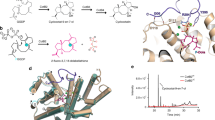
Abstract
Terpene synthases (TSs) catalyse the most complex cyclization cascades in nature, with generation and taming of reactive carbocations. Although deprotonation–reprotonation sequences are frequently relevant for TS catalysis, little is known how the enzyme acts in these processes. Here we show, through quantum mechanics (density functional theory)/molecular mechanics molecular dynamics simulations that the main-chain carbonyl oxygen of Gly182 of selina-4(15),7(11)-diene synthase (SdS) has a dual role as a base and an acid and acts in synchrony with one water molecule. The computational model is supported by isotopic labelling experiments confirming the predicted stereochemical course associated with the deprotonation–reprotonation sequence. Gly182 is located within the G1/2 helix break of SdS, with all backbone carbonyl oxygens pointing into the active site having functions in recognizing substrate conformation, stabilizing carbocation intermediates and anchoring their poses. The strict conservation of the G1/2 helix break in type I TSs from bacteria, fungi and plants suggests that its functions as described here may be of general importance in TS catalysis.







Similar content being viewed by others
Data availability
The authors declare that data supporting the findings of this study are available within the article and its Supplementary Information file. FPP parameters and key structures are given as Supplementary Data files. Data that support the plots within the paper and other findings of this study are available from the corresponding authors on reasonable request.
References
Christianson, D. W. Structural and chemical biology of terpenoid cyclases. Chem. Rev. 117, 11570–11648 (2017).
Christianson, D. W. Structural biology and chemistry of the terpenoid cyclases. Chem. Rev. 106, 3412–3442 (2006).
Dickschat, J. S. Bacterial diterpene biosynthesis. Angew. Chem. Int. Ed. 58, 15964–15976 (2019).
Hong, Y. J. & Tantillo, D. J. Biosynthetic consequences of multiple sequential post-transition-state bifurcations. Nat. Chem. 6, 104–111 (2014).
Tantillo, D. J. Biosynthesis via carbocations: theoretical studies on terpene formation. Nat. Prod. Rep. 28, 1035–1053 (2011).
Hong, Y. J. & Tantillo, D. J. Branching out from the bisabolyl cation. Unifying mechanistic pathways to barbatene, bazzanene, chamigrene, chamipinene, cumacrene, cuprenene, dunniene, isobazzanene, iso-gamma-bisabolene, isochamigrene, laurene, microbiotene, sesquithujene, sesquisabinene, thujopsene, trichodiene, and widdradiene sesquiterpenes. J. Am. Chem. Soc. 136, 2450–2463 (2014).
Hong, Y. J. & Tantillo, D. J. Consequences of conformational preorganization in sesquiterpene biosynthesis: theoretical studies on the formation of the bisabolene, curcumene, acoradiene, zizaene, cedrene, duprezianene, and sesquithuriferol sesquiterpenes. J. Am. Chem. Soc. 131, 7999–8015 (2009).
Mahadevi, A. S. & Sastry, G. N. Cation-pi interaction: its role and relevance in chemistry, biology, and material science. Chem. Rev. 113, 2100–2138 (2013).
Tantillo, D. J. Importance of inherent substrate reactivity in enzyme-promoted carbocation syclization/rearrangements. Angew. Chem. Int. Ed. 56, 10040–10045 (2017).
Chen, M. et al. Mechanistic insights from the binding of substrate and carbocation intermediate analogues to aristolochene synthase. Biochemistry 52, 5441–5453 (2013).
Noel, J. P. et al. Structural elucidation of cisoid and transoid cyclization pathways of a sesquiterpene synthase using 2-fluorofarnesyl diphosphates. ACS Chem. Biol. 5, 377–392 (2010).
Minami, A., Ozaki, T., Liu, C. & Oikawa, H. Cyclopentane-forming di/sesterterpene synthases: widely distributed enzymes in bacteria, fungi, and plants. Nat. Prod. Rep. 35, 1330–1346 (2018).
Rinkel, J. et al. Mechanisms of the diterpene cyclases β-pinacene synthase from Dictyostelium discoideum and hydropyrene synthase from Streptomyces clavuligerus. Chem. Eur. J. 23, 10501–10505 (2017).
Rinkel, J., Lauterbach, L. & Dickschat, J. S. Spata-13,17-diene synthase—an enzyme with sesqui-, di-, and sesterterpene synthase activity from Streptomyces xinghaiensis. Angew. Chem. Int. Ed. 56, 16385–16389 (2017).
Bian, G. K. et al. A clade II-D fungal chimeric diterpene synthase from Colletotrichum gloeosporioides produces dolasta-1(15),8-diene. Angew. Chem. Int. Ed. 57, 15887–15890 (2018).
Rinkel, J., Lauterbach, L. & Dickschat, J. S. A branched diterpene cascade: the mechanism of spinodiene synthase from Saccharopolyspora spinosa. Angew. Chem. Int. Ed. 58, 452–455 (2019).
Zhang, F., Chen, N. H., Zhou, J. W. & Wu, R. B. Protonation-dependent diphosphate cleavage in FPP cyclases and synthases. ACS Catal. 6, 6918–6929 (2016).
Hong, Y. J. & Tantillo, D. J. Feasibility of intramolecular proton transfers in terpene biosynthesis-guiding principles. J. Am. Chem. Soc. 137, 4134–4140 (2015).
Seemann, M. et al. Pentalenene synthase. Analysis of active site residues by site-directed mutagenesis. J. Am. Chem. Soc. 124, 7681–7689 (2002).
Jiang, J., He, X. & Cane, D. E. Biosynthesis of the earthy odorant geosmin by a bifunctional Streptomyces coelicolor enzyme. Nat. Chem. Biol. 3, 711–715 (2007).
Baer, P. et al. Induced-fit mechanism in class I terpene cyclases. Angew. Chem. Int. Ed. 53, 7652–7656 (2014).
Köksal, M., Chou, W. K. W., Cane, D. E. & Christianson, D. W. Structure of 2-methylisoborneol synthase from Streptomyces coelicolor and implications for the cyclization of a noncanonical C-methylated monoterpenoid substrate. Biochemistry 51, 3011–3020 (2012).
Aaron, J. A., Lin, X., Cane, D. E. & Christianson, D. W. Structure of epi-isozizaene synthase from Streptomyces coelicolor A3(2), a platform for new terpenoid cyclization templates. Biochemistry 49, 1787–1797 (2010).
Xu, H., Rinkel, J. R. & Dickschat, J. S. Isoishwarane synthase from Streptomyces lincolnensis. Org. Chem. Front. 8, 1177–1184 (2021).
Lauterbach, L., Rinkel, J. & Dickschat, J. S. Two bacterial diterpene synthases from Allokutzneria albata for bonnadiene and for phomopsene and allokutznerene. Angew. Chem. Int. Ed. 57, 8280–8283 (2018).
Tantillo, D. J. Walking in the woods with quantum chemistry—applications of quantum chemical calculations in natural products research. Nat. Prod. Rep. 30, 1079–1086 (2013).
Ganguly, A., Thaplyal, P., Rosta, E., Bevilacqua, P. C. & Hammes-Schiffer, S. Quantum mechanical/molecular mechanical free energy simulations of the self-cleavage reaction in the hepatitis Delta virus ribozyme. J. Am. Chem. Soc. 136, 1483–1496 (2014).
Rooklin, D. W., Lu, M. & Zhang, Y. Revelation of a catalytic calcium-binding site elucidates unusual metal dependence of a human apyrase. J. Am. Chem. Soc. 134, 15595–15603 (2012).
Ke, Z., Smith, G. K., Zhang, Y. & Guo, H. Molecular mechanism for eliminylation, a newly discovered post-translational modification. J. Am. Chem. Soc. 133, 11103–11105 (2011).
Wang, Y.-H., Xie, H., Zhou, J., Zhang, F. & Wu, R. Substrate folding modes in trichodiene synthase: a determinant of chemo- and stereoselectivity. ACS Catal. 7, 5841–5846 (2017).
Diao, H. et al. Biosynthetic mechanism of lanosterol: a completed story. ACS Catal. 10, 2157–2168 (2020).
Chen, N., Wang, S., Smentek, L., Hess, B. A. & Wu, R. Biosynthetic mechanism of lanosterol: cyclization. Angew. Chem. Int. Ed. 54, 8693–8696 (2015).
Das, S., Dixit, M. & Major, D. T. First principles model calculations of the biosynthetic pathway in selinadiene synthase. Bioorg. Med. Chem. 24, 4867–4870 (2016).
Rabe, P. et al. Terpene cyclases from social Amoebae. Angew. Chem. Int. Ed. 55, 15420–15423 (2016).
Rinkel, J. & Dickschat, J. S. Addressing the chemistry of germacrene A by isotope labeling experiments. Org. Lett. 21, 2426–2429 (2019).
Hahn, F. M., Hurlburt, A. P. & Poulter, C. D. Escherichia coli open reading frame 696 is idi, a nonessential gene encoding isopentenyl diphosphate isomerase. J. Bacteriol. 181, 4499–4504 (1999).
Cornforth, J. W., Cornforth, R. H., Popjak, G. & Yengoyan, L. Sudies on the biosynthesis of cholesterol XX. Steric course of decarboxylation of 5-pyrophosphomevalonate and of the carbon to carbon bond formation in the biosynthesis of farnesyl pyrophosphate. J. Biol. Chem. 241, 3970–3987 (1966).
Zhou, J. et al. Protonation-triggered carbon-chain elongation in geranyl pyrophosphate synthase (GPPS). ACS Catal. 5, 4466–4478 (2015).
Rabe, P. et al. Conformational analysis, thermal rearrangement and EI-MS-fragmentation mechanism of (1(10)E,4E,6S,7R)-germacradien-6-ol by 13C-labeling experiments. Angew. Chem. Int. Ed. 54, 13448–13451 (2015).
Lin, F.-L. et al. Mechanistic characterization of the fusicoccane-type diterpene synthase for myrothec-15(17)-en-7-ol. ACS Catal. 10, 4306–4312 (2020).
Dereka, B. et al. Crossover from hydrogen to chemical bonding. Science 371, 160–164 (2021).
Rabe, P., Pahirulzaman, K. A. K. & Dickschat, J. S. Structures and biosynthesis of corvol ethers—sesquiterpenes from the actinomycete Kitasatospora setae. Angew. Chem. Int. Ed. 54, 6041–6045 (2015).
Rabe, P., Rinkel, J., Klapschinski, T. A., Barra, L. & Dickschat, J. S. A method for investigating the stereochemical course of terpene cyclisations. Org. Biomol. Chem. 14, 158–164 (2016).
Xu, H. & Dickschat, J. S. Germacrene A–a central intermediate in sesquiterpene biosynthesis. Chem. Eur. J. 26, 17318–17341 (2020).
Potter, K., Criswell, J., Zi, J., Stubbs, A. & Peters, R. J. Novel product chemistry from mechanistic analysis of ent-copalyl diphosphate synthases from plant hormone biosynthesis. Angew. Chem. Int. Ed. 53, 7198–7202 (2014).
Potter, K. C. et al. Blocking deprotonation with retention of aromaticity in a plant ent-copalyl diphosphate synthase leads to product rearrangement. Angew. Chem. Int. Ed. 55, 634–638 (2016).
Srivastava, P. L. et al. Redesigning the molecular choreography to prevent hydroxylation in germacradien-11-ol synthase catalysis. ACS Catal. 11, 1033–1041 (2021).
Acknowledgements
This work was supported by the German Research Foundation DFG (no. DI1536/7-2 to J.S.D.); the National Key Research and Development Program of China (nos. 2018YFA0903200 and 2018YFA0903201 to H.G.); the National Natural Science Foundation of China (nos. 21803080 and 32070042 to Y.-H.W., 81925037 to H.G., 81872759 to P.-H.S. and 21773313 to R.W.); the National High-level Personnel of Special Support Program (no. 2017RA2259 to H.G.); the Chang Jiang Scholars Program (Young Scholar) from the Ministry of Education of China (to H.G.); and the K. C. Wong Education Foundation (to H.G.). We thank X.-S. Yao (Guangzhou) for his support of this study through the 111 Project of the Ministry of Education of the People’s Republic of China (no. B13038), and the Guangzhou and Shenzhen Supercomputer Center for providing the computational source.
Author information
Authors and Affiliations
Contributions
J.S.D., R.W. and Y.-H.W. designed the research. J.S.D. supervised the experimental procedures. R.W. and P.-H.S. supervised the computational work. R.W. provided the Qchem–Tinker software package. Y.-H.W. and J.Z. performed the QM/MM MD simulations. H.X. and E.C. carried out the experimental work. Y.-H.W., J.Z., X.-B.C., Y.-Q.Z., W.-L.L., G.-D.C., D.H., H.G. and P.-H.S. analysed computational data. Y.-H.W., J.S.D., J.Z. and H.X. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Catalysis thanks Hideaki Oikawa, Per-Olof Syren and Marc van der Kamp for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Methods, Tables 1 and 2 and Figs. 1–26.
Supplementary Data 1
Computed structure FPP-exo.
Supplementary Data 2
Computed structure FPP-endo.
Supplementary Data 3
Computed structure I-endo.
Supplementary Data 4
Computed structure I-exo.
Supplementary Data 5
Computed structure TS II.
Supplementary Data 6
Computed structure II.
Supplementary Data 7
Computed structure TS II–III.
Supplementary Data 8
Computed structure III.
Supplementary Data 9
Computed structure TS III–IV.
Supplementary Data 10
Computed structure IV.
Supplementary Data 11
Computed structure TS IV–V.
Supplementary Data 12
Computed structure V.
Supplementary Data 13
Computed structure TS V–VI.
Supplementary Data 14
Computed structure VI.
Rights and permissions
About this article
Cite this article
Wang, YH., Xu, H., Zou, J. et al. Catalytic role of carbonyl oxygens and water in selinadiene synthase. Nat Catal 5, 128–135 (2022). https://doi.org/10.1038/s41929-022-00735-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-022-00735-0
- Springer Nature Limited
This article is cited by
-
Molecular insights into the catalytic promiscuity of a bacterial diterpene synthase
Nature Communications (2023)