Abstract
As thermal stress and disease outbreaks decimate coral reefs throughout the tropics, there is growing evidence that higher latitude marine environments may provide crucial refuges for many at-risk, temperature-sensitive coral species. However, our understanding of how coral populations expand into new areas and sustain themselves over time is constrained by the limited scope of modern observations. Here, we provide geological insights into coral range expansions by reconstructing the composition of a Late Holocene-aged subfossil coral death assemblage on the southeast Florida reef tract and comparing it to modern reefs throughout the region. Our findings show that the Late Holocene coral assemblages were dominated by now critically endangered Acropora species between ~3500 and 1800 years before present, mirroring classic zonation patterns characteristic of healthy pre-1970s Caribbean reefs. In contrast, the modern reefs off southeast Florida are becoming increasingly dominated by stress-tolerant species like Porites astreoides and Siderastrea siderea despite modest expansions of Acropora cervicornis over the past several decades. Our results suggest that ongoing anthropogenic stressors, not present during the Late Holocene, are likely limiting the ability of modern higher latitude reefs in Florida to function as long-term climate refugia.
Similar content being viewed by others
Introduction
Coral reefs are among the planet’s most biologically diverse and economically valuable ecosystems, supporting over 25% of all marine species and providing critical goods and services to millions living in coastal communities worldwide1. However, over the past several decades, rising ocean temperatures and the increased frequency of extreme thermal stress events have caused substantial declines in the health of coral reef ecosystems globally2,3. This trend is particularly evident in the western Atlantic region, where climate-driven bleaching and disease outbreaks have resulted in widespread mortality of key reef-building coral species, including Acropora palmata, Acropora cervicornis, and Orbicella spp. complex2,4,5,6. Although historically dominant on western Atlantic reefs since at least the late Pleistocene (~600 thousand years ago7), all three species have undergone unprecedented population declines in just a few decades4,8,9,10, prompting their listing as threatened under the U.S. Endangered Species Act in 2006 and 201411,12.
Despite widespread population declines in the tropics, there is growing evidence that warming ocean temperatures are also driving the poleward range expansion of thermally sensitive coral species into some subtropical and temperate marine regions, including Japan13, the Mediterranean Sea14, and Western Australia15. In the western Atlantic, threatened Acropora spp. have also been observed well north of their historical ranges off southeast Florida16,17 and at the Flower Garden Banks in the northern Gulf of Mexico18. These observations suggest that subtropical regions may provide critical ‘refuges’ for tropical reef-building coral assemblages in the face of ongoing climate warming18,19,20. Understanding the ecological dynamics and environmental constraints on coral-reef development at subtropical and temperate latitudes has, therefore, emerged as a crucial objective for both scientists and resource managers19,21.
Although recent trends suggest that tropical coral communities are expanding their ranges poleward into higher latitude reef environments around the globe, it is unclear if those expansions will persist long-term under future climate-change scenarios. Compared to the tropics, higher latitude subtropical and temperate reef environments are highly dynamic, experiencing both cold and warm temperature extremes22,23 and variable aragonite saturation states24. As a result, they are typically characterized by lower coral diversity, slower growth rates, and limited accretion25. Since tropical coral taxa are adapted to a narrow range of environmental conditions22, their long-term persistence in higher latitude environments will depend on their ability to tolerate more dynamic environments with more intense disturbance regimes. However, the increasing rate of global climate change is also expected to result in more severe temperature, wind, and wave events26, posing a substantial threat to higher latitude and tropical reef communities alike19. Thus, as the climate continues to warm, there is considerable uncertainty about whether poleward expanding coral taxa will be more or less susceptible to climate change than in the tropics23.
Paleoecological reconstructions and interpretations of past coral reef environments provide the unique opportunity to examine how coral communities respond to climate and environmental change over temporal scales not available in modern ecological studies23,27,28,29,30,31. They also provide a valuable pre-anthropogenic baseline that can be used to evaluate the state and long-term stability of modern coral reef ecosystems, allowing resource managers to prioritize specific areas for restoration and protection7,10,21. This application is especially useful in determining the potential of higher latitude reef sites to serve as climate refuges, particularly where analogous range expansions occurred in the geological past15,17,23,32. Reconstructions of past coral range expansions from the fossil record not only inform where future expansions might occur, but also provide the necessary framework for determining whether recent range expansions are likely to persist in subtropical or temperate regions threatened by climate and environmental change.
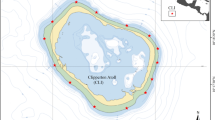
Here, we provide the first comprehensive paleoecological surveys of Late Holocene subfossil coral assemblages recently discovered off the coast of northern Broward County, Florida, in an area herein referred to as Pompano Ridge (26°13’N, 80°5’W) (Fig. 1). The assemblages represent a period of Acropora-dominated reef development corresponding to regional, centennial-scale warming at the northern extent of the Atlantic Warm Pool (AWP) between ~3500 and 1800 years B.P.30. Their well-constrained age structure, combined with high-quality taphonomic preservation, make them particularly well-suited as a baseline for evaluating the potential for modern subtropical range expansions of western Atlantic coral taxa under anthropogenic climate change. By comparing the structure of the Late Holocene coral assemblages to modern ones, we assess the natural range expansion potential of several reef-building coral species relative to their pre-anthropogenic baseline and explore the anthropogenic stressors likely to inhibit further reef development. Additionally, we use taphonomic descriptions and the presence of certain indicator species to reconstruct the overall depositional environment and species zonation across the paleoreef. Our findings have important implications for the present ecological state of southeast Florida’s subtropical reefs and their present and future ability to support coral refugia in the face of climate change.
a Map showing full extent of Southeast Florida reef tract (SFRT; red) with Southeast Florida Coral Reef Evaluation and Monitoring (SECREMP) sites (orange) and Pompano Ridge study area (PMP RIDG; blue) labeled. The Southeast Florida reef tract hardbottom extent is shown in red. Map image is the intellectual property of Esri and is used herein under license. Copyright 2020 Esri and its licensors. All rights reserved. b Bathymetric map of Pompano Ridge study area with depths in meters below mean sea level (bMSL) generated with lidar-based digital elevation models acquired by U.S. Army Corp of Engineers in 2017. Estimated extent of subfossil coral rubble and potential original growth location are bordered with dashed line, and sampling sites marked by black dots (Supplementary Table S1).
Results
Chronological framework
U-Th dating of subfossil Acropora cervicornis (n = 5), Pseudodiploria strigosa (n = 5), and Colpophyllia natans (n = 1) yielded Late Holocene ages ranging between 1937 ± 4 and 3218 ± 6 years B.P. (Supplementary Table 6). These findings align with the previously established age ranges for coral community development within the study area, based on radiometric dating of A. palmata and Orbicella spp. (Fig. 2)30. This was also the case for subfossil Acropora prolifera and Dendrogyra cylindrus samples previously dated by Modys et al.31,33. These results confirm that despite D. cylindrus, A. cervicornis, A. prolifera, Ps. strigosa, and C. natans being documented alive throughout parts of the SFRT34, their subfossil occurrences in the Late Holocene assemblages at Pompano Ridge are temporally distinct. This temporal distinction is likely attributed to the region’s current low live coral cover, which minimizes the inclusion of modern corals in the subfossil record and ensures limited sampling bias from post-depositional intermixing. Furthermore, all but three of the additional coral ages were within error of at least one other previously dated A. palmata or Orbicella spp. sample (Fig. 2; Supplementary Table 6), suggesting coeval growth. Finally, consecutive non-overlapping ages of all subfossil coral samples combined were separated by an average of ~110 years, indicating that our sampling strategy likely captured centennial-scale records of coral community persistence at Pompano Ridge during the Late Holocene.
a Displays the distribution of U-Th ages for various coral taxa. b Shows the density distribution of the U-Th ages, with the bandwidth determined using the default “nrd0” method. The blue points and density curve represent corals dated in the current study and from previous studies, which are not presently alive at the study site. The red points and density curve correspond to previously dated corals from ref. 30, indicating the temporal extent of the Late Holocene range expansion. Apalm = Acropora palmata, Oann = Orbicella spp., Dcyl = Dendrogyra cylindrus, Pstrig = Pseudodiploria strigosa, Acer = Acropora cervicornis, Apro = Acropora prolifera. Plots were generated using the ggplot2 package in R (RStudio Team111).
Late Holocene coral composition
We identified 21 unique coral taxa (six branching, seven massive, and seven with other morphologies) from the 16 Late Holocene pit/transect samples (Supplementary Table 2). Consistent with previous studies evaluating fossil coral assemblages5,35,36, branching coral taxa dominated the Late Holocene assemblages, accounting for almost 90% of the relative coral composition in our samples (Supplementary Table 2). Of the six branching coral taxa, A. palmata and A. cervicornis were most abundant, averaging 43.2% ± 4.8% (mean ± standard error) and 33.6% ± 5.2% of the Late Holocene subfossil record, respectively. Branching varieties of Porites spp., Millepora alcicornis, and Millepora complanata were also relatively common, but together only accounted for ~15% of the Late Holocene record (8.0% ± 1.7%, 4.3% ± 1.1%, and 2.6% ± 1.4%, respectively). The hybrid A. prolifera was also recovered from several pit/transect samples but accounted for less than 0.5% of all identified subfossil corals collected and identified from the Late Holocene assemblages.
Whereas branching coral taxa dominated the Late Holocene assemblages, massive corals accounted for only ~4% of all subfossil coral specimens identified (Supplementary Table 2). Of the seven massive coral taxa identified, only Orbicella spp. accounted for more than 1% of the subfossil composition (2.3% ± 0.8%). Less common massive coral taxa, including Ps. strigosa, Porites astreoides, and D. cylindrus, comprised just under 2% of the Late Holocene assemblages (0.6% ± 0.1%, 0.5% ± 0.2%, and 0.5% ± 0.5%, respectively). Colpophyllia natans and Dichocoenia stokesi were present but rare, with each averaging 0.2% of the relative coral composition in our samples. Of the remaining small, massive (Favia fragum and Siderastrea siderea), plating (Agaricia spp.), free-living (Manicina aereolata, Isophyllia sinuosa), and phaceloid (Eusmilia fastigiata) coral taxa, only Agaricia spp. (2.0% ± 0.5%) averaged more than 0.2% of all identified subfossil branching and massive corals collected from the Late Holocene assemblages.
Preservation and taphonomy
Pits excavated at random points along each of the 16 transects revealed a relatively homogenous internal structure characterized by poorly cemented piles of large A. palmata and A. cervicornis branches and trunks (>20 cm length) infilled by an unconsolidated matrix of sand and coral-dominated gravel. Large Acropora branches and trunks were typically encrusted by crustose coralline algae, sciaphilic foraminifera (columnar Carpentaria utricularis and globulose and branching Homotrema rubrum), and various serpulid worms and bryozoans. Encruster thicknesses ranged from thin, single layers with sparse coverage to thick intergrowths (~1 cm) encompassing entire clasts (Supplementary Fig. 1). Internal coral gravel exhibited a similar range of encrustation and preservation except in pockets with high volumes of unconsolidated sand and finer rubble. In these areas, pristine coral clasts with remarkably well-preserved original corallite structures were found entombed in sediment (Supplementary Fig. 2A). Poorly preserved coral fragments were also intermixed in well-preserved coral gravel layers, some of which were encrusted by pristine early successional corals including Porites spp., Agaricia spp., and Favia fragum (Supplementary Fig. 4). The coral rubble sampled from within the interstitial spaces of larger interlocking A. palmata branches, particularly in areas with high unconsolidated sand, contained the highest species richness identified throughout the study area.
Surficial clusters of subfossil massive corals were found interspersed among lower-relief Acropora-dominated coral rubble accumulations and discontinuous patches of sand. Whereas the larger subfossil colonies (>1 m diameter) occurred in an upright position with no signs of lateral transport, smaller colonies (<1 m diameter) were typically found overturned and deposited in broad, low-relief mounds. The exposed surfaces of both upright and overturned colonies were highly bioeroded and encrusted; however, the buried surfaces of overturned colonies were well-preserved with clearly defined corallites, sparse encrustation (some globulose H. rubrum and serpulids) and little to no bioerosion (Supplementary Fig. 5). Smaller (<0.25 m) subfossil massive corals were occasionally recovered from internal excavation pits; however, the majority were encountered along surface transects.
Spatial variability
Hierarchical cluster analysis of the Late Holocene coral assemblages surveyed at Pompano Ridge identified two distinct transect groups (Fig. 3a). The first group was comprised mainly of shallow (2.8 to 3.5 bMSL) transects located along the eastern margin of the study area, while the second group consisted mostly of western transects located in slightly deeper water (3.5 to 4.5 m bMSL). Statistical comparison revealed significant differences in coral community composition between the two groups (ANOSIM: R = 0.3734, p = 0.004) largely due to dissimilarities in the average relative abundances of A. cervicornis, A. palmata, branching Porites spp., and Orbicella spp. (Fig. 3b; SIMPER average dissimilarity = 31.1%). Specifically, the western transects were characterized by higher relative abundances of A. cervicornis and branching Porites spp., whereas the eastern transects were characterized by higher abundances of A. palmata (Supplementary Table 7). D. cylindrus, C. natans, D. stokesi, and all but one colony of Orbicella spp. were found exclusively at the western transects. Conversely, all coral taxa present in the eastern transects were also present in the western transects.
Species composition from each transect at Pompano Ridge summarized with (a) a dendrogram of the sampling sites created using hierarchical cluster analysis and (b) non-multidimensional scaling (NMDS). In b, colored polygon hulls encompass the transects sampled from the eastern and western area of the study site. The length and direction of species vectors reflect the strength and correlation of the species with the specific sites. Only the three species (Acropora palmata [Apalm], A. cervicornis [Acer], and branching Porites porites [Ppor]) contributing to more than 60% in the similarity percentage (SIMPER) analysis and with envfit p-values below 0.001were plotted. c Representative photographs of eastern transects and (d) western transects taken by ABM in 2018.
Comparison between modern and Late Holocene assemblages
There was a significant difference between the Late Holocene and modern assemblages at Pompano Ridge (ANOSIM: R = 0.7164, p = 0.001; Fig. 4a) mainly due to dissimilarities in the average relative abundances of A. palmata, Po. astreoides, and A. cervicornis (SIMPER average dissimilarity = 76.9%; Supplementary Table 8). Together, A. palmata and A. cervicornis dominated the Late Holocene assemblages, accounting for 48.2% ± 5.3% and 36.2% ± 5.3% of coral relative abundance, respectively. Although A. cervicornis was the second most abundant species in the modern assemblages at Pompano Ridge (20.3% ± 8.8%), it was subdominant to Po. astreoides, which accounted for 42.5% ± 10%. In contrast, Po. astreoides was rare in the Late Holocene assemblage (0.5% ± 0.2%).
a Non-multidimensional scaling (NMDS) plot comparing subfossil and live species composition at Pompano Ridge. Colored polygon hulls encompass transects sampled for modern and subfossil corals across the study area. The length and direction of species vectors reflect the strength and correlation of the species to the specific site. Only the three species (Acropora palmata [Apalm], A. cervicornis [Acer], and Po. astreoides [Past]) contributing to more than 60% in the similarity percentage (SIMPER) analysis and with envfit p-values below 0.001were plotted. b Relative abundances of three species contributing to more than 60% in the similarity percentage (SIMPER) analysis.
The relative species composition of the Late Holocene assemblages at Pompano Ridge was also significantly different from the modern coral assemblages sampled throughout the SFRT by the Southeast Florida Coral Reef Evaluation and Monitoring Program (SECREMP) (ANOSIM, R = 0.7528, p = 0.001; Fig. 5A). SIMPER analysis indicated that average dissimilarity between assemblages was 92.6%, mainly due to just seven species that contributed over 80% to cumulative dissimilarity (Supplementary Table 9). The most striking difference between the modern and Late Holocene assemblages was the shift from A. palmata and A. cervicornis dominance in the Late Holocene to S. siderea and Po. astreoides in the modern records. Whereas A. palmata and A. cervicornis were the most abundant corals in the Late Holocene assemblages, they were absent (A. palmata) and rare (A. cervicornis, 0.2% ± 0.4%) in the modern assemblages. In contrast, S. siderea and Po. astreoides dominated the modern assemblages (35.4% ± 8.8% and 21.5% ± 6.9%, respectively) but were absent (S. siderea) and rare (Po. astreoides, <1%) in the Late Holocene assemblages (Supplementary Table 9). Although our results indicate similar levels of divergence between the Late Holocene assemblages at Pompano Ridge and modern assemblages in 2013 (ANOSIM, R = 0.8039, p = 0.001; SIMPER average dissimilarity = 93.8%; Supplementary Table 10) and 2021 (ANOSIM, R = 0.8284, p = 0.001; SIMPER average dissimilarity = 91.4%, Supplementary Table 11), the relative abundances of A. cervicornis, S. siderea, and Po. astreoides increased by ~300%, ~47%, and ~45%, respectively between 2013 and 2021.
A Non-multidimensional scaling (NMDS) plot comparing subfossil species composition at Pompano Ridge and the live species composition extrapolated from SECREMP survey sites. Colored polygon hulls encompass transects sampled for live and subfossil corals from the Late Holocene, 2013, and 2021 in the study area. Representations for each county are as follows: triangles for Miami-Dade, points for Broward, squares for Palm Beach, and crosses for Martin County. The length and direction of species vectors reflect the strength and correlation of the species to the specific site. A total of four species (Acropora cervicornis [Acer], Acropora palmata [Apalm], Porites astreoides [Past], and Siderastrea siderea [Sidsid]) contributing to more than 60% in the similarity percentage (SIMPER) analysis and with envfit p-values below 0.001were plotted. B Relative abundances of the four species contributing to more than 60% in the similarity percentage (SIMPER) analysis.
Discussion
Paleoecological records of subtropical reef development in southeast Florida are crucial for evaluating the present and future biogeographical response of coral populations to climate change5,17. However, previous studies of Holocene reef development in the region have focused primarily on A. palmata23,37,38,39, thereby limiting our understanding of broader coral community dynamics. This study addresses this limitation by utilizing a well-preserved coral death assemblage located off the coast of northern Broward County to provide a comprehensive paleoecological reconstruction of coral assemblage composition during the Late Holocene in southeast Florida. Our results reveal significant differences in coral composition between Late Holocene and modern assemblages, suggesting that modern range expansions of tropical coral communities on the subtropical reefs off southeast Florida may be limited compared to those that occurred over millennial timescales. Below, we explore the potential implications of paleoenvironment and preservation on interpretations of relative coral abundance in the Late Holocene record, discuss the differences between Late Holocene and modern coral assemblages, and examine potential factors that may be contributing to assemblage differences.
Paleo-reef environment
The overall structure and preservation of the subfossil coral rubble deposits observed at Pompano Ridge are consistent with storm-generated deposition in which coral colonies growing on limestone ridges were repeatedly fragmented or dislodged by storm-induced waves and currents and transported shoreward to surrounding depositional lows (e.g., Refs. 40,41). Rapid storm burial with varying exposure to light is supported by the prevalence of broken and fragmented A. palmata and A. cervicornis clasts with varying thicknesses of sciaphilic-dominated encrusters42. Episodes of rapid burial in sediment and limited to no reworking are also supported by the presence of pristinely preserved coral clasts with limited to no encrustations or abrasion43. Overturned massive corals with well-preserved buried surfaces were also likely dislodged from their original growth position by large waves and transported shoreward, where they were partially buried by sand and rubble. Although input of allochthonous coral material from offshore sources (such as the inner reef ~400 m offshore) is possible, an absence of coral debris between the outer wave-cut-cliff terminus of the NRC44 and the rest of the reef tract suggests that the Late Holocene assemblages in the area most likely originated within the NRC. However, because we found no basal attachment surfaces, it is difficult to pinpoint the exact location where coral colonies existed within the study area45.
The composition of the Late Holocene assemblages also aligns with earlier descriptions of shallow-water coral reef assemblages from the tropical western Atlantic. Prior to the emergence of white-band disease in the late 1970s and since at least the late Pleistocene (~600,000 years ago), A. palmata was the overwhelmingly dominant species on shallow-water reef crests, often forming monospecific assemblage zones in depths less than 5 m2,46,47,48,49,50. Conversely, deeper forereef zones and protected backreefs and lagoons were typically dominated by more delicate A. cervicornis and massive colonies of Orbicella spp. We suggest a similar wave-energy-driven zonation pattern at Pompano Ridge during the Late Holocene, in which dense A. palmata thickets formed a shallow reef crest (<3 m paleodepths) at the eastern (seaward) extent of the study area, while more delicate branching corals and Orbicella spp. dominated a potentially slightly deeper and more protected rear zone (sensu46) at the western (shoreward) extent (Fig. 6). This is supported by the lower relative abundances of A. cervicornis and branching Porites spp. at eastern sites, where A. palmata was more abundant (Supplementary Table 7). Although the lower abundance of smaller, delicate coral taxa at the study area’s eastern extent is likely partly due to depositional grading, all but one of the in situ Orbicella spp. colonies encountered in the study area were present at the western extent, confirming the above zonation patterns.
Preservation bias and implications on coral composition
Unlike modern coral survey data, which represent ‘snapshots’ of living coral community composition at a singular point in time, the Late Holocene coral assemblages consist of a mixture of once temporally distinct coral assemblages time-averaged over multiple centuries. As a result, they are subject to considerable taphonomic bias as the living assemblage is converted to the subfossil assemblage over time35,36. This is especially the case on shallow-water, high-energy reefs where smaller branching and more delicate corals are typically underrepresented in death and fossil assemblages because they are easily destroyed or transported off the reef by storms51,52. However, in areas where the coral material is rapidly buried, and the death assemblage is quickly preserved with little transport away from the reef site or mixing from adjacent reef sites, high-energy reef facies have been shown to produce well-preserved coral clasts and have high fidelity to the life assemblage53,54,55,56. This latter scenario is likely the case at Pompano Ridge, where the taphonomy and spatial configuration of the Late Holocene assemblages indicate rapid or instantaneous entrapment and burial of autochthonous coral debris into depositional lows during storms.
Because the physiographic setting at Pompano Ridge likely supported high preservation of more delicate and fragile coral clasts, taphonomic bias due to differences in coral growth form must also be considered (reviewed in ref. 36). In environments where delicate branching coral fragments are retained, they are typically overrepresented in the death assemblage because they grow considerably faster than massive corals, have short regeneration/recovery times after storms, and are highly susceptible to storm fragmentation. This may explain the nearly equal proportions of A. palmata and A. cervicornis in the Late Holocene record despite the limited spatial extent of the semi-protected Orbicella-occupied rear zone and shallow depths (<4 m) across the entire reef. Because A. cervicornis is more delicate and has a higher branch density compared to A. palmata57, it is likely that its contribution to the Late Holocene record via fragmentation by storm waves was exaggerated relative to its live abundance on the reef. This is also likely the case with slower-growing but extremely delicate branching Porites spp., typically sparse on shallow, high-energy reef crests and shallow rear zones46,58. Although slower-growing and longer-lived massive corals are likely to be underestimated to some extent in the Late Holocene assemblages due to time averaging, lower abundances of those corals are generally expected on shallow-reef crests and rear zones dominated by faster-growing A. palmata and A. cervicornis46,59. This likely explains the conspicuous absence of both Mo. cavernosa and S. siderea in the Late Holocene assemblages.
Limits on modern coral range expansions
Despite potential limitations in assessing absolute coral composition from the geologic record, our results reveal a large fundamental difference between the Late Holocene assemblages at Pompano Ridge and the modern coral assemblages surveyed across the SFRT. Late Holocene assemblages consisted almost entirely of A. palmata and A. cervicornis, while Po. astreoides and S. siderea dominated the modern assemblages (Figs. 4b and 5B). Discordance between living and fossil assemblages can indicate substantial changes in coral ecosystem condition over time, particularly when it occurs between species with differing environmental requirements and life history traits32,60,61. This has been well-demonstrated in the tropical western Atlantic, where the loss of specialized reef-builders like Acropora and Orbicella spp. has also coincided with an emergence of novel communities dominated by more stress-tolerant and weedy coral taxa28,62. In recent years, Po. astreoides and S. siderea have been particularly successful on the subtropical reefs off southeast Florida, partly due to their resistance to thermal stress, sedimentation, and, more recently, stony coral tissue loss disease (SCTLD)63,64. Since the initial outbreak of SCTLD in 201464,65, the relative abundance of both species increased by nearly 50% on the shallow-water reefs between Dade and Martin counties (Supplementary Table 12), consistent with trends observed throughout the Florida reef tract66,67, Mexico68, and the Turks and Caicos69.
In addition to increases in the relative abundances of Po. astreoides and S. siderea, the contribution of A. cervicornis to the modern shallow-water coral assemblages on the SFRT rose dramatically between 2013 and 2021 (Fig. 5B, Supplementary Table 12). However, even with a nearly 400% increase in its relative abundance over that time period, it still only represented less than 1% of the coral assemblage in 2021. Likewise, at Pompano Ridge, A. cervicornis was the second most abundant living species surveyed in 2018, although it remained consistently subdominant to Po. astreoides (Fig. 4b). Expansions of large, monotypic thickets of A. cervicornis were first documented on the NRC off Broward County in the late 1990s but have since been relatively difficult to track because fragmentation events can move them in and out of long-term monitoring sites70. Interestingly, recent demographic surveys of multiple large A. cervicornis thickets in the same area indicated that disease and predation are rising, with reduced time between disturbances such as tropical storms potentially inhibiting natural recovery from those stressors71. The increasing dominance of stress-tolerant and weedy coral taxa like Po. astreoides and S. siderea, coupled with the recent challenges faced by A. cervicornis at the northern extent of its present geographic range, imply that the potential for persistent modern range expansions on the SFRT may be presently limited, particularly when compared to the Late Holocene.
Although long-term, gradual climate warming during the Holocene may have been the primary driver of coral range expansions in southeast Florida23,30,72, the effect of more recent changes in the intensity and frequency of thermal anomalies on these expansions remains uncertain. As climate warms, extreme temperature events are becoming more frequent, severe, and widespread, posing a substantial threat to subtropical reef communities existing near their thermal and environmental thresholds19. Yet, while acute deviations from regional long-term averages may be affecting the corals that are found alive on the SFRT73, between 2007 and 2021, only Miami-Dade County reefs experienced days with average temperatures above the upper thermal threshold for tropical Atlantic corals (>30.5 °C)74, and only Martin County reefs experienced days with average temperatures below the lower threshold (<16 °C)75 (Supplementary Tables 13 and 14; Supplementary Fig. 6)34. In contrast, both summer maximum and winter minimum sea-surface temperatures in Broward and Palm Beach Counties appear to be generally favorable for thermophilic species like A. palmata and A. cervicornis (>20 °C and <30 °C; Supplementary Fig. 4). This suggests that temperature stress is not presently inhibiting the expansion of tropical coral taxa to the subtropical reefs off southeast Florida. However, whether – or how long –thermal conditions off the coast of southeast Florida remain favorable is uncertain, especially given the rapid rate of warming in the region over the past few decades76.
Poleward range expansion of tropical coral species into higher latitude marine environments also strongly depends on successful larval dispersal from lower-latitude source populations13,19. In southeast Florida, coral larvae are transported from source populations in the Florida Keys via the northward-flowing Florida Current77. Despite the strong oceanographic connectivity between these subregions77, dramatic A. palmata and A. cervicornis population declines have resulted in lowered fertilization success (i.e., Allee Effect)78, further diminishing larval supply and recruitment across the Florida reef tract79,80,81. In contrast, Po. astreoides is genetically well-mixed throughout the region, indicating high levels of larval dispersal82,83 that may contribute to its recent success off southeast Florida. This scenario likely differs from the Late Holocene when acroporid coral populations on the FRKT were still likely maintaining most of their ecological functionality during the Late Holocene10,28. Better-developed source reefs with more expansive populations of Acropora spp. would have likely resulted in a higher regional larval supply84 and thus greater chances of successful recruitment on the subtropical reefs off southeast Florida during Holocene warm periods.
The ability of southeast Florida’s subtropical reefs to support expanding tropical coral populations is also likely limited by a variety of local-scale anthropogenic stressors associated with urbanization and coastal development. The modern reefs in this region are situated offshore of one of the most densely populated and urbanized coastlines in the continental United States and, as a result, are regularly exposed to land-based pollution sources, such as agricultural and urban runoff, sewage, and sedimentation65,85,86. Numerous studies have shown that nutrient enrichment in coral-reef ecosystems can not only reduce coral bleaching tolerances by increasing algal symbiont densities but also increase the severity of coral disease outbreaks87,88. Nutrients can also promote macroalgal growth, which in turn inhibits successful recruitment of reef-building corals89. Additionally, runoff of low salinity and high CO2 surface waters can cause localized coastal acidification and potentially lowered coral calcification on nearshore reefs near engineered inlets and tidal outflow currents86. Although moderate increases in turbidity can benefit corals by reducing thermal stress through shading90, excessive sedimentation can also lead to coral mortality or morbidity through sediment smothering and burial or by reducing light availability through increased turbidity91. While freshwater runoff and sedimentation were likely present to some extent during the Late Holocene, their effects on modern reef communities in the region are likely exacerbated by the alteration of southeast Florida’s natural watershed due to various human activities. In particular, increases in wastewater and stormwater discharges, associated with the rapid population growth of southeast Florida over the past century92, have likely played a substantial role. In addition, large-scale beach nourishment and coastal construction projects are likely impacting coral populations at local scales93,94. Together, these added anthropogenic stressors could potentially limit range expansions of tropical reef-building coral communities throughout the region.
Management implications
The persistence of Acropora-dominated coral communities off northern Broward County during Late Holocene warm periods underscores the potential for these ecosystems to serve as refugia for critically threatened coral species in the face of ongoing climate change. However, without natural recruitment from sexually reproductive source populations and mitigation of local anthropogenic impacts, the long-term viability of these refugia is uncertain. Strategic conservation efforts, such as assisted migration and targeted restoration programs, could reduce Allee effects and enhance natural recruitment throughout their full Holocene range84. Such efforts are likely to be most successful in Broward and Palm Beach Counties, where monitored sea-surface temperatures have been well within the optimal ranges for A. palmata, the taxon that offers the greatest potential for reef building and coastal protection from storms95. On the other hand, it is important to note that northern Broward County marks a distinct physiographic boundary in which nearshore hardbottom habitats in the optimal depth range for A. palmata become sparse96,97. Cold-water upwelling also becomes more prevalent at Palm Beach County’s northern extent, where the Gulf Stream current begins to veer offshore98. As a result, coastal communities in Palm Beach County would have to invest not only in coral restoration and rehabilitation programs, but also in the implementation of elevated artificial reef structures to provide the foundation required to successfully reintroduce shallow-water Acropora-dominated coral communities to the area.
While our study suggests that modern climate warming may be enhancing the capacity of Florida’s subtropical reefs to support range expansions of thermophilic reef-building species reminiscent of the Late Holocene, it is critical to acknowledge that these expansions are occurring within an ecosystem in which environmental conditions and ecology have substantially deviated from long-term Holocene baselines. Therefore, the restoration of a more natural reef state in this anthropogenically altered system will undoubtedly require substantial human intervention. Future restoration and artificial reef programs will likely be most successful if they emulate not only the species composition of the Late Holocene reef assemblages at Pompano Ridge, but also the historical paleodepths and zonation patterns. A strategic approach focusing on establishing resilient Acropora-dominated coral communities similar to their Late Holocene counterparts could potentially facilitate the most effective restoration and establishment of subtropical refugia. However, it is important to emphasize that the long-term sustainability of these restoration activities will ultimately depend on whether modern climate warming can be slowed or reversed.
Methods
Regional Setting and study area
The southeast Florida reef tract (SFRT) is an antecedent reef system spanning ~150 km of Florida’s Atlantic coastline from Biscayne National Park in south Miami (25.2°N) to the St. Lucie inlet in southern Martin County (27.3°N). It is located at the boundary of the subtropical and temperate climate zones99, and, as a result, it is periodically exposed to low air temperatures after the passage of mid-latitude cold fronts during the winter100. Although the intensity of winter cold events on southeast Florida’s reefs is muted by the warm waters of the Florida Current100,101,102, it has likely played a prominent role in limiting the distribution of reef-building corals throughout the reef tract since the end of the Holocene23,27,83. Over the past century, coral-reef communities on the SFRT have been dominated by eurytopic, non-reef-building corals as well as macroalgae, soft corals, and sponges103,104; however, recent expansions of A. palmata and A. cervicornis where they were previously rare (A. cervicornis) or absent (A. palmata) suggest that warming climate conditions may be tropicalizing the region’s subtropical reefs17. Because these trends have only been observed over the past several decades, however, it is unclear whether those populations will be able to persist for the long term under future climate change and other environmental disturbances23.
Geological investigations of the underlying structure of the SFRT indicate that past range expansions of tropical reef-building corals are not unprecedented and that Acropora-dominated reef communities persisted ~120 km north of their recent geographical distribution during the Holocene thermal maximum (HTM) (~8000–5000 years ago). Warmer, more stable climate conditions during this period likely permitted a more northerly distribution of cold-sensitive acroporids as far north as Palm Beach County (26.7°N)17,23,37. This pattern persisted until ~7000 years ago when deteriorating climate conditions initiated a stepwise southward contraction of Acropora spp. By ~5000 years B.P., the offshore reefs had contracted to modern-day Miami-Dade County, and by ~3000 years B.P., reef development had largely ceased regionwide23. Today, the coral-reef assemblages off southeast Florida are situated offshore one of the most densely populated and urbanized coastlines in the continental United States and, as a result, are exposed to a multitude of local-scale anthropogenic stressors (nutrient pollution, overfishing, etc.) not present during the Holocene27,65.
Although prior studies of the mid-Holocene reefs off southeast Florida suggest that primary A. palmata-dominated reef development in the region contracted south of modern-day Broward County by ~5000 years B.P.23,39, recent investigations of newly discovered subfossil coral deposits off northern Broward County have revealed that both A. palmata and Orbicella spp. returned to the region again between ~3500 and 1800 years B.P30. Although there are currently no local sea-surface temperature reconstructions for the area, the timing of this subsequent range expansion corresponds to regional records of sea-surface temperature warming at the northern extent of the AWP105. Based on this overlap and modern oceanographic observations in the area, Modys et al.30 hypothesized that centennial-scale expansions of the AWP may have enhanced the general buffering effect that the Florida Current has on coastal sea-surface temperatures off southeast Florida after the passage of winter cold-fronts. The Late Holocene record off northern Broward County thus provides valuable insights into the long-term ecological dynamics of the region’s high-latitude reefs in response to climate change over finer time scales not attainable from existing mid-Holocene fossil-reef records.
The Late Holocene subfossil coral assemblages examined in this study are located at the northern extent of the nearshore ridge complex (NRC) ~ 600 m off the coast of northern Broward County in southeast Florida (26°13’N, 80°5’W) (Fig. 1). The NRC, which is the shallowest geomorphological subunit of the SFRT, is characterized by a series of shore-parallel Pleistocene coquina ridges ranging in water depth from 2–6 m below mean sea level (MSL)38. The subfossil coral assemblages are scattered across the surface of these ridges, forming veneers (<1.5 m) of poorly consolidated to unconsolidated coral rubble interspersed with partially buried in situ massive coral colonies30,31. Whereas the outer edge of the coral rubble deposits terminates abruptly ~300 m from the offshore terminus of the NRC, the inshore edge forms a gradual, shoreward-sloping talus that grades into fine rubble and then mixed sand and hardbottom. The estimated spatial coverage of the NRC rubble deposits at Pompano Ridge is ~2.5 km3; however, exposure may vary over time with sediment movement.
Quantifying Late Holocene coral composition
To determine the taxonomic composition of the Late Holocene coral assemblages, we conducted surveys along 16 shore-parallel, line-intercept transects (60-m long) distributed haphazardly across the full extent of the coral rubble zone as determined by SCUBA-based ground-truthing and examination of high-resolution bathymetric data (Fig. 1b, Supplementary Table 1). Surficial coral rubble and dead massive colonies encountered every 5 cm were identified to the lowest taxonomic level recognizable without physically breaking them apart or removing algal and other encrusting overgrowths. Additionally, sub-surface coral rubble was collected from a total of 16 50 cm diameter pits excavated at a random point along each transect. Coral fragments were removed from each pit until a 2-liter mesh bag was filled (pit depths between 30 and 60 cm). Coral rubble from pit samples was also identified to the lowest taxonomic level and then pooled with data obtained from the line-point intercept transects. Because the physical sizes and taphonomic characteristics of surface and sub-surface coral skeletons varied, collection and pooling of data from pit and transect samples was done to ensure that the complete taxonomic composition of the subfossil coral assemblages were incorporated into the comparative analyses. Although coral clast size varied between transect and pit collections, the number of identifiable corals was relatively consistent (60‒100 corals each). In total, 1949 coral skeletons from 16 pit/transect samples were identified (Supplementary Table 2).
Previous studies have shown that understanding the taphonomy and depositional dynamics of time-averaged fossil marine deposits is critical to accurately reconstructing their community composition106. This is especially true with fossil coral-reef deposits where varying depth, energy regime, and coral growth form can have a substantial impact on what is preserved, destroyed, or exported off the reef52,53,107. To address this, we also qualitatively evaluated representative subfossil coral samples collected from pit samples and transects based on their overall preservation in terms of abrasion, presence of original corallite material, and encruster communities [following43,108 and references therein]. These parameters were used as the basis for evaluating paleoenvironmental and depositional setting as well as the potential effects of taphonomic bias in the Late Holocene record due to time-averaging.
U-Th dating
Previous high-precision U-Th dating of 40 subfossil A. palmata and Orbicella spp. samples collected from the same coral death assemblages surveyed in this study yielded mixed ages between ~900 and 4500 yr B.P.30. Given that both taxa are locally rare or absent on modern reefs throughout the region, it is likely that the subfossil A. palmata and Orbicella spp. corals sampled in the present study are also Late Holocene-aged. However, several other taxa encountered in the coral death assemblages are also found living on nearshore reef habitats throughout the SFRT. To confirm that these additional taxa were indeed Late Holocene-aged and not recently incorporated into the subfossil coral death assemblages, we dated an additional ten samples of A. cervicornis (n = 4), Ps. strigosa (n = 5), and C. natans (n = 1) using the same high-precision U-Th dating methods. We also incorporated ten previously published ages obtained from subfossil D. cylindrus (n = 8) and A. prolifera (n = 2) samples collected from the same study area31,33. Smaller and more delicate coral species also found in both the living and subfossil coral death assemblages, including Porites and Agaricia, were not dated because they were typically contaminated with secondary cements (high-Mg calcite and iron oxide), making it difficult to extract unaltered coral aragonite required for U-Th dating.
Prior to U-Th dating, each subfossil coral sample was sectioned laterally using a diamond saw, and small pieces of internal coral aragonite (~1 g) free of visually observable secondary alteration and dissolution were extracted along the primary growth axis. The coral pieces were soaked in an 8% sodium hypochlorite solution for ~24 h and then sonicated in deionized water to remove any remaining organic or detrital material. Because diagenesis is a considerable source of error in determining U-Th ages from corals, additional sub-samples were powdered and analyzed for secondary calcite by X-ray diffraction (XRD) using a Philips XPert Powder Diffractometer. Only samples with “no detectable” calcite (less than 0.2 wt. %) were processed further for U-Th dating. Although we did not evaluate the additional samples dated for this study with a scanning electron microscope (SEM), previous SEM analysis of representative samples collected from the same study area yielded little to no secondary aragonite30. U-Th isotopic measurements were determined by Multi-collector Inductively Coupled Plasma Mass Spectrometry (MC-ICP-MS) at Rutgers University following the methods described in ref. 109 and revised in ref. 110.
Spatial variability
Spatial variability in Late Holocene coral assemblage composition within the study area at Pompano Ridge was evaluated at the transect level (pooled data from line-point intercept transects and pits) using hierarchical average linkage cluster analysis based on Bray–Curtis similarities of square-root transformed abundance data. The cluster analysis and calculation of Bray–Curtis dissimilarities was carried out using the ‘hclust’ and ‘metaMDS’ functions in the R package vegan111,112. Differences in coral community composition among spatially varying transect groups were then evaluated by ordinating the Bray–Curtis dissimilarities using non-metric multidimensional scaling (NMDS). Groups were also compared by conducting an analysis of similarities (ANOSIM) in vegan (“ANOSIM” function). Once differences in dissimilarities among transects were confirmed, we further investigated the specific changes in coral community composition that contributed to spatial variability within the study area by calculating similarity percentages using the ‘simper’ function in vegan. We also evaluated contributions of individual species to dissimilarities between groups by fitting species correlation vectors to the ordination (‘envfit’ in vegan with 999 permutations). Only species that cumulatively contributed 60‒70% to average dissimilarity in SIMPER analysis were plotted.
Comparison of modern and Late Holocene assemblages
To determine how contemporary assemblages at our study site compare with the Late Holocene assemblages within the study area at Pompano Ridge, we collected live coral abundance data along the same 16 transects in the summer and fall of 2018. For each transect, all living corals larger than 4 cm within a 1-m wide belt were identified to the species level and counted by SCUBA divers. To determine how modern reef assemblages throughout the SFRT compare with the Late Holocene assemblages surveyed in this study, we also evaluated stony coral demographic data from the Florida Fish and Wildlife Conservation Commission’s (FWC) Southeast Florida Coral Reef Evaluation and Monitoring Program (SECREMP) (cf. Toth et al.10). The SECREMP monitoring program has conducted annual surveys on coral reef and hardbottom sites throughout the SFRT since 200334. At each site, the percent cover of stony corals and other benthos is estimated using a random-point-count method on images taken along four permanent 22 × 0.4 m belt transects. We selected only SECREMP sites in depths less than 8 m below mean sea level (bMSL) to best match the shallow, high-energy reef environment present at the study site during the Late Holocene30. This included two inner reef and five NRC sites between Miami-Dade and Martin counties (Fig. 1a; Supplementary Table 3). The closest SECREMP site to the study area is ~3 km south on the NRC off Fort Lauderdale, Florida.
Although 28 unique scleractinian coral taxa were identified in the modern and Late Holocene coral assemblages, we only compared 18 taxa that can contribute substantially to reef accumulation (Supplementary Tables 4 and 5). This excluded rare, free-living, and relatively small genera such as Manicina, Favia, Isophyllia, Stephanocoenia, and Eusmilia, which likely have variable preservation in the subfossil record. The hydrocorals Mi. complanata and Mi. alcicornis were also excluded because they were not included in the SECREMP count surveys. Reef-building coral taxa that were present in the modern assemblages but absent in the Late Holocene assemblages were only included if they could be perceived as having an equal chance of being preserved as other present taxa without obvious taphonomic or sampling biases. For example, although Mo. cavernosa was absent in the Late Holocene assemblages, other massive corals like Ps. strigosa and C. natans were present. Given that both species share similar physical properties (average size and growth form) and occupy similar habitats on the modern reef tract63,104, we assume that the absence of Mo. cavernosa is due to actual absence, rather than systematic sampling bias or taphonomic bias. The other species that were absent in the Late Holocene assemblages but included in this study based on the likelihood of preservation were Solenastrea bournoni, Madracis decactis, Meandrina meandrites, and S. siderea.
Prior to comparative analysis, relative abundance was calculated for both the modern and Late Holocene assemblages based on the count data for the 18 taxa selected for this study. Non-metric multidimensional scaling (NMDS) ordination and ANOSIM were used to examine overall differences in composition between the Late Holocene and modern assemblages within Pompano Ridge and across SECREMP sites located throughout the SFRT, while similarity percentages (simper) and species vectors (envfit) were used to identify and visualize specific taxa that contributed to those measured dissimilarities (as described above). This approach was based off of several previous studies that also compared modern reef assemblages to fossil assemblages10,29,52,53,107.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All Late Holocene coral composition and age data generated or analyzed during this study are published in USGS data releases, accessible via the following links: https://doi.org/10.5066/P9VTNG2N and https://doi.org/10.5066/P9F0VW9L. All original coral composition and reef temperature data from the Southeast Coral Reef Evaluation and Monitoring Project (SECREMP) were obtained from https://geodata.myfwc.com, and the specific datasets used in this study are available for download at https://doi.org/10.6084/m9.figshare.c.7060112. Additionally, a subset of the coral composition data utilized for each analysis in the above study is downloadable at https://doi.org/10.6084/m9.figshare.c.7060112. Coral specimens collected and analyzed for this research are stored in the Florida Atlantic University Department of Geosciences sample repository, ensuring their preservation and availability for future studies.
References
Grafeld, S., Oleson, K. L. L., Teneva, L. & Kittinger, J. N. Follow that fish: Uncovering the hidden blue economy in coral reef fisheries. PloS One 12, e0182104 (2017).
Aronson, R. B., & Precht, W. F. 9. Evolutionary Paleoecology of Caribbean Coral Reefs. In Evolutionary paleoecology, 171–234 (Columbia University Press, 2001).
Sully, S., Burkepile, D. E., Donovan, M. K., Hodgson, G. & Van Woesik, R. A global analysis of coral bleaching over the past two decades. Nat. Commun. 10, 1264 (2019).
Aronson, R. B., & Precht, W. F. White-band disease and the changing face of Caribbean coral reefs. In The Ecology and Etiology of Newly Emerging Marine Diseases, 25–38, https://doi.org/10.1023/A:1013103928980 (2001).
Kuffner, I. B. & Toth, L. T. A geological perspective on the degradation and conservation of western Atlantic coral reefs. Conserv. Biol. 30, 706–715 (2016).
Bruno, J. F., Côté, I. M. & Toth, L. T. Climate change, coral loss, and the curious case of the parrotfish paradigm: Why don’t marine protected areas improve reef resilience? Ann. Rev. Marine Sci. 11, 307–334 (2019).
Pandolfi, J. M. & Jackson, J. B. C. Ecological persistence interrupted in Caribbean coral reefs. Ecol. Lett. 9, 818–826 (2006).
Gardner, T. A., Côté, I. M., Gill, J. A., Grant, A. & Watkinson, A. R. Long-term region-wide declines in Caribbean corals. Science 301, 958–960 (2003).
Edmunds, P. J. A quarter‐century demographic analysis of the Caribbean coral, Orbicella annularis, and projections of population size over the next century. Limnol. Oceanogr. 60, 840–855 (2015).
Toth, L. T. et al. The unprecedented loss of Florida’s reef‐building corals and the emergence of a novel coral‐reef assemblage. Ecology 100, e02781 (2019).
NMFS-NOAA. Endangered and threatened species: final listing determinations for elkhorn coral and staghorn coral. Federal Register 71, 26852−26861 (2006).
NMFS-NOAA. Final listing determinations on proposal to list 66 reef-building coral species and to reclassify elkhorn and staghorn corals, final rule 79, Federal Register 53852–54123, https://www.federalregister.gov/documents/2014/09/10/2014-20814/endangered-and-threatened-wildlife-andplants-final-listing-determinations-on-proposal-to-list-66 (2014).
Yamano, H., Sugihara, K., & Nomura, K. Rapid poleward range expansion of tropical reef corals in response to rising sea surface temperatures. Geophys. Res. Lett., 38, https://doi.org/10.1029/2010GL046474 (2011).
Serrano, E. et al. Rapid northward spread of a zooxanthellate coral enhanced by artificial structures and sea warming in the western Mediterranean. PLoS One 8, e52739 (2013).
Greenstein, B. J. & Pandolfi, J. M. Escaping the heat: range shifts of reef coral taxa in coastal Western Australia. Global Change Biol. 14, 513–528 (2008).
Vargas-Ángel, B., Thomas, J. D. & Hoke, S. M. Higher latitude Acropora cervicornis thickets off Fort Lauderdale, Florida, USA. Coral Reefs 22, 465–473 (2003).
Precht, W. F. & Aronson, R. B. Climate flickers and range shifts of reef corals. Front. Ecol. Environ. 2, 307–314 (2004).
Precht, W. F. et al. Back to the future: the history of acroporid corals at the Flower Garden Banks, Gulf of Mexico, USA. Marine Geol. 349, 152–161 (2014).
Beger, M., Sommer, B., Harrison, P. L., Smith, S. D. A. & Pandolfi, J. M. Conserving potential coral reef refuges at high latitudes. Divers. Distributions 20, 245–257 (2014).
Schoepf, V., et al. Corals at the edge of environmental limits: A new conceptual framework to re-define marginal and extreme coral communities. Sci. Total Environ. 163688 https://doi.org/10.1016/j.scitotenv.2023.163688 (2023).
Makino, A. et al. Spatio‐temporal marine conservation planning to support high‐latitude coral range expansion under climate change. Divers. Distributions 20, 859–871 (2014).
Kleypas, J. A., McManus, J. W. & Meñez, L. A. B. Environmental limits to coral reef development: where do we draw the line? Am. Zoologist 39, 146–159 (1999).
Toth, L. T. et al. Climate and the latitudinal limits of subtropical reef development. Sci. Rep. 11, 13044 (2021).
Fabricius, K. E. Factors determining the resilience of coral reefs to eutrophication: a review and conceptual model. Coral Reefs, 493–505, https://doi.org/10.1007/978-94-007-0114-4_28 (2011).
Denis, V. et al. Coverage, diversity, and functionality of a higher latitude coral community (Tatsukushi, Shikoku Island, Japan). PLoS One 8, e54330 (2013).
Stott, P. A. et al. Attribution of extreme weather and climate‐related events. Wiley Interdiscip. Rev. Clim. Change 7, 23–41 (2016).
Precht, W. F., & Miller, S. L. Ecological shifts along the Florida reef tract: the past as a key to the future. Geological Approaches Coral Reef Ecol. 237–312, https://doi.org/10.1007/978-0-387-33537-7_9 (2007).
Toth, L. T., Kuffner, I. B., Stathakopoulos, A. & Shinn, E. A. A 3,000‐year lag between the geological and ecological shutdown of Florida’s coral reefs. Global Change Biol. 24, 5471–5483 (2018).
O’Dea, A. et al. Defining variation in pre-human ecosystems can guide conservation: An example from a Caribbean coral reef. Sci. Rep. 10, 2922 (2020).
Modys, A. B., Olenik, A. E., Mortlock, R. A., Toth, L. T. & Precht, W. F. Climate-modulated range expansion of reef-building coral communities off southeast Florida during the Late Holocene. Front. Marine Sci. 9, 2390 (2022).
Modys, A.B., Toth, L.T., Mortlock, R.A., Oleinik, A.E. and Precht, W.F. Discovery of a rare pillar coral (Dendrogyra cylindrus) death assemblage off southeast Florida reveals multi-century persistence during the late Holocene. Coral Reefs, 1–7, https://doi.org/10.1007/s00338-023-02387-3 (2023).
Jackson, J. B. C. Reefs since Columbus. Coral Reefs 16, S23–S32 (1997).
Modys, A. B., Mortlock, R. A., Vollmer, S. V., Kaufman, L. & Precht, W. F. U-Th dating of Holocene age Acropora prolifera (Lamarck, 1816) colonies confirms coral hybridization is not a recent phenomenon. Proc. Biol. Soc. Washington 133, 72–75 (2020).
Gilliam D. S., Hayes N. K., Ruzicka R. R., Colella M. A. Southeast Florida coral reef evaluation and monitoring project 2020 year 18 final report, 82 (Florida DEP: FWC, 2021).
Greenstein, B. J. & Pandolfi, J. M. Taphonomic alteration of reef corals: effects of reef environment and coral growth form II: the Florida Keys. Palaios 18, 495–509 (2003).
Greenstein, B. J. Taphonomy: detecting critical events in fossil reef-coral assemblages. Geological Approaches Coral Reef Ecol. 192, 31–60 (2007).
Lighty, R. G., Macintyre, I. G. & Stuckenrath, R. Submerged early Holocene barrier reef south-east Florida shelf. Nature 276, 59–60 (1978).
Banks, K. W., Riegl, B. M., Shinn, E. A., Piller, W. E. & Dodge, R. E. Geomorphology of the southeast Florida continental reef tract (Miami-Dade, Broward, and Palm Beach counties, USA). Coral Reefs 26, 617–633 (2007).
Stathakopoulos, A. & Riegl, B. M. Accretion history of mid-Holocene coral reefs from the southeast Florida continental reef tract, USA. Coral Reefs 34, 173–187 (2014).
Scoffin, T. P. & Hendry, M. D. Shallow-water sclerosponges on Jamaican reefs and a criterion for recognition of hurricane deposits. Nature 307, 728–729 (1984).
Blanchon, P. et al. Retrograde accretion of a Caribbean fringing reef controlled by hurricanes and sea-level rise. Front. Earth Sci. 5, 78 (2017).
Perry, C. T. & Hepburn, L. J. Syn-depositional alteration of coral reef framework through bioerosion, encrustation and cementation: taphonomic signatures of reef accretion and reef depositional events. Earth Sci. Rev. 86, 106–144 (2008).
Perry, C. Storm-induced coral rubble deposition: Pleistocene records of natural reef disturbance and community response. Coral Reefs 20, 171–183 (2001).
Raymond, W.F. A geologic investigation of the offshore sands and reefs of Broward County, Florida. Doctoral dissertation (Florida State University, 1972).
Stathakopoulos, A., Riegl, B. M. & Toth, L. T. A revised Holocene coral sea-level database from the Florida reef tract, USA. PeerJ 8, e8350 (2020).
Goreau, T. F. The ecology of Jamaican coral reefs I. Species composition and zonation. Ecology 40, 67–90 (1959).
Mesolella, K. J. Zonation of uplifted Pleistocene coral reefs on Barbados, West Indies. Science 156, 638–640 (1967).
Adey, W. H. & Burke, R. Holocene bioherms (algal ridges and bank-barrier reefs) of the eastern Caribbean. Geol. Soc. Am. Bull. 87, 95–109 (1976).
Gladfelter, E. H., Monahan, R. K. & Gladfelter, W. B. Growth rates of five reef-building corals in the northeastern Caribbean. Bull. Marine Sci. 28, 728–734 (1978).
Jackson, J. B. C. Pleistocene perspectives on coral reef community structure. Am. Zoologist 32, 719–731 (1992).
Hubbard, D. K., Miller, A. I. & Scaturo, D. Production and cycling of calcium carbonate in a shelf-edge reef system (St. Croix, US Virgin Islands); applications to the nature of reef systems in the fossil record. J. Sediment. Res. 60, 335–360 (1990).
Edinger, E. N., Pandolfi, J. M. & Kelley, R. A. Community structure of Quaternary coral reefs compared with Recent life and death assemblages. Paleobiology 27, 669–694 (2001).
Pandolfi, J. M. & Minchin, P. R. A comparison of taxonomic composition and diversity between reef coral life and death assemblages in Madang Lagoon, Papua New Guinea. Palaeogeogr. Palaeoclimatol. Palaeoecol. 119, 321–341 (1996).
Greenstein, B. J., & Moffat, H. A. Comparative taphonomy of modern and Pleistocene corals, San Salvador, Bahamas. Palaios, 57–63, https://doi.org/10.2307/3515116 (1996).
Bishop, D., & Greenstein, B. J. The effects of Hurricane Floyd on the fidelity of coral life and death assemblages in San Salvador, Bahamas: Does a hurricane leave a signature in the fossil record. In Geological Society of America, Abstracts with Programs, North-Central Regional Meeting, 33, A51 (Geological Society of America, 2001).
Riegl, B. Inhibition of reef framework by frequent disturbance: examples from the Arabian Gulf, South Africa, and the Cayman Islands. Palaeogeogr. Palaeoclimatol. Palaeoecol. 175, 79–101 (2001).
Highsmith, R. C. Geographic patterns of coral bioerosion: a productivity hypothesis. J. Exp. Marine Biol. Ecol. 46, 177–196 (1980).
Roos, P. J. The shallow-water stony corals of the Netherlands Antilles. Stud. Fauna Curaçao Other Caribbean Islands 37, 1–108 (1971).
Geister, J. The influence of wave exposure on the ecological zonation of Caribbean coral reefs. Proc. 3rd Coral Reef Symp. 1, 23–29 (1977).
Aronson, R. B., Precht, W. F. & Macintyre, I. G. Extrinsic control of species replacement on a Holocene reef in Belize: the role of coral disease. Coral Reefs 17, 223–230 (1998).
Hammerman, N. M., Roff, G., Lybolt, T., Eyal, G. & Pandolfi, J. M. Unraveling Moreton Bay reef history: An urban higher latitude setting for coral development. Front. Ecol. Evol. 10, 884850 (2022).
Green, D. H., Edmunds, P. J. & Carpenter, R. C. Increasing relative abundance of Porites astreoides on Caribbean reefs mediated by an overall decline in coral cover. Marine Ecol. Progr. Ser. 359, 1–10 (2008).
Burman, S. G., Aronson, R. B. & van Woesik, R. Biotic homogenization of coral assemblages along the Florida reef tract. Marine Ecol. Progr. Ser. 467, 89–96 (2012).
Precht, W. F., Gintert, B. E., Robbart, M. L., Fura, R. & Van Woesik, R. Unprecedented disease-related coral mortality in Southeastern Florida. Sci. Rep. 6, 1–11 (2016).
Enochs, I. C. et al. Coral persistence despite marginal conditions in the Port of Miami. Sci. Rep. 13, 6759 (2023).
Walton, C. J., Hayes, N. K. & Gilliam, D. S. Impacts of a regional, multi-year, multi-species coral disease outbreak in Southeast Florida. Front. Marine Sci. 5, 323 (2018).
Hayes, N. K., Walton, C. J. & Gilliam, D. S. Tissue loss disease outbreak significantly alters the Southeast Florida stony coral assemblage. Front. Marine Sci. 9, 975894 (2022).
Alvarez-Filip, L., Estrada-Saldívar, N., Pérez-Cervantes, E., Molina-Hernández, A. & González-Barrios, F. J. A rapid spread of the stony coral tissue loss disease outbreak in the Mexican Caribbean. PeerJ 7, e8069 (2019).
Heres, M. M., Farmer, B. H., Elmer, F. & Hertler, H. Ecological consequences of stony coral tissue loss disease in the Turks and Caicos Islands. Coral Reefs 40, 609–624 (2021).
Walker, B. K., Larson, E. A., Moulding, A. L. & Gilliam, D. S. Small-scale mapping of indeterminate arborescent acroporid coral (Acropora cervicornis) patches. Coral Reefs 31, 885–894 (2012).
Goergen, E. A., Moulding, A. L., Walker, B. K. & Gilliam, D. S. Identifying causes of temporal changes in Acropora cervicornis populations and the potential for recovery. Front. Marine Sci. 6, 36 (2019).
Bova, S., Rosenthal, Y., Liu, Z., Godad, S. P. & Yan, M. Seasonal origin of the thermal maxima at the Holocene and the last interglacial. Nature 589, 548–553 (2021).
Jones, N. P., Figueiredo, J. & Gilliam, D. S. Thermal stress-related spatiotemporal variations in higher latitude coral reef benthic communities. Coral Reefs 39, 1661–1673 (2020).
Manzello, D. P., Berkelmans, R. & Hendee, J. C. Coral bleaching indices and thresholds for the Florida reef tract, Bahamas, and St. Croix, US Virgin Islands. Marine Pollut. Bull. 54, 1923–1931 (2007).
Burns, T. P. Hard-coral distribution and cold-water disturbances in South Florida: variation with depth and location. Coral Reefs 4, 117–124 (1985).
Bove, C. B., Mudge, L. & Bruno, J. F. A century of warming on Caribbean reefs. PloS Clim. 1, e0000002 (2022).
Frys, C. et al. Fine-scale coral connectivity pathways in the Florida Reef Tract: implications for conservation and restoration. Front. Marine Sci. 7, 312 (2020).
Knowlton, N. The future of coral reefs. Proc. Natl Acad. Sci. 98, 5419–5425 (2001).
Baums, I. B., Miller, M. W. & Hellberg, M. E. Regionally isolated populations of an imperiled Caribbean coral, Acropora palmata. Mol. Ecol. 14, 1377–1390 (2005).
Williams, D. E., Miller, M. W. & Kramer, K. L. Recruitment failure in Florida Keys Acropora palmata, a threatened Caribbean coral. Coral Reefs 27, 697–705 (2008).
van Woesik, R., Scott, W. J. IV & Aronson, R. B. Lost opportunities: coral recruitment does not translate to reef recovery in the Florida Keys. Marine Pollut. Bull. 88, 110–117 (2014).
Gallery, D. N., Green, M. L., Kuffner, I. B., Lenz, E. A. & Toth, L. T. Genetic structure and diversity of the mustard hill coral Porites astreoides along the Florida Keys reef tract. Marine Biodivers. 51, 1–16 (2021).
Shilling, E.N., Eckert, R.J., Sturm, A.B. and Voss, J.D., Porites astreoides coral populations demonstrate high clonality and connectivity in southeast Florida. Coral Reefs 1–15, https://doi.org/10.1007/s00338-023-02417-0 (2023).
Kuffner, I. B., Stathakopoulos, A., Toth, L. T. & Bartlett, L. A. Reestablishing a stepping-stone population of the threatened elkhorn coral Acropora palmata to aid regional recovery. Endanger. Species Res. 43, 461–473 (2020).
Futch, J. C., Griffin, D. W., Banks, K. & Lipp, E. K. Evaluation of sewage source and fate on southeast Florida coastal reefs. Marine Pollut. Bull. 62, 2308–2316 (2011).
Enochs, I. C., Manzello, D. P., Jones, P. R., Stamates, S. J. & Carsey, T. P. Seasonal carbonate chemistry dynamics on southeast Florida coral reefs: localized acidification hotspots from navigational inlets. Front. Marine Sci. 6, 160 (2019).
Bruno, J. F., Petes, L. E., Drew Harvell, C. & Hettinger, A. Nutrient enrichment can increase the severity of coral diseases. Ecol. Lett. 6, 1056–1061 (2003).
Burkepile, D. E. et al. Nitrogen identity drives differential impacts of nutrients on coral bleaching and mortality. Ecosystems 23, 798–811 (2020).
Lirman, D. Competition between macroalgae and corals: effects of herbivore exclusion and increased algal biomass on coral survivorship and growth. Coral Reefs 19, 392–399 (2001).
Cacciapaglia, C. & van Woesik, R. Climate‐change refugia: Shading reef corals by turbidity. Global Change Biol. 22, 1145–1154 (2015).
Jones, R., Fisher, R. & Bessell-Browne, P. Sediment deposition and coral smothering. PLoS One 14, e0216248 (2019).
Huq, E. & Abdul-Aziz, O. I. Climate and land cover change impacts on stormwater runoff in large-scale coastal-urban environments. Sci. Total Environ. 778, 146017 (2021).
Jordan, L. K. B., Banks, K. W., Fisher, L. E., Walker, B. K. & Gilliam, D. S. Elevated sedimentation on coral reefs adjacent to a beach nourishment project. Marine Pollut. Bull. 60, 261–271 (2010).
Gintert, B. E. et al. Regional coral disease outbreak overwhelms impacts from a local dredge project. Environ. Monit. Assess. 191, 630 (2019).
Toth, L. T. et al. The potential for coral reef restoration to mitigate coastal flooding as sea levels rise. Nat. Commun. 14, 2313 (2023).
Walker, B. K. Spatial analyses of benthic habitats to define coral reef ecosystem regions and potential biogeographic boundaries along a latitudinal gradient. PloS One 7, e30466 (2012).
Wirt, K. E., Hallock, P., Palandro, D. & Daly, K. L. Potential habitat of Acropora spp. on Florida reefs. Appl. Geogr. 39, 118–127 (2013).
Walker, B. K. & Gilliam, D. S. Determining the extent and characterizing coral reef habitats of the northern latitudes of the Florida Reef Tract (Martin County). PloS One 8, e80439 (2013).
Chen, E. & Gerber, J. F. Climate. Ecosystems of Florida. Univ. Central Florida Press 11, 35 (1990).
Banks, K. W., et al The reef tract of continental southeast Florida (Miami-Dade, Broward and Palm Beach counties, USA). Coral Reefs USA, 175–220, https://doi.org/10.1007/978-1-4020-6847-8_5 (2008).
Lee, T. N. Florida Current spin-off eddies. Deep Sea Res. Oceanogr. Abstr. 22, 753–765 (1975).
Jaap W. The ecology of the south Florida coral reefs: a community profile. Florida Department of Natural Resources. Technical Report FWS/OBS-82/08, https://www.osti.gov/biblio/5486458 (1984).
Vaughan, T. W. Investigations of the geology and geologic processes of the reef tracts and adjacent areas of the Bahamas and Florida, 12, 1–183 (Carnegie Institute Washington Yearbook, 1914).
Goldberg, W. M. The ecology of the coral-octocoral communities off the southeast Florida coast: geomorphology, species composition, and zonation. Bull. Marine Sci. 23, 465–488 (1973).
Thirumalai, K., Richey, J. N. & Quinn, T. M. Holocene Evolution of Sea‐Surface Temperature and Salinity in the Gulf of Mexico. Paleoceanogr. Paleoclimatol. 36, e2021PA004221 (2021).
Kidwell, S. M. Time‐averaging and fidelity of modern death assemblages: building a taphonomic foundation for conservation palaeobiology. Palaeontology 56, 487–522 (2013).
Greenstein, B. J. & Pandolfi, J. M. Preservation of community structure in modern reef coral life and death assemblages of the Florida Keys: implications for the Quaternary fossil record of coral reefs. Bull. Marine Sci. 61, 431–452 (1997).
Martindale, W. Calcified epibionts as palaeoecological tools: examples from the Recent and Pleistocene reefs of Barbados. Coral Reefs 11, 167–177 (1992).
Mortlock, R. A., Fairbanks, R. G., Chiu, T. & Rubenstone, J. 230Th/234U/238U and 231Pa/235U ages from a single fossil coral fragment by multi-collector magnetic-sector inductively coupled plasma mass spectrometry. Geochim. Cosmochimica Acta 69, 649–657 (2005).
Abdul, N. A., Mortlock, R. A., Wright, J. D. & Fairbanks, R. G. Younger Dryas sea level and meltwater pulse 1B recorded in Barbados reef crest coral Acropora palmata. Paleoceanography 31, 330–344 (2016).
RStudio Team. RStudio: Integrated Development Environment for R (version 1.4.1106). RStudio, PBC, Available at: http://www.rstudio.com/ (2021).
Oksanen, J. et al. Package ‘vegan’. Community Ecology Package, Version 2, 1–295 https://github.com/vegandevs/vegan (2013).
Acknowledgements
We would like to thank Scott Hurwood for his assistance in the field. Fieldwork was conducted under a Florida Fish and Wildlife Conservation Commission (FWC) Special Activity License (SAL-18-1650(A)-SRP) and with permission from Broward County Environmental Protection & Growth Management. Funding for the study was provided by the U.S. Geological Survey Coastal/Marine Hazards and Resources Program. Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the United States Government.
Author information
Authors and Affiliations
Contributions
ABM, AEO, LTT, and WFP conceived and designed the research. ABM and AEO conducted fieldwork. ABM wrote the original manuscript draft and conducted sample preparation/identification and data analysis. RAM performed radiometric dating. All authors contributed to the reviewing and editing of the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Earth & Environment thanks Emmanuel Hanert and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Aliénor Lavergne. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Modys, A.B., Oleinik, A.E., Toth, L.T. et al. Modern coral range expansion off southeast Florida falls short of Late Holocene baseline. Commun Earth Environ 5, 119 (2024). https://doi.org/10.1038/s43247-024-01283-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s43247-024-01283-0
- Springer Nature Limited