Abstract
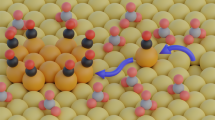
The electrochemical CO2 reduction reaction on Cu is widely believed to occur via two consecutive and orthogonal reaction steps, that is, CO2-to-CO and CO-to-C2+, on the same sites. Here we provide compelling experimental evidence that challenges these long-held assumptions. We show that the presence of CO2 promotes the electrochemical CO reduction reaction, and there are at least two distinct types of Cu sites, with one (CuCO2) more active in the CO2-to-CO conversion and the other (CuCO) favouring the further reduction of CO to C2+ products. CO adsorbed on CuCO is at least six times more active towards the formation of C2+ products than that on CuCO2. Isotopic labelling experiments on Cu(111) and Cu(100) surfaces indicate that CuCO2 and CuCO probably correspond to Cu(111)-like and defect sites, respectively. These insights highlight the possibility of selectivity control in the CO2 reduction reaction.







Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request. Source data are provided with this paper.
References
Centi, G., Quadrelli, E. A. & Perathoner, S. Catalysis for CO2 conversion: a key technology for rapid introduction of renewable energy in the value chain of chemical industries. Energy Environ. Sci. 6, 1711 (2013).
Weber, R. S. Effective use of renewable electricity for making renewable fuels and chemicals. ACS Catal. 9, 946–950 (2019).
Luna, P. D. et al. What would it take for renewably powered electrosynthesis to displace petrochemical processes? Science 364, 350 (2019).
Chu, S., Cui, Y. & Liu, N. The path towards sustainable energy. Nat. Mater. 16, 16–22 (2017).
Hori, Y. et al. Production of methane and ethylene in electrochemical reduction of carbon dioxide at copper electrode in aqueous hydrogencarbonate solution. Chem. Lett. 15, 897–898 (1986).
Kuhl, K. P., Cave, E. R., Abram, D. N. & Jaramillo, T. F. New insights into the electrochemical reduction of carbon dioxide on metallic copper surfaces. Energy Environ. Sci. 5, 7050–7059 (2012).
Raciti, D. & Wang, C. Recent advances in CO2 reduction electrocatalysis on copper. ACS Energy Lett. 3, 1545–1556 (2018).
Wang, L. et al. Electrochemical carbon monoxide reduction on polycrystalline copper: effects of potential, pressure, and pH on aelectivity toward multicarbon and oxygenated products. ACS Catal. 8, 7445–7454 (2018).
Hori, Y., Murata, A., Takahashi, R. & Suzuki, S. Electroreduction of CO to CH4 and C2H4 at a copper electrode in aqueous-solutions at ambienttemperature and pressure. J. Am. Chem. Soc. 109, 5022–5023 (1987).
Li, H., Jiang, K., Zou, S. Z. & Cai, W. B. Fundamental aspects in CO2 electroreduction reaction and solutions from in situ vibrational spectroscopies. Chin. J. Catal. 43, 2772–2791 (2022).
Wuttig, A., Yoon, Y., Ryu, J. & Surendranath, Y. Bicarbonate is not a general acid in Au-catalyzed CO2 electroreduction. J. Am. Chem. Soc. 139, 17109–17113 (2017).
Xiao, H., Cheng, T., Goddard, W. A. 3rd & Sundararaman, R. Mechanistic explanation of the pH dependence and onset potentials for hydrocarbon products from electrochemical reduction of CO on Cu (111). J. Am. Chem. Soc. 138, 483–486 (2016).
Wu, G. L. et al. Selective electroreduction of CO2 to n-propanol in two-step tandem catalytic system. Adv. Energy Mater. 12, 2202054 (2022).
Overa, S. et al. Tandem and hybrid processes for carbon dioxide utilization. Joule 5, 8–13 (2021).
Huang, J. F., Mensi, M., Oveisi, E., Mantella, V. & Buonsanti, R. Structural sensitivities in bimetallic catalysts for electrochemical CO2 reduction revealed by Ag–Cu nanodimers. J. Am. Chem. Soc. 141, 2490–2499 (2019).
Ting, L. R. L. et al. Enhancing CO2 electroreduction to ethanol on copper–silver composites by opening an alternative catalytic pathway. ACS Catal. 10, 4059–4069 (2020).
Lv, X. M. et al. Electron-deficient Cu sites on Cu3Ag1 catalyst promoting CO2 electroreduction to alcohols. Adv. Energy Mater. 10, 2001987 (2020).
Jia, H. L. et al. Symmetry-broken Au–Cu heterostructures and their tandem catalysis process in electrochemical CO2 reduction. Adv. Funct. Mater. 31, 2101255 (2021).
Wang, X. et al. Mechanistic reaction pathways of enhanced ethylene yields during electroreduction of CO2–CO co-feeds on Cu and Cu-tandem electrocatalysts. Nat. Nanotechnol. 14, 1063–1070 (2019).
Li, J. et al. Electrokinetic and in situ spectroscopic investigations of CO electrochemical reduction on copper. Nat. Commun. 12, 3264 (2021).
Chang, X. et al. C–C coupling is unlikely to be the rate-determining step in the formation of C2+ Products in the copper-catalyzed electrochemical reduction of CO. Angew. Chem. Int. Ed. 134, e202111167 (2022).
Chang, X. X. et al. Determining intrinsic stark tuning rates of adsorbed CO on copper surfaces. Catal. Sci. Technol. 11, 6825 (2021).
Chang, X. et al. Understanding the complementarities of surface-enhanced infrared and Raman spectroscopies in CO adsorption and electrochemical reduction. Nat. Commun. 13, 2656 (2022).
Gunathunge, C. M., Ovalle, V. J., Li, Y., Janik, M. J. & Waegele, M. M. Existence of an electrochemically inert CO population on Cu electrodes in alkaline pH. ACS Catal. 8, 7507–7516 (2018).
Lu, X. F., Shinagawa, T. & Takanabe, K. Product distribution control guided by a microkinetic analysis for CO reduction at high-flux electrocatalysis using gas-diffusion Cu electrodes. ACS Catal. 13, 1791–1803 (2023).
Hollins, P. & Pritchard, J. Interactions of CO molecules adsorbed on Cu (111). Surf. Sci. 89, 486–495 (1979).
Lum, Y. & Ager, J. W. Evidence for product-specific active sites on oxide-derived Cu catalysts for electrochemical CO2 reduction. Nat. Catal. 2, 86–93 (2019).
Hou, J. J., Chang, X. X., Li, J., Xu, B. J. & Lu, Q. Correlating CO coverage and CO electroreduction on Cu via high-pressure in situ spectroscopic and reactivity investigations. J. Am. Chem. Soc. 144, 22202–22211 (2022).
Xiong, H. C. et al. Correlating the experimentally determined CO adsorption enthalpy with the electrochemical CO reduction performance on Cu surfaces. Angew. Chem. Int. Ed. 62, e202218447 (2023).
Hori, Y., Takahashi, I., Koga, O. & Hoshi, N. Electrochemical reduction of carbon dioxide at various series of copper single crystal electrodes. J. Mol. Catal. A 199, 39–47 (2003).
Zhong, D. Z. et al. Coupling of Cu (100) and (110) facets promotes carbon dioxide conversion to hydrocarbonsand alcohols. Angew. Chem. Int. Ed. 60, 4879–4885 (2021).
Hori, Y., Wakebe, H., Tsukamoto, T. & Koga, O. Adsorption of CO accompanied with simultaneous charge transfer on copper single crystal electrodes related with electrochemical reduction of CO2 to hydrocarbons. Surf. Sci. 335, 258–263 (1995).
Bagger, A., Ju, W., Varela, A. S., Strasser, P. & Rossmeisl, J. Electrochemical CO2 reduction: a classification problem. ChemPhysChem 18, 3266–3273 (2017).
Li, J. H., Maresi, I., Lum, Y. W. & Ager, J. W. Effects of surface diffusion in electrocatalytic CO2 reduction on Cu revealed by kinetic Monte Carlo simulations. J. Chem. Phys. 155, 164701 (2021).
Schreier, M., Yoon, Y., Jackson, M. N. & Surendranath, Y. Competition between H and CO for active sites governs copper-mediated electrosynthesis of hydrocarbon fuels. Angew. Chem. Int. Ed. 57, 10221–10225 (2018).
Lee, S. H. et al. Oxidation state and surface reconstruction of Cu under CO2 reduction conditions from in situ X-ray characterization. J. Am. Chem. Soc. 143, 588–592 (2021).
Simon, G. H., Kley, C. S. & Cuenya, B. R. Potential-dependent morphology of copper catalysts during CO2 electroreduction revealed by in situ atomic force microscopy. Angew. Chem. Int. Ed. 60, 2561–2568 (2021).
Huang, Y., Handoko, A. D., Hirunsit, P. & Yeo, B. S. Electrochemical reduction of CO2 using copper single-crystal surfaces: effects of CO* coverage on the selective formation of ethylene. ACS Catal. 7, 1749–1756 (2017).
Nitopi, S. et al. Progress and perspectives of electrochemical CO2 reduction on copper in aqueous electrolyte. Chem. Rev. 119, 7610–7672 (2019).
Liu, X. et al. Understanding trends in electrochemical carbon dioxide reduction rates. Nat. Commun. 8, 15438 (2017).
Tang, W. et al. The importance of surface morphology in controlling the selectivity of polycrystalline copper for CO2 electroreduction. Phys. Chem. Chem. Phys. 14, 76–81 (2021).
Jiang, K. et al. Metal ion cycling of Cu foil for selective C–C coupling in electrochemical CO2 reduction. Nat. Catal. 1, 111–119 (2018).
Dunwell, M. et al. The central role of bicarbonate in the electrochemical reduction of carbon dioxide on Gold. J. Am. Chem. Soc. 139, 3774–3783 (2017).
Acknowledgements
This work is supported by the Beijing National Laboratory for Molecular Sciences. W.G. acknowledges the support of the Project funded by China Postdoctoral Science Foundation (2022M710183). The authors thank Y. Zhang and C. Fan for the assistance with numerical analysis.
Author information
Authors and Affiliations
Contributions
W.G., Y.X. and B.X. conceived the idea and designed experiments in this study. W.G. synthesized and characterized the catalysts, and conducted the electrochemical tests. Y.X. performed SEIRAS experiments. L.F. performed part of the electrochemical tests. W.G., Y.X., X.C. and B.X. analysed data, and co-wrote the manuscript, with input from all other co-authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Catalysis thanks Joel Ager III, Wen-Bin Cai and the other, anonymous, reviewer for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–31, Notes 1–6, Tables 1–4 and References 1–8.
Supplementary Data 1
Source data for supplementary figures.
Source data
Source Data Fig. 1
Co-electrolysis of CO/CO2 and in situ spectroscopic characterization.
Source Data Fig. 2
Co-electrolysis of 13CO/12CO2 (4:1) on Den-Cu.
Source Data Fig. 3
Distribution of ethylene isotopologues in the co-electrolysis of 13CO/12CO2.
Source Data Fig. 4
Isotopologue distribution of C2 products in the co-electrolysis of 13CO/12CO2.
Source Data Fig. 5
The two-site model and its predictions.
Source Data Fig. 6
Distribution of ethylene isotopologues on single crystal Cu surface.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gao, W., Xu, Y., Fu, L. et al. Experimental evidence of distinct sites for CO2-to-CO and CO conversion on Cu in the electrochemical CO2 reduction reaction. Nat Catal 6, 885–894 (2023). https://doi.org/10.1038/s41929-023-01002-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-023-01002-6
- Springer Nature Limited
This article is cited by
-
Stabilized Cu0 -Cu1+ dual sites in a cyanamide framework for selective CO2 electroreduction to ethylene
Nature Communications (2024)
-
Turning copper into an efficient and stable CO evolution catalyst beyond noble metals
Nature Communications (2024)
-
How local electric field regulates C–C coupling at a single nanocavity in electrocatalytic CO2 reduction
Nature Communications (2024)
-
Interfacially coupled Cu-cluster/GaN photocathode for efficient CO2 to ethylene conversion
Nature Synthesis (2024)
-
Revealing the structural evolution of CuAg composites during electrochemical carbon monoxide reduction
Nature Communications (2024)