. J. How antibiotics kill bacteria: from targets to networks. Nat. Rev. Microbiol. 8, 423–435 (2010).
Lam, S. J. et al. Bionano interaction study on antimicrobial star-shaped peptide polymer nanoparticles. ACS Appl. Mater. Interf. 8, 33446–33456 (2016).
Zhang, Y. et al. Theoretical insights into the interactions between star-shaped antimicrobial polypeptides and bacterial membranes. Langmuir 34, 13438–13448 (2018).
Chen, Y.-F. et al. Star-shaped polypeptides exhibit potent antibacterial activities. Nanoscale 11, 11696–11708 (2019).
Wong, E. H. H. et al. Modulating antimicrobial activity and mammalian cell biocompatibility with glucosamine-functionalized star polymers. Biomacromolecules 17, 1170–1178 (2016).
Liu, H. et al. Efficient synthesis and excellent antimicrobial activity of star-shaped cationic polypeptides with improved biocompatibility. Biomater. Sci. 9, 2721–2731 (2021).
Zhou, C. et al. High potency and broad-spectrum antimicrobial peptides synthesized via ring-opening polymerization of α-amino acid-N-carboxyanhydrides. Biomacromolecules 11, 60–67 (2010).
Ding, X. et al. A macromolecule reversing antibiotic resistance phenotype and repurposing drugs as potent antibiotics. Adv. Sci. 7, 2001374 (2020).
Bai, S. et al. A polymeric approach toward resistance-resistant antimicrobial agent with dual-selective mechanisms of action. Adv. Healthc. Mater. 7, eabc9917 (2021).
Tan, J., Zhao, Y., Hedrick, J. L. & Yang, Y. Y. Effects of hydrophobicity on antimicrobial activity, selectivity, and functional mechanism of guanidinium-functionalized polymers. Sci. Adv. 7, 2100482 (2021).
Shirbin, S. J. et al. Architectural effects of star‐shaped “structurally nanoengineered antimicrobial peptide polymers” (SNAPPs) on their biological activity. Adv. Healthc. Mater. 7, 1800627 (2018).
Li, W. et al. Star-peptide polymers are multi-drug-resistant Gram-positive bacteria killers. ACS Appl. Mater. Interf. 14, 25025–25041 (2022).
Lázár, V. et al. Antibiotic-resistant bacteria show widespread collateral sensitivity to antimicrobial peptides. Nat. Microbiol. 3, 718–731 (2018).
Wyrsta, M. D., Cogen, A. L. & Deming, T. J. A parallel synthetic approach for the analysis of membrane interactive copolypeptides. J. Am. Chem. Soc. 123, 12919–12920 (2001).
Kuroki, A., Tay, J., Lee, G. H. & Yang, Y. Y. Broad-spectrum antiviral peptides and polymers. Adv. Healthc. Mater. 10, e2101113 (2021).
Ito, M. et al. Inhibitory effect of dextran sulfate and heparin on the replication of human immunodeficiency virus (HIV) in vitro. Antivir. Res. 7, 361–367 (1987).
Huskens, D., Vermeire, K., Profy, A. T. & Schols, D. The candidate sulfonated microbicide, PRO 2000, has potential multiple mechanisms of action against HIV-1. Antivir. Res. 84, 38–47 (2009).
Kim, H.-O. et al. Reactive oxygen species-regulating polymersome as an antiviral agent against influenza virus. Small 13, 1700818 (2017).
Roemer, T. & Krysan, D. J. Antifungal drug development: challenges, unmet clinical needs, and new approaches. Cold Spring Harb. Perspect. Med. 4, a019703 (2014).
Campoy, S. & Adrio, J. L. Antifungals. Biochem. Pharmacol. 133, 86–96 (2017).
Fernández de Ullivarri, M., Arbulu, S., Garcia-Gutierrez, E. & Cotter, P. D. Antifungal peptides as therapeutic agents. Front. Cell. Infect. Microbiol. 10, 105 (2020).
Ramamourthy, G., Park, J., Seo, C., Vogel, H. J. & Park, Y. Antifungal and antibiofilm activities and the mechanism of action of repeating lysine–tryptophan peptides against Candida albicans. Microorganisms 8, 758 (2020).
Yang, H., Zhao, J., Yan, M., Pispas, S. & Zhang, G. Nylon 3 synthesized by ring opening polymerization with a metal-free catalyst. Polym. Chem. 2, 2888–2892, (2011).
Liu, R. et al. Nylon-3 polymers with selective antifungal activity. J. Am. Chem. Soc. 135, 5270–5273 (2013).
Liu, R. et al. Nylon-3 polymers active against drug-resistant Candida albicans biofilms. J. Am. Chem. Soc. 137, 2183–2186 (2015).
Ejim, L. et al. Combinations of antibiotics and nonantibiotic drugs enhance antimicrobial efficacy. Nat. Chem. Biol. 7, 348–350 (2011).
Si, Z. et al. A glycosylated cationic block poly(β-peptide) reverses intrinsic antibiotic resistance in all ESKAPE Gram-negative bacteria. Angew. Chem. Int. Edn Engl. 59, 6819–6826 (2020).
Thappeta, K. R. V., Vikhe, Y. S., Yong, A. M. H., Chan-Park, M. B. & Kline, K. A. Combined efficacy of an antimicrobial cationic peptide polymer with conventional antibiotics to combat multidrug-resistant pathogens. ACS Infect. Dis. 6, 1228–1237 (2020).
Zhen, J.-B., Kang, P.-W., Zhao, M.-H. & Yang, K.-W. Silver nanoparticle conjugated star PCL-b-AMPs copolymer as nanocomposite exhibits efficient antibacterial properties. Bioconjug. Chem. 31, 51–63 (2019).
Ekladious, I., Colson, Y. L. & Grinstaff, M. W. Polymer–drug conjugate therapeutics: advances, insights and prospects. Nat. Rev. Drug Discov. 18, 273–294 (2019).
Gao, J., Wang, M., Wang, F. & Du, J. Synthesis and mechanism insight of a peptide-grafted hyperbranched polymer nanosheet with weak positive charges but excellent intrinsically antibacterial efficacy. Biomacromolecules 17, 2080–2086 (2016).
Chin, W. et al. A macromolecular approach to eradicate multidrug resistant bacterial infections while mitigating drug resistance onset. Nat. Commun. 9, 917 (2018).
Eom, K. H. et al. Synthetic polypeptides with cationic arginine moieties showing high antimicrobial activity in similar mineral environments to blood plasma. Polymers 14, 1868 (2022).
Galanakou, C., Dhumal, D. & Peng, L. Amphiphilic dendrimers against antibiotic resistance: light at the end of the tunnel? Biomater. Sci. 11, https://doi.org/10.1039/d2bm01878k (2023).
Si, Z., Pethe, K. & Chan-Park, M. B. Chemical basis of combination therapy to combat antibiotic resistance. JACS Au. 3, 276–292 (2023).
Assoni, L. et al. Resistance mechanisms to antimicrobial peptides in gram-positive bacteria. Front. Microbiol. 11, 593215 (2020).
Yu, G., Baeder, D. Y., Regoes, R. R. & Rolff, J. Combination effects of antimicrobial peptides. Antimicrob. Agents Chemother. 60, 1717–1724 (2016).
Yu, G., Baeder, D. Y., Regoes, R. R. & Rolff, J. Predicting drug resistance evolution: insights from antimicrobial peptides and antibiotics. Proc. Biol. Sci. 285, 20172687 (2018).
Liu, Y. & Yin, L. α-Amino acid N-carboxyanhydride (NCA)-derived synthetic polypeptides for nucleic acids delivery. Adv. Drug Deliv. Rev. 171, 139–163 (2021).
Zhu, R. et al. Spherical α-helical polypeptide-mediated E2F1 silencing against myocardial ischemia-reperfusion injury (MIRI). Biomater. Sci. 10, 6258–6266 (2022).
Bauer, T. A. et al. Photocleavable core cross-linked polymeric micelles of polypept(o)ides and ruthenium(II) complexes. J. Mater. Chem. B 9, 8211–8223 (2021).
Hasannia, M. et al. Synthesis of doxorubicin-loaded peptosomes hybridized with gold nanorod for targeted drug delivery and CT imaging of metastatic breast cancer. J. Nanobiotechnol. 20, 391 (2022).
Zhang, Q. et al. Phenylboronic acid-functionalized copolypeptides: facile synthesis and responsive dual anticancer drug release. Biomacromolecules 23, 2989–2998 (2022).
Mokrus, M. & Menzel, H. Thermoresponsive glycopolypeptide containing block copolymers, particle formation, and lectin interaction. Macromol. Biosci. 22, e2100518 (2022).
Dong, L. et al. Poly(l-cysteine) peptide amphiphile derivatives containing disulfide bonds: synthesis, self-assembly-induced β-sheet nanostructures, pH/reduction dual response, and drug release. Biomacromolecules 22, 5374–5381 (2021).
Pottanam Chali, S., Hüwel, S., Rentmeister, A. & Ravoo, B. J. Self-assembled cationic polypeptide supramolecular nanogels for intracellular DNA delivery. Chemistry 27, 12198–12206 (2021).
Kimmins, S. D. et al. Antimicrobial and degradable triazolinedione (TAD) crosslinked polypeptide hydrogels. J. Mater. Chem. B 9, 5456–5464 (2021).
Augustine, R. et al. Multi-stimuli-responsive nanomicelles fabricated using synthetic polymer polylysine conjugates for tumor microenvironment dependent drug delivery. J. Mater. Chem. B 8, 5745–5755 (2020).
Zhou, J., Li, L., Wang, W., Zhao, Y. & Feng, S. pH-responsive polymeric vesicles from branched copolymers. RSC Adv. 9, 41031–41037 (2019).
Sang, X., Yang, Q., Wen, Q., Zhang, L. & Ni, C. Preparation and controlled drug release ability of the poly[N-isopropylacryamide-co-allyl poly(ethylene glycol)]-b-poly(γ-benzyl-l-glutamate) polymeric micelles. Mater. Sci. Eng. C 98, 910–917 (2019).
Ahmed, M. Peptides, polypeptides and peptide–polymer hybrids as nucleic acid carriers. Biomater. Sci. 5, 2188–2211 (2017).
Byrne, M. et al. Molecular weight and architectural dependence of well-defined star-shaped poly (lysine) as a gene delivery vector. Biomater. Sci. 1, 1223–1234 (2013).
Lam, S. J. et al. Peptide-based star polymers as potential siRNA carriers. Aust. J. Chem. 67, 592–597 (2013).
Walsh, D. P. et al. Bioinspired star-shaped poly(l-lysine) polypeptides: efficient polymeric nanocarriers for the delivery of DNA to mesenchymal stem cells. Mol. Pharmaceutics 15, 1878–1891 (2018).
Skoulas, D. et al. Amphiphilic star polypept(o)ides as nanomeric vectors in mucosal drug delivery. Biomacromolecules 21, 2455–2462 (2020).
Yu, Z. et al. Guanidine-rich helical polypeptides bearing hydrophobic amino acid pendants for efficient gene delivery. Biomater. Sci. 9, 2670–2678 (2021).
Paunovska, K., Loughrey, D. & Dahlman, J. E. Drug delivery systems for RNA therapeutics. Nat. Rev. Genet. 23, 265–280 (2022).
Terzopoulou, Z. et al. Biocompatible synthetic polymers for tissue engineering purposes. Biomacromolecules 23, 1841–1863 (2022).
Zhou, X. & Li, Z. Advances and biomedical applications of polypeptide hydrogels derived from α-amino acid N-carboxyanhydride (NCA) polymerizations. Adv. Healthc. Mater. 7, 1800020 (2018).
Zhou, J. et al. A Simple and versatile synthetic strategy to functional polypeptides via vinyl sulfone-substituted l-cysteine N-carboxyanhydride. Macromolecules 46, 6723–6730 (2013).
Gao, Q. et al. Methacrylate-ended polypeptides and polypeptoids for antimicrobial and antifouling coatings. Polym. Chem. 8, 6386–6397 (2017).
Kricheldorf, H. R. & Tönnes, K.-U. Copolymerization of sarcosine-NCA and l-leucine-NCA or l-phenylalanine-NCA. Makromol. Symp. 42-43, 313–328 (1991).
Birke, A., Ling, J. & Barz, M. Polysarcosine-containing copolymers: synthesis, characterization, self-assembly, and applications. Prog. Polym. Sci. 81, 163–208 (2018).
Gao, Q. et al. Fabrication of mixed-charge polypeptide coating for enhanced hemocompatibility and anti-infective effect. ACS Appl. Mater. Interf. 12, 2999–3010 (2020).
Gao, Q. et al. Antibacterial and hydroxyapatite-forming coating for biomedical implants based on polypeptide-functionalized titania nanospikes. Biomater. Sci. 8, 278–289 (2020).
Farrenkopf, P. M. The cost of ignoring vaccines. Yale J. Biol. Med. 95, 265–269 (2022).
Teijaro, J. R. & Farber, D. L. COVID-19 vaccines: modes of immune activation and future challenges. Nat. Rev. Immunol. 21, 195–197 (2021).
O’Brien-Simpson, N. M., Ede, N. J., Brown, L. E., Swan, J. & Jackson, D. C. Polymerisation of unprotected synthetic peptides: a view toward synthetic peptide vaccines. J. Am. Chem. Soc. 119, 1183–1188 (1997).
Liarou, E. et al. Smart polymersomes and hydrogels from polypeptide-based polymer systems through α-amino acid N-carboxyanhydride ring-opening polymerization. From chemistry to biomedical applications. Prog. Polym. Sci. 83, 28–78 (2018).
Geeraedts, F. et al. Superior immunogenicity of inactivated whole virus H5N1 influenza vaccine is primarily controlled by Toll-like receptor signalling. PLoS Pathog. 4, e1000138 (2008).
Di Pasquale, A., Preiss, S., Tavares Da Silva, F. & Garçon, N. Vaccine adjuvants: from 1920 to 2015 and beyond. Vaccines 3, 320–343 (2015).
Nash, A. A., Dalziel, R. G. & Fitzgerald, J. R. In Mims’ Pathogenesis of Infectious Disease 6th edn (eds Nash, A. A., Dalziel, R. G. & Fitzgerald, J. R.), 291–303 (Academic Press, 2015).
Khong, H. & Overwijk, W. W. Adjuvants for peptide-based cancer vaccines. J. Immunother. Cancer 4, 56 (2016).
Nevagi, R. J., Toth, I. & Skwarczynski, M. In Peptide Applications in Biomedicine, Biotechnology and Bioengineering (ed. Koutsopoulos, S.) 327–358 (Woodhead Publishing, 2018).
Kawai, T. & Akira, S. Toll-like receptors and their crosstalk with other innate receptors in infection and immunity. Immunity 34, 637–650 (2011).
Kanneganti, T. D., Lamkanfi, M. & Núñez, G. Intracellular NOD-like receptors in host defense and disease. Immunity 27, 549–559 (2007).
Geijtenbeek, T. B. & Gringhuis, S. I. Signalling through C-type lectin receptors: shaping immune responses. Nat. Rev. Immunol. 9, 465–479 (2009).
Loo, Y. M. & Gale, M. Jr. Immune signaling by RIG-I-like receptors. Immunity 34, 680–692 (2011).
Lim, J.-W. et al. Cationic poly(amino acid) vaccine adjuvant for promoting both cell-mediated and humoral immunity against influenza virus. Adv. Healthc. Mater. 8, 1800953 (2019).
He, Y. et al. Synthetic charge-invertible polymer for rapid and complete implantation of layer-by-layer microneedle drug films for enhanced transdermal vaccination. ACS Nano 12, 10272–10280 (2018).
Zhang, X. et al. Poly(l-lysine) nanostructured particles for gene delivery and hormone stimulation. Biomaterials 31, 1699–1706 (2010).
Beg, S. et al. Lipid/polymer-based nanocomplexes in nucleic acid delivery as cancer vaccines. Drug Discov. Today 26, 1891–1903 (2021).
Lee, H., Jeong, J. H. & Park, T. G. PEG grafted polylysine with fusogenic peptide for gene delivery: high transfection efficiency with low cytotoxicity. J. Controlled Rel. 79, 283–291 (2002).
Mandal, H. et al. ε-Poly-l-lysine/plasmid DNA nanoplexes for efficient gene delivery in vivo. Int. J. Pharmaceutics 542, 142–152 (2018).
Sun, V. Z. et al. Transfection of mammalian cells using block copolypeptide vesicles. Macromol. Biosci. 13, 539–550 (2013).
Rodriguez, A. R., Choe, U.-J., Kamei, D. T. & Deming, T. J. Fine tuning of vesicle assembly and properties using dual hydrophilic triblock copolypeptides. Macromol. Biosci. 12, 805–811 (2012).
Chremos, A. & Douglas, J. F. Communication: when does a branched polymer become a particle? J. Chem. Phys. 143, 111104 (2015).
Acknowledgements
G.G.Q. and N.M.O’B.-S. acknowledge funding support from The National Health and Medical Research Council (NHMRC) of Australia (APP1142472, APP1158841 and APP1185426) and the Australian Research Council (ARC) (DP210102781 and DP160101312).
Author information
Authors and Affiliations
Contributions
S.S., S.H., W.L., Z.S. and D.P. researched data for the article, made a substantial contribution to discussion of content and wrote the article. M.B.C.-P., G.G.Q. and N.M.O’B.-S. made a substantial contribution to discussion of content, wrote and reviewed/edited the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Bioengineering thanks Aaron Lau, who co-reviewed with Ruchika Goyal; Annelise Barron, who co-reviewed with Josefine Eilsø Nielsen; and the other, anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shabani, S., Hadjigol, S., Li, W. et al. Synthetic peptide branched polymers for antibacterial and biomedical applications. Nat Rev Bioeng 2, 343–361 (2024). https://doi.org/10.1038/s44222-023-00143-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s44222-023-00143-4
- Springer Nature Limited