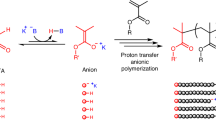
Abstract
Anion-binding interactions in nature have enabled the development of organocatalytic transformations; however, even though ionic species act as intermediates or precursors in many polymerizations, these interactions are underappreciated in polymerization catalysis. Here we introduce a powerful anion-binding catalytic strategy for cationic polymerization. In our approach, selenocyclodiphosph(V)azanes were designed as bench-stable hydrogen-bond donors to reversibly activate dormant covalent bonds (C–X, X=Cl, carboxylate and phosphate), in turn to precisely control the equilibrium between dormant covalent precursors and active cationic species under mild conditions. Experimental and computational analysis of this catalytic system revealed the key role of non-covalent anion-binding interactions between the catalyst and substrates. The living and controlled nature of this strategy, coupled with its capability for recycling catalysts and addressing certain fundamental constraints, such as metal residue and rigorous reaction conditions, delivers a versatile and robust living cationic polymerization methodology for precision polymer synthesis.






Similar content being viewed by others
Data availability
All data that support the findings of this study are available within the article and its Supplementary Information. Crystallographic data for the structures in this article have been deposited at the Cambridge Crystallographic Data Centre (CCDC) under deposition nos. 2125756 (5a·CH3CN) and 2142031 (5a·TEACl). Copies of the data can be obtained free of charge from www.ccdc.cam.ac.uk/structures/.
References
Dutzler, R., Campbell, E. B., Cadene, M., Chait, B. T. & MacKinnon, R. X-ray structure of a ClC chloride channel at 3.0 Å reveals the molecular basis of anion selectivity. Nature 415, 287–294 (2002).
Davis, A. P., Sheppard, D. N. & Smith, B. D. Development of synthetic membrane transporters for anions. Chem. Soc. Rev. 36, 348–357 (2007).
Hedstrom, L. Serine protease mechanism and specificity. Chem. Rev. 102, 4501–4524 (2002).
Zhang, Z. & Schreiner, P. R. (Thio)urea organocatalysis—what can be learnt from anion recognition? Chem. Soc. Rev. 38, 1187–1198 (2009).
Phipps, R. J., Hamilton, G. L. & Toste, F. D. The progression of chiral anions from concepts to applications in asymmetric catalysis. Nat. Chem. 4, 603–614 (2012).
Brak, K. & Jacobsen, E. N. Asymmetric ion-pairing catalysis. Angew. Chem. Int. Ed. 52, 534–561 (2013).
Neel, A. J., Hilton, M. J., Sigman, M. S. & Toste, F. D. Exploiting non-covalent π interactions for catalyst design. Nature 543, 637–646 (2017).
Macheño, O. G. (ed.) Anion-Binding Catalysis (Wiley-VCH GmbH, 2022).
Dove, A. P., Pratt, R. C., Lohmeijer, B. G. G., Waymouth, R. M. & Hedrick, J. L. Thiourea-based bifunctional organocatalysis: supramolecular recognition for living polymerization. J. Am. Chem. Soc. 127, 13798–13799 (2005).
Zhu, J.-B. & Chen, E. Y. X. From meso-lactide to isotactic polylactide: epimerization by B/N Lewis pairs and kinetic resolution by organic catalysts. J. Am. Chem. Soc. 137, 12506–12509 (2015).
Zhang, X., Jones, G. O., Hedrick, J. L. & Waymouth, R. M. Fast and selective ring-opening polymerizations by alkoxides and thioureas. Nat. Chem. 8, 1047–1053 (2016).
Lin, B. & Waymouth, R. M. Urea anions: simple, fast, and selective catalysts for ring-opening polymerizations. J. Am. Chem. Soc. 139, 1645–1652 (2017).
Li, M. et al. Synergetic organocatalysis for eliminating epimerization in ring-opening polymerizations enables synthesis of stereoregular isotactic polyester. J. Am. Chem. Soc. 141, 281–289 (2019).
Thomas, C. & Bibal, B. Hydrogen-bonding organocatalysts for ring-opening polymerization. Green Chem. 16, 1687–1699 (2014).
Ottou, W. N., Sardon, H., Mecerreyes, D., Vignolle, J. & Taton, D. Update and challenges in organo-mediated polymerization reactions. Prog. Polym. Sci. 56, 64–115 (2016).
Li, M. et al. Unimolecular anion-binding catalysts for selective ring-opening polymerization of O-carboxyanhydrides. Angew. Chem. Int. Ed. 60, 6003–6012 (2021).
Kato, M., Kamigaito, M., Sawamoto, M. & Higashimura, T. Polymerization of methyl methacrylate with the carbon tetrachloride/dichlorotris(triphenylphosphine)ruthenium(II)/methylaluminum bis(2,6-di-tert-butylphenoxide) initiating system: possibility of living radical polymerization. Macromolecules 28, 1721–1723 (1995).
Wang, J.-S. & Matyjaszewski, K. Controlled/‘living’ radical polymerization. Atom transfer radical polymerization in the presence of transition-metal complexes. J. Am. Chem. Soc. 117, 5614–5615 (1995).
Grubbs, R. H. & Tumas, W. Polymer synthesis and organotransition metal chemistry. Science 243, 907–915 (1989).
Miyamoto, M., Sawamoto, M. & Higashimura, T. Living polymerization of isobutyl vinyl ether with hydrogen iodide/iodine initiating system. Macromolecules 17, 265–268 (1984).
Faust, R. & Kennedy, J. P. Living carbocationic polymerization. Polym. Bull. 15, 317–323 (1986).
Sawamoto, M. Modern cationic vinyl polymerization. Prog. Polym. Sci. 16, 111–172 (1991).
Matyjaszewski, K. & Sigwalt, P. Unified approach to living and non-living cationic polymerization of alkenes. Polym. Int. 35, 1–26 (1994).
Sigwalt, P. & Moreau, M. Carbocationic polymerization: mechanisms and kinetics of propagation reactions. Prog. Polym. Sci. 31, 44–120 (2006).
Aoshima, S. & Kanaoka, S. A renaissance in living cationic polymerization. Chem. Rev. 109, 5245–5287 (2009).
Michaudel, Q., Kottisch, V. & Fors, B. P. Cationic polymerization: from photoinitiation to photocontrol. Angew. Chem. Int. Ed. 56, 9670–9679 (2017).
Kamigaito, M. & Sawamoto, M. Synergistic advances in living cationic and radical polymerizations. Macromolecules 53, 6749–6753 (2020).
Uchiyama, M., Satoh, K. & Kamigaito, M. Cationic RAFT and DT polymerization. Prog. Polym. Sci. 124, 101485 (2022).
Uchiyama, M., Satoh, K. & Kamigaito, M. Cationic RAFT polymerization using ppm concentrations of organic acid. Angew. Chem. Int. Ed. 54, 1924–1928 (2015).
Teator, A. J. & Leibfarth, F. A. Catalyst-controlled stereoselective cationic polymerization of vinyl ethers. Science 363, 1439–1443 (2019).
Kottisch, V. et al. Controlled cationic polymerization: single-component initiation under ambient conditions. J. Am. Chem. Soc. 141, 10605–10609 (2019).
Kottisch, V. et al. Hydrogen bond donor-catalyzed cationic polymerization of vinyl ethers. Angew. Chem. Int. Ed. 60, 4535–4539 (2021).
Zhang, X., Jiang, Y., Ma, Q., Hu, S. & Liao, S. Metal-free cationic polymerization of vinyl ethers with strict temporal control by employing an organophotocatalyst. J. Am. Chem. Soc. 143, 6357–6362 (2021).
Abel, B. A., Snyder, R. L. & Coates, G. W. Chemically recyclable thermoplastics from reversible-deactivation polymerization of cyclic acetals. Science 373, 783–789 (2021).
Lin, S. & Jacobsen, E. N. Thiourea-catalysed ring opening of episulfonium ions with indole derivatives by means of stabilizing non-covalent interactions. Nat. Chem. 4, 817–824 (2012).
Ford, D. D., Lehnherr, D., Kennedy, C. R. & Jacobsen, E. N. On- and off-cycle catalyst cooperativity in anion-binding catalysis. J. Am. Chem. Soc. 138, 7860–7863 (2016).
Kennedy, C. R. et al. Mechanism-guided development of a highly active bis-thiourea catalyst for anion-abstraction catalysis. J. Am. Chem. Soc. 138, 13525–13528 (2016).
Zhao, C., Sojdak, C. A., Myint, W. & Seidel, D. Reductive etherification via anion-binding catalysis. J. Am. Chem. Soc. 139, 10224–10227 (2017).
Chen, L., Berry, S. N., Wu, X., Howe, E. N. W. & Gale, P. A. Advances in anion receptor chemistry. Chem 6, 61–141 (2020).
Busschaert, N., Caltagirone, C., Van Rossom, W. & Gale, P. A. Applications of supramolecular anion recognition. Chem. Rev. 115, 8038–8155 (2015).
Plajer, A. J. et al. Tailoring the binding properties of phosphazane anion receptors and transporters. J. Am. Chem. Soc. 141, 8807–8815 (2019).
Gheewala, C. D., Collins, B. E. & Lambert, T. H. An aromatic ion platform for enantioselective Brønsted acid catalysis. Science 351, 961–965 (2016).
Kütt, A. et al. Strengths of acids in acetonitrile. Eur. J. Org. Chem. 2021, 1407–1419 (2021).
Sawamoto, M., Kamigaito, M. & Higashimura, T. Living cationic polymerization of isobutyl vinyl ether by the diphenyl phosphate/zinc iodide initiating system. Polym. Bull. 20, 407–412 (1988).
Thordarson, P. Determining association constants from titration experiments in supramolecular chemistry. Chem. Soc. Rev. 40, 1305–1323 (2011).
Kamigaito, M., Maeda, Y., Sawamoto, M. & Higashimura, T. Living cationic polymerization of isobutyl vinyl ether by hydrogen chloride/Lewis acid initiating systems in the presence of salts: in-situ direct NMR analysis of the growing species. Macromolecules 26, 1643–1649 (1993).
Ford, D. D., Lehnherr, D., Kennedy, C. R. & Jacobsen, E. N. Anion-abstraction catalysis: the cooperative mechanism of α-chloroether activation by dual hydrogen-bond donors. ACS Catal. 6, 4616–4620 (2016).
Johnson, E. R. et al. Revealing noncovalent interactions. J. Am. Chem. Soc. 132, 6498–6506 (2010).
Takagi, K., Yamauchi, K. & Murakata, H. Halogen-bonding-mediated and controlled cationic polymerization of isobutyl vinyl ether: expanding the catalytic scope of 2-iodoimidazolium salts. Chem. Eur. J. 23, 9495–9500 (2017).
Haraguchi, R., Nishikawa, T., Kanazawa, A. & Aoshima, S. Metal-free living cationic polymerization using diaryliodonium salts as organic Lewis acid catalysts. Macromolecules 53, 4185–4192 (2020).
Takagi, K., Murakata, H., Yamauchi, K. & Hashimoto, K. Cationic polymerization of vinyl monomers using halogen bonding organocatalysts with varied activity. Polym. Chem. 11, 6739–6744 (2020).
Takagi, K., Sakakibara, N., Hasegawa, T. & Hayashi, S. Controlled/living cationic polymerization of p-methoxystyrene using tellurium-based chalcogen bonding catalyst—discovery of a new water-tolerant Lewis acid catalyst. Macromolecules 55, 3671–3680 (2022).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (grant U21A2089, Y.T., and grant 22001243, M.L.) and the Jilin Science and Technology Bureau (Grant 20210402066GH, M.L.). We thank X. Zhang for helpful discussions with the theoretical calculations. We thank the Molecular Scale Lab at Shenzhen University for the MALDI–TOF MS characterization.
Author information
Authors and Affiliations
Contributions
M.L. and Y.T. developed the concept, and co-wrote the manuscript. M.L. and Y.T. designed the experiments, and analysed the results. M.L., Z.Z., Y.Y. and W.L. performed the experiments. Z.L. performed the MALDI–TOF MS experiments. M.L. performed the DFT calculations. Y.T. and X.W. directed the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Frank Leibfarth and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary handling editor: Alison Stoddart, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–49, Tables 1–4, materials, experimental procedures, product characterization, computational methodology and supplementary discussion.
Supplementary Data 1
Crystallographic data for compound 5a, CCDC 2125756.
Supplementary Data 2
Crystallographic data for compound 5a•TEACl, CCDC 2142031.
Supplementary Data 3
DFT computational data in .log files for all the transition states and intermediates.
Source data
Source Data Fig. 2
Statistical Source Data.
Source Data Fig. 3
Statistical Source Data.
Source Data Fig. 4
Statistical Source Data.
Source Data Fig. 5
Statistical Source Data.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, M., Zhang, Z., Yan, Y. et al. Anion-binding catalysis enables living cationic polymerization. Nat. Synth 1, 815–823 (2022). https://doi.org/10.1038/s44160-022-00142-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s44160-022-00142-0
- Springer Nature Limited
This article is cited by
-
Organic Brønsted Acid-Catalyzed Stereoselective Cationic RAFT Polymerization: The Effect of RAFT Agents
Chinese Journal of Polymer Science (2024)