Abstract
Platelets have been shown to be associated with pathophysiological processes beyond thrombosis, demonstrating critical additional roles in homeostatic processes, such as immune regulation and vascular remodeling. Platelets themselves can have multiple functional states and can communicate with and regulate other cells, including immune cells and vascular smooth muscle cells, to serve such diverse functions. Although traditional platelet functional assays are informative and reliable, they are limited in their ability to unravel platelet phenotypic heterogeneity and interactions. Developments in methods such as electron microscopy, flow cytometry, mass spectrometry and ‘omics’ studies have led to new insights. In this Review, we focus on advances in platelet biology and function, with an emphasis on current and promising methodologies. We also discuss technical and biological challenges in platelet investigations. Using coronavirus disease 2019 (COVID-19) as an example, we further describe the translational relevance of these approaches and the possible ‘bench-to-bedside’ utility in patient diagnosis and care.
Similar content being viewed by others
Main
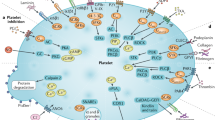
Many studies in the past decade have elaborated the emerging functions of platelets beyond hemostasis and thrombosis. Molecular and functional studies have shown diverse roles of platelets in vascular integrity and remodeling, immunoregulation and tissue regeneration (Fig. 1). Although the function of platelets in the development and progression of cardiovascular diseases is well established, platelets were also demonstrated to be critical players in the pathophysiology of cancer, inflammatory diseases and infections such as that with severe acute respiratory syndrome coronavirus (SARS-CoV)-2, giving rise to COVID-19. Investigations of platelets and their heterogeneous states, platelet releasates and other target cells in various diseases have provided important mechanistic insights for diagnosis, prognosis and therapeutics. In this Review, we discuss the pathophysiological role of platelets and the latest developments in basic and clinical platelet methodologies. We also provide new insights into the development of guidelines for platelet investigation in which knowledge of underlying mechanisms might be important for diagnosis.
Although classically considered as mediators of hemostasis and thrombosis, platelets have been demonstrated to play a wide range of other physiological and pathological roles. Their physiological functions include the maintenance of vascular integrity through interaction with the vascular wall and contributions to wound healing and angiogenesis14,166. Platelets also maintain plasma content homeostasis by storing and releasing many essential plasma factors such as growth factors (VEGF, TGF-β, etc.) and vascular tone regulators such as serotonin166. Platelets are intricately linked to physiological inflammatory responses through cytokine storage and release and by interacting with diverse circulating immune cells167. Many of these homeostatic functions are mediated through transfer to other cells (for example, mRNA or miRNA) or acceptance from other cells. Platelets are well studied for their pathological roles in thrombotic disorders168, such as enhanced vascular thrombosis in diabetes mellitus and many other disease processes14. Platelets have also been linked to the pathogenesis of disease states such as infections (sepsis and viral infections including COVID-19 (refs. 19,20,21)) and malignancies56 in addition to inflammatory disorders, autoimmune disorders, liver disease and kidney disease57,58. This functional diversity in both physiological and pathological contexts is unusual for a small, short-lived cell with no nucleus. Complementing basic investigations with clinical methodology can provide critical mechanistic insights into these diverse functions. DIC, differential interference contrast.
Platelets are heterogeneous first responders to vascular stress
Platelets are anucleate, small (2–4 µm), short-lived (7–10 d) circulating cells in the blood, shed from megakaryocytes1. Approximately, 1011 platelets are produced (and consumed) daily, maintaining a platelet count of between 150 and 450 × 109 platelets per l in the bloodstream of a healthy individual. Platelets have an essential function in thrombosis. Platelet releasates can initiate and propagate thrombosis following mechanical stress or injury, endothelial damage and collagen exposure. Circulating von Willebrand factor (VWF) binding to platelet glycoprotein (GP)Ib (GPIb–IX–V receptor complex) and platelet GPVI–collagen interactions contribute to initial thrombus formation at the damaged site. After initial adhesion to the damaged vessel wall and platelet activation, platelet-derived thromboxane A2 (TxA2) and release of ADP from δ-granules mediates recruitment of additional platelets. Platelet fibrinogen receptor αIIbβ3 primarily contributes to the formation and stabilization of the thrombus. The activation of coagulation factors and other cells, including monocytes, vascular smooth muscle cells (VSMCs), macrophages and the inflamed endothelium further facilitates thrombus development.
In addition to secreted thrombosis mediators including ADP, fibrinogen and VWF, platelets also contain other factors such as cytokines and chemokines (interleukin (IL)-1β, RANTES, MCP-1 and others), which allows them to respond to changes in the local environment and influence other cells (for example, leukocytes). The ability of platelets to influence other cells arises not only from granules (α-granules, dense granules and lysosomes) but also possibly via microvesicles, exosomes, apoptotic bodies and uptake of whole platelets. The procoagulant microvesicles (phosphatidylserine+CD41+CD42b+) released by activated platelets in circulation bearing receptors for collagen and VWF contribute to hemostasis and thrombosis. Platelet-released microvesicles have also been proposed to promote thrombosis in clinical settings such as heparin-induced thrombocytopenia.
Platelets contain a heterogeneous content of coding and noncoding RNA species, machinery for processing RNA transcripts and the capacity to synthesize new proteins in response to stress stimuli. Indeed, the gradual loss of RNA content in ageing platelets might be a determinant of platelet lifespan2. MicroRNA (miRNA) species have been detected in human platelets, and platelet reactivity has been associated with the abundance of selective miRNA species, such as miR-223 and miR-126 (refs. 3,4). Activated platelets can also release miRNA species and probably contribute to the increased levels of miRNA species in plasma3,4,5,6. Several platelet-derived miRNA species predominantly present in extracellular vesicles have been linked to cardiovascular diseases including coronary artery disease (CAD) and thrombotic events. Furthermore, select miRNA species have been shown to have considerable clinical utility in predicting risk and mortality and have been proposed as biomarkers for the assessment of the efficacy of an antiplatelet regimen7,8,9.
Of note, it is also becoming apparent that circulating platelets can exist in different states, and their heterogeneity in size, age and responsiveness might be reflected at rest, upon activation or stimulation by agonists and as observed within the hemostatic plug. Newly synthesized, younger platelets are larger in size, with a higher mean platelet volume, also reflecting their greater prothrombotic potential. Elevated platelet size is associated with increased platelet aggregation and higher risk of cardiovascular and peripheral arterial disease10, and mean platelet volume might be an important prognostic marker10,11. Immature platelets might be more resistant to inhibition with aspirin and clopidogrel, a P2Y12 receptor antagonist12. In addition to differences in function based on age, platelets can be hyperactive13, procoagulant14 or apoptotic13,15, all potentiating a thrombotic tendency. Our studies have shown that diabetes-induced oxidative stress (hyperglycemia, inflammation, lipids) leads to platelet dysfunction and apoptosis13. Paradoxically, platelet apoptosis can induce thrombosis and can result in a higher proportion of younger, more reactive platelets. Apoptotic bodies shed by dying platelets can promote hyperactivity of the remaining platelets16 and lead to increased thrombosis risk, such as that seen in heparin-induced thrombocytopenia17. These and other such studies have highlighted the need to understand and identify the different states of platelets and their contributions to disease processes.
Intercellular interactions of platelets
Platelets execute many of their emerging pathophysiological functions (Fig. 1) by interacting with other cells directly or through platelet releasates (for example, growth factors, hemostatic factors, miRNA), reflecting further functional diversity. With endothelial damage, for example, VSMCs are exposed to circulating platelets. Platelets not only release growth factors such as platelet-derived growth factor (PDGF) that stimulate VSMC dedifferentiation to promote wound healing, but platelets themselves can also be taken up by VSMCs2,7. Platelet inhibition reduces endothelial dysfunction and limits the progression of atherosclerosis and atherothrombotic events18. Major antiplatelet therapies including aspirin and clopidogrel can improve endothelial function in patients with CAD and patients undergoing percutaneous coronary intervention. Platelet–endothelial interactions have also been observed in COVID-19 (refs. 19,20,21). Activated platelets can also interact with monocytes to form platelet–monocyte aggregates, which participate in a broad spectrum of cardiovascular and other inflammatory diseases including atherosclerosis, venous thromboembolism and myocardial infarction22,23,24. The levels of circulating platelet–leukocyte aggregates are elevated in CAD and peripheral artery disease, and studies have shown evidence for platelet–neutrophil interactions at the sites of atherosclerotic plaque rupture and atherothrombosis25. Table 1 summarizes the interactions of platelets with other cells including endothelial cells, VSMCs, red blood cells (RBCs), pathogens and tumor cells that can occur during normal physiology as well as in disease pathology and the major techniques (to be described) that have been successfully used to study such interactions.
Over the past decade, advances in platelet studies have demonstrated platelet heterogeneity in functional states, content and interaction with other cells. Detecting these different states, contents and interactions will lead to recognition of important platelet roles in disease pathogenesis. However, studying platelets can be difficult and requires meticulous approaches (Box 1). Research laboratory techniques might prove clinically useful when complemented with clinical investigative methodology. We will outline some of these promising laboratory research approaches followed by currently used clinical approaches to provide a broad spectrum of what is currently available to investigate the latest developments in platelet heterogeneity and platelet–cellular interactions.
Current platelet techniques in the laboratory
Major progress has been made in the development of methods, both conventional and emerging, to study platelets in both the laboratory research setting and the clinic. Although classical platelet tests such as light transmission aggregometry (LTA) are still used globally to understand platelet responses, the emerging technologies reviewed below provide an expanded set of tools for a more in-depth evaluation of platelet biology and function. Although advantages and limitations are associated with each method (Table 2), the range of available techniques allows the researcher to choose the assay method best suited for the specific research goal. Owing to increasing evidence showing the critical roles of platelets in the prognosis of not only cardiovascular diseases but also in diseases with a previously unknown involvement of platelets, these technologies are ushering in an exciting new era for platelet studies and many might ultimately be used for clinical studies.
Aggregometry to study mechanisms
Platelet aggregometry is considered the gold standard test for platelet functional analysis. Aggregometry measurements take only minutes after sample processing and can be performed on whole blood (impedance-based), platelet-rich plasma (PRP) or washed platelet samples (LTA) with the use of specialized equipment (platelet aggregometer, reviewed by Hvas and Favaloro)26. Multiple-electrode aggregometry uses multiple electrodes and is high throughput. Although multiple-electrode aggregometry can be performed in whole blood, even in samples with low platelet counts, it remains less sensitive than traditional LTA27. In basic research, platelet aggregometry is commonly conducted to assess the effects of deleted or overexpressed genes in animal models or for testing antiplatelet activities of various therapeutic agents. Mouse platelet aggregation is often performed in pooled blood samples because of the scarcity of blood volume that can be drawn from live animals. To allow for individual mouse measurements, our group has optimized whole-blood impedance aggregometry, eliminating the need for euthanasia or sample pooling28.
Flow cytometry
Flow cytometry is a diverse and reliable assay for studying multiple aspects of platelet function in both humans and mice. Flow cytometry allows for quantitation and detection of interactions between activated platelets and other blood cells including leukocytes. Interestingly, platelet aggregation can also be studied with the use of a flow cytometry assay. Flow cytometry assays offer the advantage of being highly sensitive and can be performed with platelet counts as low as 40,000 platelets per mm3 or lower. Numerous platelet-specific markers have been incorporated into flow cytometry assays to study a variety of platelet functions. Platelet-activated glycoprotein αIIbβ3 antibody (PAC-1) is a specific monoclonal antibody that recognizes an epitope present on the activated form of human αIIbβ3 and can be used to differentiate resting platelets from activated platelets29. The presence of CD62P (P-selectin) and receptor TREML1 on the platelet surface, usually found in α-granules of platelets, is a marker of activation-coupled degranulation. Measurement of intraplatelet calcium levels with the use of calcium-binding fluorescent dyes can also be used to study platelet-activation kinetics and activation states. Although a Fluo-3-based assay has been shown to measure activation-associated intraplatelet calcium kinetics in whole-blood platelets, Fluo-4 and Fluo-5 have been used to study platelet-activation states and distinguish procoagulant platelets from activated but non-coagulant platelets16. Other ratiometric dyes such as Fura-2 have also been used to quantitate absolute calcium concentration changes during agonist-induced platelet activation30. Platelet mitochondrial depolarization, which is an important parameter for mitochondrial function, apoptosis31 and externalization of phosphatidylserine, another property of an apoptotic, procoagulant platelet, can be also detected by flow cytometry32. Measurement of intraplatelet reactive oxygen species by flow cytometry with the use of specific cell-permeable dyes has been used in the study of platelet aging and oxidative stress28.
Performing flow cytometry with the use of thiazole orange (a fluorescent dye that binds nucleic acids) can be used to distinguish the reticulated platelet subpopulation and is especially useful in the estimation of platelet production and megakaryopoiesis in thrombocytopenic disorders33,34. Automated blood analyzers (Sysmex XE-/XN-series) can also measure the immature platelet fraction in whole blood and are used often in clinical settings34.
As platelet function inherently depends on the dynamic interplay between activating and inhibitory signaling, multiple methods have now been developed to study phosphoprotein patterns by flow cytometry, offering large advantages over more conventional approaches including immunoblotting and mass spectrometry that rely on lysis and extraction35. A smart application of the flow cytometry assay to platelet function analysis has been developed to identify heterogeneity in platelet responsiveness to agonists in healthy donors. Coined as a ‘Phenomic’ study by the authors, Dunster and coworkers used multiple agonists at varying concentrations to generate a platelet-response profile of each individual on the basis of surface marker expression36. Although this technique requires a more careful and elaborate assay design compared with that of conventional flow cytometry, this analysis could be potentially helpful in studying platelet function variability in a cohort with better reproducibility.
Proteomics
Platelets contain a vast pool of proteins (estimated to be more than 3,000) that includes membrane proteins, cytoskeletal proteins, signaling proteins, enzymes, coagulation factors and transcription factors. The discovery of this rich and diverse repertoire of platelet proteins has been largely achieved by platelet proteomic analysis. Platelet two-dimensional electrophoresis–mass spectrometry (2DE–MS) proteomics to study thrombin-induced activation has led to the identification of specific proteins released by platelets37. In addition, platelet proteomic-analysis studies have identified numerous proteins that mediate hypoxia-induced platelet hyper-reactivity38.
Proteomic advances have made it possible to study platelet subproteomes on the basis of post-translational modifications, and phospho-proteome analyses have revealed phosphorylation patterns in resting and activated platelets39,40. With the use of immobilized metal affinity chromatography (IMAC) and titanium dioxide coupled with nano-liquid chromatography (LC)–MS and isobaric tags for relative and absolute quantification (iTRAQ) (modified proteomic technique)41, specific nodes in platelet-activation signaling have been identified. Studies of the glycosylated proteome showed a major role for N-linked glycosylation of adhesive proteins following binding to collagen42. Platelet protein ubiquitination is another important post-translational modification involved in protein degradation, signaling and protein–protein interactions and has been analyzed by studying the platelet ubiquitinome43.
Mass cytometry time of flight (mass CyTOF) is a fairly new methodology that combines features of flow cytometry and mass spectrometry, improving upon the limitations of traditional flow cytometry through the use of antibodies covalently tagged by heavy-metal isotypes rather than fluorophores44. Despite the antibody dependence, the greatest advantage of mass cytometry is the large number of simultaneous parameters that can be analyzed, which allows for comprehensive characterization of cellular proteins and pathways. Mass cytometry has been used to identify previously unappreciated platelet subpopulations45,46 and is emerging as a powerful single-cell proteomic technique.
Lipidomics
Diverse lipids and fatty acids including phospholipids, sphingolipids, steroids, eicosanoids (prostaglandins), phosphatidylinositides, diglycerides, triglycerides and cholesteryl esters are present in varying proportions in platelets and can regulate major aspects of platelet function including aggregation, morphological changes, coagulation and granule release47. Advances in mass spectrometry analysis of lipids, ‘lipidomics’, using high-resolution rapid scanning instruments have provided new insights into understanding the role of the platelet lipidome in the regulation of hemostasis and its contribution to the progression of thrombosis and atherosclerosis. Currently, lipidomics is commonly performed with the use of either ‘shotgun’ lipidomics, a high-throughput approach that detects the most abundant species, or LC–MS, a targeted and highly sensitive approach. A comprehensive analysis of the platelet lipidome has revealed that the platelet lipidome is altered during platelet activation and probably has a role in platelet procoagulant function48,49. Furthermore, studies on the composition of stored platelets50 and extracellular vesicles have confirmed the presence of cholesterol and other lipids in platelets. High cholesterol levels as observed in hypocholesterolemia correlate with the elevated amounts of cholesterol found in platelet membranes and greater thrombosis risk51. Altered lipidomic profiles have been observed in patients with CAD52 and myocardial infarction. Therefore, platelet lipidomic profiles could be potentially used as disease signatures (such as plaque progression in atherosclerosis), for risk stratification of patients and to determine treatment efficacy.
Genomics and transcriptomics
Although platelets lack nuclei, genomic sequencing (exon or whole genome) and/or genome-wide association studies (GWAS) can be informative by studying genetic mutations or single-nucleotide polymorphisms associated with platelet phenotypic traits or bleeding disorders. A whole-genome-sequencing study identified regulatory noncoding genomic loci affecting platelet function53. One single-nucleotide polymorphism in the GNB3 gene associated with enhanced platelet function was identified in African American families with heritable premature CAD54. Platelet transcriptomics with the use of microarray and serial analysis of gene expression (SAGE) strategies have revealed platelets to represent around 15–25% of the human genes present in gene libraries37,55. All these genomic analyses are especially important for heritable platelet disorders and in cases of family history of cardiovascular risk. RNA sequencing and platelet RNA profiling of tumor-educated circulating platelets from over 220 patients with various types of cancers have revealed a unique platelet transcriptome profile that can accurately identify primary tumor sites56. Unique platelet transcriptomes that do not necessarily correlate with platelet hyperactivation signatures have also been characterized in myocardial infarction, lupus, kidney disease, obesity and sepsis57,58. Transcriptome analyses have highlighted the diverse roles of platelets beyond thrombotic activation and provide an important strategy for future investigations.
Despite advances over the past few years in high-throughput and high-sensitivity techniques, a challenge in platelet ‘omics’ studies is to attain a high level of purity of platelet samples. The possible presence of small numbers of leukocytes or RBCs in the isolated platelet population, even after centrifugation-based washing steps, makes it mandatory to estimate the purity of an isolated platelet population. An average of one to five leukocytes per 10 million platelets (<0.00005% contamination) has been suggested as the acceptable limit. Flow cytometry assays make it possible to gate platelet populations with the use of CD41- or CD42b-specific antibodies. PCR or western blotting are other reliable methods, using a CD45-targeted primer or antibody, respectively, to detect leukocyte contamination. The low yields of platelet RNA and the potential for leukocyte contamination makes RNA analysis challenging. However, this challenge can be overcome by adding RNA-amplification steps into the standard protocols. Leukocyte depletion with magnetic bead-based antibodies is preferred for platelet RNA studies, although carefully conducted centrifugation steps along with PCR-based purity checks might bypass the need for leukocyte depletion. Platelet RNA analysis can also be performed by PCR-based arrays with the use of standard primers for the candidate genes or using commercially available high-throughput arrays28.
Platelet imaging
Many imaging tools using fluorescence or phase contrast have advanced our understanding of platelet biology. A range of specialized microscopy techniques (Table 2) has allowed the visualization of platelet activation and receptor profiles, the formation of platelets from megakaryocytes and even the cytoskeletal rearrangements for the formation of thrombi59. Microscopy methods include wide-field microscopy, encompassing phase-contrast, total internal reflection fluorescence, reflectance interference-contrast and differential interference-contrast microscopy59,60,61. A platelet-adhesion assay tests the ability of platelets to adhere to and spread on different assay surfaces and can be visualized in static mode or under flow with the use of perfusion chambers and immunofluorescence microscopy62. Similarly, time-lapse phase-contrast microscopy with high magnification has been used to visualize and quantitate platelet migration after activation in the context of innate immune responses to bacterial challenges63. The development of in vivo imaging techniques such as intravital microscopy allows for dynamic analysis of interactions between platelets and vascular cells in real time64.
In the last decade, the emergence of super-resolution microscopy and other nanoscopy techniques such as structured illumination microscopy and single-molecular-localization microscopy have allowed visualization at the nanometer resolution while overcoming the limitations of light micrsocopy65. Given that biological function is preserved during sample preparation for these techniques, these approaches have provided useful insights into cytoskeletal rearrangement, actin and tubulin fibers in platelets and receptor-colocalization studies66,67,68. However, these methods have been largely limited to in vitro and ex vivo studies because they are invasive and technically challenging to perform.
The photolithography technique coupled with immunofluorescence microscopy has been applied to quantitate the mechanical traction force generated by the platelet cytoskeleton during adhesion and spreading69. Customized atomic force microscopy has also been used to study the mechanics of platelet contraction at the single-cell level during clotting, which examines single platelets spread between two fibrinogen-coated surfaces70. Although less physiologic than in vivo imaging, microfluidic device imaging represents another approach to study platelet interactions with vascular cells and leukocytes in real time71. In conjunction with other technologies (for example, flow cytometry), these assays can provide useful information on the role of platelets in inflammation and immunity72.
The development of high-throughput imaging and deep learning has taken platelet imaging to the next level. Intelligent image-activated cell sorting (iLACS) technology, which was initially developed for nucleated cells73,74, has been applied successfully to platelets in the form of ‘intelligent platelet morphometry’. This technique combines optofluidic time-stretch microscopy with a convolutional neural network in a deep learning algorithm to image and quantify morphological features of platelet aggregates, activated by different types of agonists in a high-throughput (>1,000 cells per second) approach75. This technology might be useful for monitoring antiplatelet therapy or identifying patients with increased thrombotic risk, such as those with cardiovascular disease or diabetes or patients hospitalized with COVID-19. Other single-cell microfluidic assays using real-time deformability flow cytometry and light-emitting diodes for mechanical phenotyping might offer additional insights into platelet physiology; however, they have yet to be validated for studying platelets76.
Electron microscopy
Electron microscopy has been traditionally used for the study of platelet morphology and ultrastructure, both as a basic research method and a diagnostic tool. Transmission electron microscopy (TEM) studies have provided detailed insights into the molecular mechanisms of platelet granule release, platelet microparticle formation and release and platelet mitochondrial dysfunction77,78,79,80,81 (Table 2). Moreover, TEM has also been a critical tool in discerning the differences in overall platelet morphology and cytoskeletal organization in platelet function defects82,83. Immunogold labeling, with the use of antibodies loaded with nanometer-size colloidal gold particles, allows the study of localized proteins and macromolecules within platelets. In combination with TEM, immunogold labeling has been used to determine the subcellular localization of many platelet proteins in α-granules, the open canalicular system and mitochondria as well as in extracellular vesicles84,85,86.
In the clinical setting, TEM is commonly recommended as part of the diagnostic work-up for storage pool disorders and other structural abnormalities87, including genetic disorders such as Hermansky–Pudlak syndrome and Chediak–Higashi syndrome82. In particular, whole-mount TEM has been shown to have clinical utility in evaluation of dense granule deficiency in patients with suspected platelet function disorders88. Given that whole-mount TEM has a superior sensitivity for dense granule deficiency than for platelet aggregometry and ADP-release assays, this technique is considered the gold standard for diagnosis of storage pool disorders. However, this method is fairly expensive, requires specialized equipment and is therefore only available in specialized centers.
In contrast to TEM, which is two-dimensional, conventional scanning electron microscopy (SEM) provides high-resolution imaging of three-dimensional (3D) surface topology. SEM is particularly useful in characterizing the composition and structure of thrombi and morphologic changes in platelets following activation81. Although SEM provides valuable information about clot and fibrin networks89,90, typical analysis is manual and tedious. Newer automated platforms are now being developed to increase accuracy and reproducibility in imaging clots91. Several studies have used SEM to facilitate understanding of thrombus formation, platelet adhesion and platelet properties under flow and mechanical stress81,92,93,94. SEM has also been used to analyze the structure and composition of thrombi in myocardial infarction and ischemic stroke and to characterize the distinct heterogeneity of arterial and venous thrombi77,95.
Cryogenic electron microscopy (cryo-EM) allows TEM of hydrated biological samples, preserving the native biological organization of cellular organelles and macromolecules81,96. Cryo-EM has been used to characterize the platelet open canalicular system and its interaction with the dense tubular system and α-granules along with platelet spreading and cytoskeletal changes at nanometer to subnanometer resolution. Serial block face SEM and focused ion beam SEM incorporate serial sectioning with SEM, allowing the acquisition of serial images with nanometer-size z-plane spacing (~20–30 nm for serial block face SEM and ~5 nm for focused ion beam SEM)81,96. These techniques have been used to obtain 3D structures of whole platelets with detailed characterization of platelet size, volume, activation events and organelles including mitochondria, Golgi and granule secretion events. Correlative light and electron microscopy (CLEM) is a new technique that combines the advantages of both fluorescence imaging and electron microscopy, with fewer limitations than conventional electron microscopy (Table 2). CLEM has been used to characterize in detail the spatial regulation of platelet activation during hemostasis94.
Although electron microscopy imaging has its challenges owing to the technical expertise and expensive instrumentation required, it remains the gold standard for studying platelet morphology and ultrastructure, both for understanding platelet biology and identifying various platelet abnormalities and storage pool deficiencies.
Non-imaging immunoassays
Although high-throughput and increasingly sensitive proteomic techniques have their unique advantages, their use is limited for routine analysis of platelet biology (Table 2). Immunodetection methods use specific antibodies and allow easy validation and a relatively inexpensive platform to identify proteins and detect quantitative differences. Enzyme-linked immunosorbent assays (ELISA) have been regularly used for the measurement of soluble platelet-activation markers including soluble P-selectin and platelet factor 4 as well as the platelet secretome (from α-granules and dense granules) and the platelet sheddome (GPIbα, GPVI and other membrane proteins)97,98,99. Changes in the levels of plasma platelet-activation markers have been associated with thromboembolic events and form a valuable tool for acute event prognosis and timely individualized adjustment of the treatment regimen to prevent thrombosis. However, despite quantitative results, ELISA is limited to analyzing the plasma compartment and does not provide isoform-specific information. By contrast, western immunoblotting has long been used as a classical tool to distinguish specific protein expression in platelets and for the investigation of both intracellular signaling pathways and surface markers. Although semi-quantitative, western immunoblotting is considered one of the most common and reliable techniques to also validate results from more extensive proteomic studies. The use of fluorescently labeled secondary antibodies has substantially improved the quantitative accuracy of platelet proteomes, thereby forming a rapid and reproducible routine platform for quantitative proteomic studies of platelets.
Several of the above-described techniques have been used to study interactions of platelets with other cells including endothelial cells, VSMCs, RBCs, pathogens and tumor cells that occur both in normal and disease states (Table 1). Therefore, basic research techniques might provide important insights with clinical relevance. We will next discuss the development and current status of the major clinical assays used for platelet function tests.
Current methodology for clinical evaluation
Traditional platelet testing and platelet aggregometry in clinical medicine
Traditional clinical laboratory testing for platelet disorders generally relies on platelet counts and platelet function testing. The bleeding time test, introduced in the early 20th century, was the first functional test to evaluate primary hemostasis and platelet disorders but is no longer used in clinical practice owing to its low sensitivity, poor reproducibility, scarring potential and absence of correlation with clinical outcome. The ability to test platelet function in the clinical laboratory has substantially improved with the introduction of platelet LTA, which rapidly became a widely used and informative methodology for diagnosing and managing platelet disorders26. Subsequent variations of aggregation-based platelet function tests have been developed and include whole-blood impedance platelet aggregometry (multiple-electrode aggregometry with the use of a Multiplate Analyzer)100,101 and VerifyNow102,103 (Table 3).
The development of multiple-electrode aggregometry that uses electrical impedance offers platelet function analysis in whole blood. Multiple-electrode aggregometry has been demonstrated to be useful for the diagnosis of heparin-induced thrombocytopenia104,105 and as a point-of-care test for pediatric patient samples106 in which sample volumes are very low. Other studies, however, found multiple-electrode aggregometry to be less sensitive for detection of abnormal platelet function and to have lower prognostic value than LTA in patients27,107. Nevertheless, multiple-electrode aggregometry offers several advantages over LTA, such as a lower sample volume requirement, and it is faster to perform on a per-sample basis.
Although platelet aggregation responses have historically been one of the most common ways of evaluating platelet disorders, these methods are limited by their sensitivity, labor intensiveness and number of parameters evaluated. However, certain factors must be considered while interpreting the aggregation assay data in which platelet function is biologically distinct such as in the case of neonates. Studies have shown that neonatal platelets are hypofunctional with agonist stimulation by aggregation-based methods. However, full-term neonates typically maintain normal physiologic hemostasis108 despite elevated hematocrit and increased VWF levels, which likely counterbalance intrinsic platelet hyporeactivity. A small but considerable proportion of patients with suspected platelet or other bleeding disorders remain without a diagnosis even after comprehensive clinical laboratory testing.
Thromboelastography
In vitro assays that simulate physiologic whole-blood hemostasis can evaluate relative contributions of platelets, fibrinogen and thrombin generation. The critical feature of such methods for platelets is that their function is assessed under more physiological conditions, with contributions from the other blood and plasma components that drive hemostasis. Thromboelastography (TEG) and rotational thromboelastometry are similar viscoelastic technologies that use whole blood to analyze dynamic changes over time, including clot formation, strength and lysis109. These methods are primarily used for the assessment of global hemostasis (that is, coagulopathy versus hemorrhage) and to support targeted transfusion therapy in patients during trauma or major surgery (for example, cardiopulmonary bypass and liver transplantation) (Table 3). Together, modifications to the standard TEG system and the addition of platelet agonists (for example, ADP, arachidonic acid and thrombin analogs) provide a new assay, known as TEG platelet mapping, that allows for a more specific assessment of platelet function110. This technique has been shown to have utility in monitoring antiplatelet therapies111. However, this approach has a fairly low sensitivity for platelet function and is therefore not recommended for diagnosing platelet functional disorders. The TEG6s Platelet Mapping assay, which uses disposable multichannel microfluidic cartridges, has been shown to have clinical utility in evaluating platelet function112,113,114. This method was shown to have less interindividual variability and greater ease of use than other viscoelastic assays. Evaluating hemostasis in patients with COVID-19 demonstrated the usefulness of TEG6s Platelet Mapping in monitoring antiplatelet and anticoagulant therapy115.
Clinical microfluidics and shear-induced platelet responses
A major limitation in current platelet function testing is the difficulty of replicating biorheological conditions in vitro. Vascular thrombus formation is a dynamic process, dependent on shear forces generated from physiologic blood flow and blood viscosity, which drive platelet adhesion, activation and aggregation116. Several platelet function tests (Table 3) have been developed to study shear-induced platelet responses to better simulate the physiologic processes that occur in the vessel. The simplest and most widely used test is the platelet function analyzer (PFA; PFA-100 and PFA-200), which uses citrated whole blood and measures the closure time of a membrane aperture coated with agonists117,118. The PFA assesses platelet adhesion and aggregation under flow and high-shear conditions that simulate primary hemostasis. Although the PFA is moderately sensitive for platelet dysfunction, it has low specificity and is primarily used as a rapid clinical tool for screening for von Willebrand disease and monitoring the efficacy of antiplatelet therapies119. Given that the PFA assay is sensitive to low platelet count and low hematocrit, results need to be adjusted for thrombocytopenia and anemia, and its clinical utility might be limited in these settings.
Although these technologies are useful for studying various aspects of thrombus formation and platelet interactions under uniform shear stress, they are unable to sufficiently simulate the dynamic flow in vessels in which bifurcation or stenosis is common. The development of microfluidic devices has allowed for more complex designs of networks that better model the vasculature120,121. The Total Thrombus-Formation Analysis System uses whole blood under physiological shear conditions with disposable microchips coated with activators such as collagen and tissue factor122 and has demonstrated promise in detecting platelet function and coagulation disorders. The method has also shown potential in monitoring antiplatelet therapies in patients with CAD and predicting periprocedural bleeding in patients undergoing percutaneous coronary intervention123,124. More recently, a microfluidic device has been developed that measures platelet contractile forces under shear gradients125; this technique might be useful for detecting platelet inhibition, guiding antiplatelet therapy and predicting bleeding risk and transfusion needs in patients with trauma. Although these newer technologies might eventually be useful complements to established laboratory evaluation of platelet function, most have not been rigorously tested in a clinical setting; a need exists for standardization and validation before routine clinical use126.
The use of rigorously optimized methods for platelet preparation and isolation and platelet functional testing is of primary importance for achieving reproducible and clinically relevant results in all platelet-investigative studies. Integrating the basic research methodologies with clinical utility assays allows both platelet biologists and clinicians to comprehensively advance the understanding of the pathophysiological role of platelets in cardiovascular and other diseases. Moreover, the combined use of these various approaches also provides a means to study the effects of antiplatelet agents and their mechanisms of action.
Investigating platelets in COVID-19
COVID-19 is a prime example in which basic investigations, beyond standard platelet counts and aggregation assays, have complemented clinical investigations to improve diagnostic and therapeutic strategies. Platelets can contribute to the overall procoagulant and inflammatory states during viral infection, potentially leading to thrombosis, vascular complications and high mortality127. COVID-19 is associated with thrombocytopenia, thrombosis and hemorrhage in the setting of underlying endotheliopathy, coagulopathy and inflammation, with each serving as biomarkers for disease severity and poor prognosis19,128. Several mechanisms have been investigated and proposed for the development of thrombocytopenia, including platelet activation and/or consumption and subsequent clearance by the reticuloendothelial system, including platelet consumption in response to increased endothelial damage, platelet auto-antibody formation and subsequent platelet clearance, bone marrow or megakaryocyte suppression owing to inflammation or direct viral infection19,129,130. Studies have used the above-described techniques to elucidate possible functions for platelets in the thrombo-inflammatory activation responses of COVID-19. A combination of flow cytometry, confocal microscopy, TEM and RNA sequencing demonstrated increased formation of platelet–neutrophil and platelet–monocyte aggregates, increased platelet activation and altered expression of over 3,000 genes in patients hospitalized with COVID-19 (ref. 131). Platelet–monocyte aggregates were also identified in another study of patients critically ill with COVID-19 with the use of flow cytometry assays132. In a separate study, Althaus and colleagues used flow cytometry assays and western blotting to investigate COVID-19-associated platelet apoptosis mediated by circulating immune complexes in patient sera133. Mass cytometry was also used to characterize a unique platelet hyperactivation phenotype of platelet surface proteins in COVID-19 (ref. 134). A remarkable application of the latest imaging technology for platelets in COVID-19 used large-scale single-cell platelet imaging by intelligent platelet morphometry135; the investigators analyzed whole-blood samples to identify platelet aggregates in nearly 90% of all patients with COVID-19. TEM demonstrated uptake of SARS-CoV-2 through attachment to microparticles, independent of angiotensin-converting enzyme 2 (ACE2) (ref. 136). Moreover, transcriptomic profiling revealed the presence of both platelet apoptosis and necroptosis136. The application of these diverse basic sciences’ platelet methodologies highlights the potential utility of these emerging assays for clinical use. Increasing evidence of COVID-19 thrombocytopathy associated with platelet–leukocyte interactions, various states of activation or hyperactivation, apoptosis and necroptosis has facilitated and encouraged the use of antiplatelet agents (for example, low-dose aspirin in addition to anticoagulation) to combat thrombosis in COVID-19 (refs. 137,138). The results of these studies have been promising with improved morbidity and mortality. Many other drugs targeting thrombosis are also being tested19. Techniques such as TEG, especially TEG6s, have proven useful in investigating the levels of thrombogenicity markers139 and also evaluating the pharmacodynamic response to anticoagulants and aspirin in patients with COVID-19 (ref. 115). Also related to patients with COVID-19, platelet function tests have proven useful in cases of vaccine-induced thrombotic thrombocytopenia, a rare but life-threatening condition, in vaccinated individuals. The use of both conventional and emerging assays in patients with vaccine-induced thrombotic thrombocytopenia is another example of the importance of these assays in the clinic140,141. Thus, using both conventional assays and cutting-edge techniques to study platelets in normal physiology and in disease and the incorporation of these assays into clinical decision making has proven to be beneficial and warranted.
Conclusion
On the basis of major advances in basic science investigations, platelets can no longer be thought of as a homogeneous population but rather as a heterogeneous interactive population with distinct subgroups that can protect against or contribute to disease processes. The classical and advanced methods and studies described in this review have enhanced our understanding of the heterogeneity of platelets, the functions of distinct platelet subsets and the mechanistic complexities of the roles of platelets in diseases (Table 4). The frontier will be to continue to characterize platelets at a single-cell, single-disease and single-person level. The study of individual-level variabilities in platelet function and response efficiently and accurately by emerging assays can be the key step towards enhanced clinical safety of antiplatelet drugs. Some of these developing techniques might also be adapted for high-throughput screening in the clinic and especially for patients with trauma for quick decision making as well as to identify new therapeutics targeting unique populations of platelets. These techniques can also potentially be used to monitor therapeutic efficacy. These technologies and discoveries might ideally lead towards the development of more precise and personalized diagnostics and point-of-care devices that will increase our ability to understand disease etiology and bleeding risk, integrating information on each patient’s history, presentation and risk factors. This knowledge will allow for customized, targeted therapies for treatment and prevention of cardiovascular diseases and other diseases.
References
van der Meijden, P. E. J. & Heemskerk, J. W. M. Platelet biology and functions: new concepts and clinical perspectives. Nat. Rev. Cardiol. 16, 166–179 (2019).
Zeng, Z. et al. Platelet-derived miR-223 promotes a phenotypic switch in arterial injury repair. J. Clin. Invest. 129, 1372–1386 (2019).
Kaudewitz, D. et al. Association of microRNAs and YRNAs with platelet function. Circ. Res. 118, 420–432 (2016).
Nagalla, S. et al. Platelet microRNA–mRNA coexpression profiles correlate with platelet reactivity. Blood 117, 5189–5197 (2011).
Zampetaki, A. et al. Prospective study on circulating microRNAs and risk of myocardial infarction. J. Am. Coll. Cardiol. 60, 290–299 (2012).
Gao, J., Ma, X., Zhang, Y., Guo, M. & Shi, D. The role of microRNAs in prethrombotic status associated with coronary artery disease. Thromb. Haemost. 117, 429–436 (2017).
Willeit, P. et al. Circulating microRNAs as novel biomarkers for platelet activation. Circ. Res. 112, 595–600 (2013).
Liu, J. et al. Platelet-derived miRNAs as determinants of the antiplatelet response in clopidogrel-treated patients with ACS. Thromb. Res. 186, 71–74 (2020).
Paseban, M. et al. Modulation of microRNAs by aspirin in cardiovascular disease. Trends Cardiovasc. Med. 30, 249–254 (2020).
Slavka, G. et al. Mean platelet volume may represent a predictive parameter for overall vascular mortality and ischemic heart disease. Arterioscler. Thromb. Vasc. Biol. 31, 1215–1218 (2011).
Pafili, K., Penlioglou, T., Mikhailidis, D. P. & Papanas, N. Mean platelet volume and coronary artery disease. Curr. Opin. Cardiol. 34, 390–398 (2019).
Armstrong, P. C. et al. Newly formed reticulated platelets undermine pharmacokinetically short-lived antiplatelet therapies. Arterioscler. Thromb. Vasc. Biol. 37, 949–956 (2017).
Tang, W. H. et al. Glucose and collagen regulate human platelet activity through aldose reductase induction of thromboxane. J. Clin. Invest. 121, 4462–4476 (2011).
Agbani, E. O. & Poole, A. W. Procoagulant platelets: generation, function, and therapeutic targeting in thrombosis. Blood 130, 2171–2179 (2017).
Leytin, V. Apoptosis in the anucleate platelet. Blood Rev. 26, 51–63 (2012).
Abbasian, N., Millington-Burgess, S. L., Chabra, S., Malcor, J. D. & Harper, M. T. Supramaximal calcium signaling triggers procoagulant platelet formation. Blood Adv. 4, 154–164 (2020).
Warkentin, T. E. et al. Sera from patients with heparin-induced thrombocytopenia generate platelet-derived microparticles with procoagulant activity: an explanation for the thrombotic complications of heparin-induced thrombocytopenia. Blood 84, 3691–3699 (1994).
van Gils, J. M., Zwaginga, J. J. & Hordijk, P. L. Molecular and functional interactions among monocytes, platelets, and endothelial cells and their relevance for cardiovascular diseases. J. Leukoc. Biol. 85, 195–204 (2009).
Gu, S. X. et al. Thrombocytopathy and endotheliopathy: crucial contributors to COVID-19 thromboinflammation. Nat. Rev. Cardiol. 18, 194–209 (2021).
Parker, W. A. E. & Storey, R. F. Platelets and the endothelium: active participants in severe COVID-19 infection. JACC Basic Transl. Sci. 6, 219–221 (2021).
Barrett, T. J. et al. Platelets amplify endotheliopathy in COVID-19. Sci. Adv. 7, eabh2434 (2021).
Allen, N. et al. Circulating monocyte–platelet aggregates are a robust marker of platelet activity in cardiovascular disease. Atherosclerosis 282, 11–18 (2019).
Nhek, S. et al. Activated platelets induce endothelial cell activation via an interleukin-1β pathway in systemic lupus erythematosus. Arterioscler. Thromb. Vasc. Biol. 37, 707–716 (2017).
Rajagopalan, S. et al. Platelet activation increases with the severity of peripheral arterial disease: implications for clinical management. J. Vasc. Surg. 46, 485–490 (2007).
Silvestre-Roig, C., Braster, Q., Ortega-Gomez, A. & Soehnlein, O. Neutrophils as regulators of cardiovascular inflammation. Nat. Rev. Cardiol. 17, 327–340 (2020).
Hvas, A. M. & Favaloro, E. J. Platelet function analyzed by light transmission aggregometry. Methods Mol. Biol. 1646, 321–331 (2017).
Al Ghaithi, R., Drake, S., Watson, S. P., Morgan, N. V. & Harrison, P. Comparison of multiple electrode aggregometry with lumi-aggregometry for the diagnosis of patients with mild bleeding disorders. J. Thromb. Haemost. 15, 2045–2052 (2017).
Jain, K. et al. Age associated non-linear regulation of redox homeostasis in the anucleate platelet: implications for CVD risk patients. EBioMedicine 44, 28–40 (2019).
Huang, J. et al. Platelet integrin αIIbβ3: signal transduction, regulation, and its therapeutic targeting. J. Hematol. Oncol. 12, 26 (2019).
Harper, M. T. & Poole, A. W. Store-operated calcium entry and non-capacitative calcium entry have distinct roles in thrombin-induced calcium signalling in human platelets. Cell Calcium 50, 351–358 (2011).
Bock, F. J. & Tait, S. W. G. Mitochondria as multifaceted regulators of cell death. Nat. Rev. Mol. Cell Biol. 21, 85–100 (2020).
Gyulkhandanyan, A. V., Mutlu, A., Freedman, J. & Leytin, V. Markers of platelet apoptosis: methodology and applications. J. Thromb. Thrombolysis 33, 397–411 (2012).
Buttarello, M., Mezzapelle, G., Freguglia, F. & Plebani, M. Reticulated platelets and immature platelet fraction: clinical applications and method limitations. Int. J. Lab. Hematol. 42, 363–370 (2020).
Hoffmann, J. J. Reticulated platelets: analytical aspects and clinical utility. Clin. Chem. Lab. Med. 52, 1107–1117 (2014).
Spurgeon, B. E. J. & Naseem, K. M. Phosphoflow cytometry and barcoding in blood platelets: technical and analytical considerations. Cytometry B Clin. Cytom. 98, 123–130 (2020).
Dunster, J. L. et al. Multiparameter phenotyping of platelet reactivity for stratification of human cohorts. Blood Adv. 5, 4017–4030 (2021).
Coppinger, J. A. et al. Characterization of the proteins released from activated platelets leads to localization of novel platelet proteins in human atherosclerotic lesions. Blood 103, 2096–2104 (2004).
Tyagi, T. et al. Altered expression of platelet proteins and calpain activity mediate hypoxia-induced prothrombotic phenotype. Blood 123, 1250–1260 (2014).
Babur, O. et al. Phosphoproteomic quantitation and causal analysis reveal pathways in GPVI/ITAM-mediated platelet activation programs. Blood 136, 2346–2358 (2020).
Hogrebe, A. et al. Benchmarking common quantification strategies for large-scale phosphoproteomics. Nat. Commun. 9, 1045 (2018).
Beck, F. et al. Temporal quantitative phosphoproteomics of ADP stimulation reveals novel central nodes in platelet activation and inhibition. Blood 129, e1–e12 (2017).
Toonstra, C., Hu, Y. & Zhang, H. Deciphering the roles of N-glycans on collagen–platelet interactions. J. Proteome Res. 18, 2467–2477 (2019).
Unsworth, A. J. et al. Human platelet protein ubiquitylation and changes following GPVI activation. Thromb. Haemost. 119, 104–116 (2019).
Behbehani, G. K. Applications of mass cytometry in clinical medicine: the promise and perils of clinical CyTOF. Clin. Lab. Med. 37, 945–964 (2017).
Blair, T. A., Michelson, A. D. & Frelinger, A. L. III Mass cytometry reveals distinct platelet subtypes in healthy subjects and novel alterations in surface glycoproteins in Glanzmann thrombasthenia. Sci. Rep. 8, 10300 (2018).
Spurgeon, B. E. J., Michelson, A. D. & Frelinger, A. L. III Platelet immunophenotyping by high-dimensional mass cytometry. Curr. Protoc. 1, e112 (2021).
O’Donnell, V. B., Murphy, R. C. & Watson, S. P. Platelet lipidomics: modern day perspective on lipid discovery and characterization in platelets. Circ. Res. 114, 1185–1203 (2014).
Peng, B. et al. Identification of key lipids critical for platelet activation by comprehensive analysis of the platelet lipidome. Blood 132, e1–e12 (2018).
Slatter, D. A. et al. Mapping the human platelet lipidome reveals cytosolic phospholipase A2 as a regulator of mitochondrial bioenergetics during activation. Cell Metab. 23, 930–944 (2016).
Ruebsaamen, K., Liebisch, G., Boettcher, A. & Schmitz, G. Lipidomic analysis of platelet senescence. Transfusion 50, 1665–1676 (2010).
Wang, N. & Tall, A. R. Cholesterol in platelet biogenesis and activation. Blood 127, 1949–1953 (2016).
Chatterjee, M. et al. Regulation of oxidized platelet lipidome: implications for coronary artery disease. Eur. Heart J. 38, 1993–2005 (2017).
Keramati, A. R. et al. Genome sequencing unveils a regulatory landscape of platelet reactivity. Nat. Commun. 12, 3626 (2021).
Bray, P. F. et al. Heritability of platelet function in families with premature coronary artery disease. J. Thromb. Haemost. 5, 1617–1623 (2007).
Gnatenko, D. V., Dunn, J. J., Schwedes, J. & Bahou, W. F. Transcript profiling of human platelets using microarray and serial analysis of gene expression (SAGE). Methods Mol. Biol. 496, 245–272 (2009).
Best, M. G. et al. RNA-seq of tumor-educated platelets enables blood-based pan-cancer, multiclass, and molecular pathway cancer diagnostics. Cancer Cell 28, 666–676 (2015).
Middleton, E. A. et al. Sepsis alters the transcriptional and translational landscape of human and murine platelets. Blood 134, 911–923 (2019).
Ple, H. et al. Alteration of the platelet transcriptome in chronic kidney disease. Thromb. Haemost. 108, 605–615 (2012).
Montague, S. J., Lim, Y. J., Lee, W. M. & Gardiner, E. E. Imaging platelet processes and function—current and emerging approaches for imaging in vitro and in vivo. Front. Immunol. 11, 78 (2020).
Jiang, Y., Fu, H., Springer, T. A. & Wong, W. P. Electrostatic steering enables flow-activated von Willebrand factor to bind platelet glycoprotein, revealed by single-molecule stretching and imaging. J. Mol. Biol. 431, 1380–1396 (2019).
Schurr, Y. et al. Platelet lamellipodium formation is not required for thrombus formation and stability. Blood 134, 2318–2329 (2019).
Liu, Y. et al. Molecular imaging of inflammation and platelet adhesion in advanced atherosclerosis effects of antioxidant therapy with NADPH oxidase inhibition. Circ. Cardiovasc. Imaging 6, 74–82 (2013).
Gaertner, F. et al. Migrating platelets are mechano-scavengers that collect and bundle bacteria. Cell 171, 1368–1382 (2017).
Sreeramkumar, V. et al. Neutrophils scan for activated platelets to initiate inflammation. Science 346, 1234–1238 (2014).
Schermelleh, L. et al. Super-resolution microscopy demystified. Nat. Cell Biol. 21, 72–84 (2019).
Mayr, S. et al. Localization microscopy of actin cytoskeleton in human platelets. Int. J. Mol. Sci. 19, 1150 (2018).
Lickert, S. et al. Morphometric analysis of spread platelets identifies integrin αIIbβ3-specific contractile phenotype. Sci. Rep. 8, 5428 (2018).
Bergstrand, J. et al. Super-resolution microscopy can identify specific protein distribution patterns in platelets incubated with cancer cells. Nanoscale 11, 10023–10033 (2019).
Feghhi, S. et al. Glycoprotein Ib–IX–V complex transmits cytoskeletal forces that enhance platelet adhesion. Biophys. J. 111, 601–608 (2016).
Lam, W. A. et al. Mechanics and contraction dynamics of single platelets and implications for clot stiffening. Nat. Mater. 10, 61–66 (2011).
Frydman, G. H. et al. Technical advance: changes in neutrophil migration patterns upon contact with platelets in a microfluidic assay. J. Leukoc. Biol. 101, 797–806 (2017).
Johnston, I. et al. Recognition of PF4–VWF complexes by heparin-induced thrombocytopenia antibodies contributes to thrombus propagation. Blood 135, 1270–1280 (2020).
Isozaki, A. et al. A practical guide to intelligent image-activated cell sorting. Nat. Protoc. 14, 2370–2415 (2019).
Nitta, N. et al. Intelligent image-activated cell sorting. Cell 175, 266–276 (2018).
Zhou, Y. et al. Intelligent classification of platelet aggregates by agonist type. eLife 9, e52938 (2020).
Sachs, L., Denker, C., Greinacher, A. & Palankar, R. Quantifying single-platelet biomechanics: an outsider’s guide to biophysical methods and recent advances. Res. Pract. Thromb. Haemost. 4, 386–401 (2020).
Joshi, S. et al. Alterations in platelet secretion differentially affect thrombosis and hemostasis. Blood Adv. 2, 2187–2198 (2018).
Nurden, P. et al. Immunolocalization of P2Y1 and TPα receptors in platelets showed a major pool associated with the membranes of α-granules and the open canalicular system. Blood 101, 1400–1408 (2003).
Ponomareva, A. A. et al. Intracellular origin and ultrastructure of platelet-derived microparticles. J. Thromb. Haemost. 15, 1655–1667 (2017).
Lee, S. H. et al. Inducing mitophagy in diabetic platelets protects against severe oxidative stress. EMBO Mol. Med. 8, 779–795 (2016).
Tomaiuolo, M., Litvinov, R. I., Weisel, J. W. & Stalker, T. J. Use of electron microscopy to study platelets and thrombi. Platelets 31, 580–588 (2020).
Clauser, S. & Cramer-Borde, E. Role of platelet electron microscopy in the diagnosis of platelet disorders. Semin. Thromb. Hemost. 35, 213–223 (2009).
Rand, M. L., Reddy, E. C. & Israels, S. J. Laboratory diagnosis of inherited platelet function disorders. Transfus. Apher. Sci. 57, 485–493 (2018).
Brisson, A. R., Tan, S., Linares, R., Gounou, C. & Arraud, N. Extracellular vesicles from activated platelets: a semiquantitative cryo-electron microscopy and immuno-gold labeling study. Platelets 28, 263–271 (2017).
Ma, Q. et al. Mitochondrial PIP3-binding protein FUNDC2 supports platelet survival via AKT signaling pathway. Cell Death Differ. 26, 321–331 (2019).
Erlandsen, S. L., Greet Bittermann, A., White, J., Leith, A. & Marko, M. High-resolution CryoFESEM of individual cell adhesion molecules (CAMs) in the glycocalyx of human platelets: detection of P-selectin (CD62P), GPI–IX complex (CD42A/CD42bα,bβ), and integrin GPIIbIIIa (CD41/CD61) by immunogold labeling and stereo imaging. J. Histochem. Cytochem. 49, 809–819 (2001).
Gresele, P. & Subcommittee on Platelet Physiology of the International Society on Thrombosis and Hemostasis. Diagnosis of inherited platelet function disorders: guidance from the SSC of the ISTH. J. Thromb. Haemost. 13, 314–322 (2015).
Brunet, J. G. et al. Electron microscopy examination of platelet whole mount preparations to quantitate platelet dense granule numbers: implications for diagnosing suspected platelet function disorders due to dense granule deficiency. Int. J. Lab. Hematol. 40, 400–407 (2018).
Weisel, J. W. & Litvinov, R. I. Fibrin formation, structure and properties. Subcell. Biochem. 82, 405–456 (2017).
Weisel, J. W. & Litvinov, R. I. Visualizing thrombosis to improve thrombus resolution. Res. Pract. Thromb. Haemost. 5, 38–50 (2021).
Daraei, A. et al. Automated fiber diameter and porosity measurements of plasma clots in scanning electron microscopy images. Biomolecules 11, 1536 (2021).
Van de Walle, A. B. et al. The role of fibrinogen spacing and patch size on platelet adhesion under flow. Acta Biomater. 8, 4080–4091 (2012).
Agbani, E. O. et al. Coordinated membrane ballooning and procoagulant spreading in human platelets. Circulation 132, 1414–1424 (2015).
Tomaiuolo, M. et al. Interrelationships between structure and function during the hemostatic response to injury. Proc. Natl Acad. Sci. USA 116, 2243–2252 (2019).
Chernysh, I. N. et al. The distinctive structure and composition of arterial and venous thrombi and pulmonary emboli. Sci. Rep. 10, 5112 (2020).
Engberts, K. B., Seinen, C., Geerts, W. J. C. & Heijnen, H. F. G. Electron tomography and correlative approaches in platelet studies. Methods Mol. Biol. 1812, 55–79 (2018).
Baidildinova, G. et al. Soluble platelet release factors as biomarkers for cardiovascular disease. Front. Cardiovasc. Med. 8, 684920 (2021).
Montague, S. J., Andrews, R. K. & Gardiner, E. E. Mechanisms of receptor shedding in platelets. Blood 132, 2535–2545 (2018).
Fong, K. P. et al. Deciphering the human platelet sheddome. Blood 117, e15–e26 (2011).
Fritsma, G. A. & McGlasson, D. L. Whole blood platelet aggregometry. Methods Mol. Biol. 1646, 333–347 (2017).
Moenen, F. et al. Screening for platelet function disorders with Multiplate and platelet function analyzer. Platelets 30, 81–87 (2019).
Larsen, P. D. et al. Comparison of Multiplate and VerifyNow platelet function tests in predicting clinical outcome in patients with acute coronary syndromes. Thromb. Res. 152, 14–19 (2017).
Dichiara, J. et al. Platelet function measured by VerifyNow identifies generalized high platelet reactivity in aspirin treated patients. Platelets 18, 414–423 (2007).
Morel-Kopp, M. C. et al. Heparin-induced multi-electrode aggregometry method for heparin-induced thrombocytopenia testing: communication from the SSC of the ISTH. J. Thromb. Haemost. 14, 2548–2552 (2016).
Jin, J. et al. Implementation of whole-blood impedance aggregometry for heparin-induced thrombocytopenia functional assay and case discussion. Am. J. Clin. Pathol. 152, 50–58 (2019).
Haas, T., Cushing, M. M., Varga, S., Gilloz, S. & Schmugge, M. Usefulness of multiple electrode aggregometry as a screening tool for bleeding disorders in a pediatric hospital. Platelets 30, 498–505 (2019).
Tsantes, A. E. et al. The prognostic value of multiple electrode aggregometry and light transmittance aggregometry in stable cardiovascular patients with type 2 diabetes mellitus. Thromb. Res. 180, 47–54 (2019).
Ferrer-Marin, F. & Sola-Visner, M. Neonatal platelet physiology and implications for transfusion. Platelets 33, 14–22 (2021).
Selby, R. “TEG talk”: expanding clinical roles for thromboelastography and rotational thromboelastometry. Hematology Am. Soc. Hematol. Educ. Program 2020, 67–75 (2020).
Chen, A. & Teruya, J. Global hemostasis testing thromboelastography: old technology, new applications. Clin. Lab. Med. 29, 391–407 (2009).
Collyer, T. C., Gray, D. J., Sandhu, R., Berridge, J. & Lyons, G. Assessment of platelet inhibition secondary to clopidogrel and aspirin therapy in preoperative acute surgical patients measured by Thrombelastography Platelet Mapping. Br. J. Anaesth. 102, 492–498 (2009).
Gurbel, P. A. et al. First report of the point-of-care TEG: a technical validation study of the TEG-6S system. Platelets 27, 642–649 (2016).
Dias, J. D. et al. TEG(R)6s system measures the contributions of both platelet count and platelet function to clot formation at the site-of-care. Platelets 31, 932–938 (2020).
Tantry, U. S. et al. The role of viscoelastic testing in assessing peri-interventional platelet function and coagulation. Platelets https://doi.org/10.1080/09537104.2021.1961709 (2021).
Gurbel, P. A. et al. Bedside thromboelastography to rapidly assess the pharmacodynamic response of anticoagulants and aspirin in COVID-19: evidence of inadequate therapy in a predominantly minority population. J. Thromb. Thrombolysis 51, 902–904 (2021).
Brass, L. F. et al. In Platelets 4th edn. (ed. Michelson, A. D.) 371–391 (Academic Press, 2019).
Kundu, S. K. et al. Description of an in vitro platelet function analyzer—PFA-100. Semin. Thromb. Hemost. 21, 106–112 (1995).
Koessler, J. et al. The new INNOVANCE® PFA P2Y carttection of the P2Y12 receptor inhibition. Platelets 22, 20–27 (2011).
Favaloro, E. J. Clinical utility of closure times using the platelet function analyzer-100/200. Am. J. Hematol. 92, 398–404 (2017).
Jain, A. et al. A shear gradient-activated microfluidic device for automated monitoring of whole blood haemostasis and platelet function. Nat. Commun. 7, 10176 (2016).
Herbig, B. A., Yu, X. & Diamond, S. L. Using microfluidic devices to study thrombosis in pathological blood flows. Biomicrofluidics 12, 042201 (2018).
Al Ghaithi, R. et al. Evaluation of the Total Thrombus-Formation System (T-TAS): application to human and mouse blood analysis. Platelets 30, 893–900 (2019).
Zheng, K. L. et al. The Total Thrombus Formation (T-TAS) platelet (PL) assay, a novel test that evaluates whole blood platelet thrombus formation under physiological conditions. Platelets https://doi.org/10.1080/09537104.2021.1882669 (2021).
Kaikita, K., Hosokawa, K., Dahlen, J. R. & Tsujita, K. Total Thrombus-Formation Analysis System (T-TAS): clinical application of quantitative analysis of thrombus formation in cardiovascular disease. Thromb. Haemost. 119, 1554–1562 (2019).
Ting, L. H. et al. Contractile forces in platelet aggregates under microfluidic shear gradients reflect platelet inhibition and bleeding risk. Nat. Commun. 10, 1204 (2019).
Mangin, P. H. et al. In vitro flow based systems to study platelet function and thrombus formation: recommendations for standardization: communication from the SSC on biorheology of the ISTH. J. Thromb. Haemost. 18, 748–752 (2020).
McMullen, P. D. et al. A descriptive and quantitative immunohistochemical study demonstrating a spectrum of platelet recruitment patterns across pulmonary infections including COVID-19. Am. J. Clin. Pathol. 155, 354–363 (2021).
Bertolin, A. J. et al. Platelet reactivity and coagulation markers in patients with COVID-19. Adv. Ther. 38, 3911–3923 (2021).
Wool, G. D. & Miller, J. L. The impact of COVID-19 disease on platelets and coagulation. Pathobiology 88, 15–27 (2021).
Zhang, Y. et al. Mechanisms involved in the development of thrombocytopenia in patients with COVID-19. Thromb. Res. 193, 110–115 (2020).
Manne, B. K. et al. Platelet gene expression and function in patients with COVID-19. Blood 136, 1317–1329 (2020).
Hottz, E. D. et al. Platelet activation and platelet–monocyte aggregate formation trigger tissue factor expression in patients with severe COVID-19. Blood 136, 1330–1341 (2020).
Althaus, K. et al. Antibody-induced procoagulant platelets in severe COVID-19 infection. Blood 137, 1061–1071 (2021).
Bongiovanni, D. et al. SARS-CoV-2 infection is associated with a pro-thrombotic platelet phenotype. Cell Death Dis. 12, 50 (2021).
Nishikawa, M. et al. Massive image-based single-cell profiling reveals high levels of circulating platelet aggregates in patients with COVID-19. Nat. Commun. 12, 7135 (2021).
Koupenova, M. et al. SARS-CoV-2 initiates programmed cell death in platelets. Circ. Res. 129, 631–646 (2021).
Meizlish, M. L. et al. Intermediate-dose anticoagulation, aspirin, and in-hospital mortality in COVID-19: a propensity score-matched analysis. Am. J. Hematol. 96, 471–479 (2021).
Chow, J. H. et al. Aspirin use is associated with decreased mechanical ventilation, intensive care unit admission, and in-hospital mortality in hospitalized patients with coronavirus disease 2019. Anesth. Analg. 132, 930–941 (2021).
Gurbel, P. A. et al. Thrombogenicity markers for early diagnosis and prognosis in COVID-19: a change from the current paradigm?. Blood Coagul. Fibrinolysis 32, 544–549 (2021).
Schultz, N. H. et al. Thrombosis and thrombocytopenia after ChAdOx1 nCoV-19 vaccination. N. Engl. J. Med. 384, 2124–2130 (2021).
Greinacher, A. et al. Thrombotic thrombocytopenia after ChAdOx1 nCov-19 vaccination. N. Engl. J. Med. 384, 2092–2101 (2021).
Coenen, D. M., Mastenbroek, T. G. & Cosemans, J. Platelet interaction with activated endothelium: mechanistic insights from microfluidics. Blood 130, 2819–2828 (2017).
Mussbacher, M., Pirabe, A., Brunnthaler, L., Schrottmaier, W. C. & Assinger, A. Horizontal microRNA transfer by platelets—evidence and implications. Front. Physiol. 12, 678362 (2021).
Lisman, T. Platelet–neutrophil interactions as drivers of inflammatory and thrombotic disease. Cell Tissue Res. 371, 567–576 (2018).
Finsterbusch, M., Schrottmaier, W. C., Kral-Pointner, J. B., Salzmann, M. & Assinger, A. Measuring and interpreting platelet–leukocyte aggregates. Platelets 29, 677–685 (2018).
Schrottmaier, W. C., Mussbacher, M., Salzmann, M. & Assinger, A. Platelet–leukocyte interplay during vascular disease. Atherosclerosis 307, 109–120 (2020).
Klatt, C. et al. Platelet–RBC interaction mediated by FasL/FasR induces procoagulant activity important for thrombosis. J. Clin. Invest. 128, 3906–3925 (2018).
Shi, G. et al. Platelet factor 4 mediates vascular smooth muscle cell injury responses. Blood 121, 4417–4427 (2013).
Qu, M. et al. Platelet-derived microparticles enhance megakaryocyte differentiation and platelet generation via miR-1915-3p. Nat. Commun. 11, 4964 (2020).
Malehmir, M. et al. Platelet GPIbα is a mediator and potential interventional target for NASH and subsequent liver cancer. Nat. Med. 25, 641–655 (2019).
Suzuki-Inoue, K. Platelets and cancer-associated thrombosis: focusing on the platelet activation receptor CLEC-2 and podoplanin. Hematology Am. Soc. Hematol. Educ. Program 2019, 175–181 (2019).
Yu, L. et al. Bidirectional interaction between cancer cells and platelets provides potential strategies for cancer therapies. Front. Oncol. 11, 764119 (2021).
Boilard, E. et al. Influenza virus H1N1 activates platelets through FcγRIIA signaling and thrombin generation. Blood 123, 2854–2863 (2014).
Thachil, J. What do monitoring platelet counts in COVID-19 teach us? J. Thromb. Haemost. 18, 2071–2072 (2020).
Celi, A. et al. Thrombus formation: direct real-time observation and digital analysis of thrombus assembly in a living mouse by confocal and widefield intravital microscopy. J. Thromb. Haemost. 1, 60–68 (2003).
Pittet, M. J. & Weissleder, R. Intravital imaging. Cell 147, 983–991 (2011).
Cooley, B. C. In vivo fluorescence imaging of large-vessel thrombosis in mice. Arterioscler. Thromb. Vasc. Biol. 31, 1351–1356 (2011).
Hagberg, I. A. & Lyberg, T. Blood platelet activation evaluated by flow cytometry: optimised methods for clinical studies. Platelets 11, 137–150 (2000).
Loosse, C., Swieringa, F., Heemskerk, J. W. M., Sickmann, A. & Lorenz, C. Platelet proteomics: from discovery to diagnosis. Expert Rev. Proteomics 15, 467–476 (2018).
Han, X., Yang, K. & Gross, R. W. Multi-dimensional mass spectrometry-based shotgun lipidomics and novel strategies for lipidomic analyses. Mass Spectrom. Rev. 31, 134–178 (2012).
Alhasan, A. A. et al. Circular RNA enrichment in platelets is a signature of transcriptome degradation. Blood 127, e1–e11 (2016).
Chen, F. et al. A randomized clinical trial comparing point-of-care platelet function assays and bleeding time in healthy subjects treated with aspirin or clopidogrel. Platelets 23, 249–258 (2012).
Gresele, P. et al. Diagnosis of suspected inherited platelet function disorders: results of a worldwide survey. J. Thromb. Haemost. 12, 1562–1569 (2014).
Pai, M. et al. Diagnostic usefulness of a lumi-aggregometer adenosine triphosphate release assay for the assessment of platelet function disorders. Am. J. Clin. Pathol. 136, 350–358 (2011).
Campbell, J., Ridgway, H. & Carville, D. Plateletworks: a novel point of care platelet function screen. Mol. Diagn. Ther. 12, 253–258 (2008).
Thomas, S. G. In Platelets 4th edn. (ed. Michelson, A. D.) 47–77 (Academic Press, 2019).
Clemetson, K. J. & Clemetson, J. M. In Platelets 4th edn. (ed. Michelson, A. D.) 169–192 (Academic Press, 2019).
Gawaz, M. & Borst, O. In Platelets 4th edn. (ed. Michelson, A. D.) 459–467 (Academic Press, 2019).
Cazenave, J. P. et al. Preparation of washed platelet suspensions from human and rodent blood. Methods Mol. Biol. 272, 13–28 (2004).
Cattaneo, M. et al. Recommendations for the standardization of light transmission aggregometry: a consensus of the Working Party from the Platelet Physiology Subcommittee of SSC/ISTH. J. Thromb. Haemost. 11, 1183–1189 (2013).
Janssen, P. W. et al. The effect of correcting VerifyNow P2Y12 assay results for hematocrit in patients undergoing percutaneous coronary interventions. J. Thromb. Haemost. 15, 618–623 (2017).
Acknowledgements
We thank the platelet research community for the rapid exciting research and clinical progress in the past 2 decades. Due to space limitations, we are unfortunately not able to cite a substantial proportion of this important work and, in some cases, have had to highlight outstanding reviews that should be a resource for further important citations. We are grateful for the following support: a JH Millstone award (S.X.G.); NIH NHLBI T32HL098069 (V.W.G.); National Health and Medical Research Council of Australia and Australian Research Council awards (E.E.G.); NIH NHLBI R01HL142090 and R01HL146101 (K.A.M.); the National Natural Science Foundation of China (grant nos. 81970437 and 82022003) (W.H.T.) and NHLBI R01HL115247, R01HL122815 and R01HL150515 (J.H.).
Author information
Authors and Affiliations
Contributions
All authors were involved in planning the review. K.J., T.T., W.H.T. and J.H. performed the major literature search and wrote the first draft. All remaining authors substantially contributed to their individual area of expertise, reviewed the manuscript, constructed graphics and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Cardiovascular Research thanks Alison Goodall, and the other, anonymous reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tyagi, T., Jain, K., Gu, S.X. et al. A guide to molecular and functional investigations of platelets to bridge basic and clinical sciences. Nat Cardiovasc Res 1, 223–237 (2022). https://doi.org/10.1038/s44161-022-00021-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s44161-022-00021-z
- Springer Nature Limited
This article is cited by
-
Blood platelet factor 4: the elixir of brain rejuvenation
Molecular Neurodegeneration (2024)
-
The association of the platelet/high-density lipoprotein cholesterol ratio with self-reported stroke and cardiovascular mortality: a population-based observational study
Lipids in Health and Disease (2024)
-
Insights into platelet pharmacology from a cryo-EM structure of the ABCC4 transporter
Nature Cardiovascular Research (2023)
-
High Autophagy Patterns in Swelling Platelets During Apheresis Platelet Storage
Indian Journal of Hematology and Blood Transfusion (2023)
-
Baseline platelet count may predict short-term functional outcome of cerebral infarction
BMC Neurology (2022)