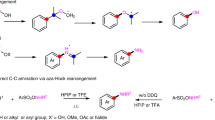
Abstract
Aryl–aryl cross-coupling constitutes one of the most widely used procedures for the synthesis of high-value materials that range from pharmaceuticals to organic electronics and conducting polymers. The assembly of (hetero)biaryl scaffolds generally requires multiple steps; coupling partners must be functionalized before the key bond-forming event is considered. The development of selective C–H arylation processes in arenes—which sidestep the need for prefunctionalized partners—is thus crucial for streamlining the construction of these key architectures. Here we report an expedient, one-pot assembly of (hetero)biaryl motifs using photocatalysis and two non-prefunctionalized arene partners. The approach is underpinned by the functionalization of a C–H bond in an arene coupling partner using the interrupted Pummerer reaction. A unique pairing of the organic photoredox catalyst and the intermediate dibenzothiophenium salts enables highly selective reduction in the presence of sensitive functionalities. The utility of the metal-free, one-pot strategy is exemplified by the synthesis of a bioactive natural product and the modification of complex molecules of societal importance.




Similar content being viewed by others
Data availability
All data are available in the main text or Supplementary Information. Metrical parameters for the structures of 2, 60 and S12 (see Supplementary Information) are available free of charge from the Cambridge Crystallographic Data Centre (https://www.ccdc.cam.ac.uk/) under reference nos. 1922367, 1922368 and 1960231.
References
Johansson Seechurn, C. C. C., Kitching, M. O., Colacot, T. J. & Snieckus, V. Palladium‐catalyzed cross‐coupling: a historical contextual perspective to the 2010 Nobel Prize. Angew. Chem. Int. Ed. 51, 5062–5085 (2012).
Roughley, S. D. & Jordan, A. M. The medicinal chemist’s toolbox: an analysis of reactions used in the pursuit of drug candidates. J. Med. Chem. 54, 3451–3479 (2011).
Brown, D. G. & Boström, J. Analysis of past and present synthetic methodologies on medicinal chemistry: where have all the new reactions gone? J. Med. Chem. 59, 4443–4458 (2016).
Yang, Y., Lan, J. & You, J. Oxidative C–H/C–H coupling reactions between two (hetero)arenes. Chem. Rev. 117, 8787–8863 (2017).
Sambiagio, C. et al. A comprehensive overview of directing groups applied in metal-catalysed C–H functionalisation chemistry. Chem. Soc. Rev. 47, 6603–6743 (2018).
Hari, D. P., Schroll, P. & Konig, B. Metal-free, visible-light-mediated direct C–H arylation of heteroarenes with aryl diazonium salts. J. Am. Chem. Soc. 134, 2958–2961 (2012).
Ghosh, I., Marzo, L., Das, A., Shaikh, R. & König, B. Visible light mediated photoredox catalytic arylation reactions. Acc. Chem. Res. 49, 1566–1577 (2016).
Nguyen, J. D., D’Amato, E. M., Narayanam, J. M. R. & Stephenson, C. R. J. Engaging unactivated alkyl, alkenyl and aryl iodides in visible-light-mediated free radical reactions. Nat. Chem. 4, 854–859 (2012).
Kim, H. & Lee, C. Visible-light-induced photocatalytic reductive transformations of organohalides. Angew. Chem. Int. Ed. 51, 12303–12306 (2012).
Ghosh, I., Ghosh, T., Bardagi, J. I. & König, B. Reduction of aryl halides by consecutive visible light-induced electron transfer processes. Science 346, 725–728 (2014).
Marzo, L., Ghosh, I., Esteban, F. & König, B. Metal-free photocatalyzed cross coupling of bromoheteroarenes with pyrroles. ACS Catal. 6, 6780–6784 (2016).
Ghosh, I. & König, B. Chromoselective photocatalysis: controlled bond activation through light-color regulation of redox potentials. Angew. Chem. Int. Ed. 55, 7676–7679 (2016).
Ghosh, I., Shaikh, R. S. & König, B. Sensitization-initiated electron transfer for photoredox catalysis. Angew. Chem. Int. Ed. 56, 8544–8549 (2017).
Smith, L. H. S., Coote, S. C., Sneddon, H. F. & Procter, D. J. Beyond the Pummerer reaction: recent developments in thionium ion chemistry. Angew. Chem. Int. Ed. 49, 5832–5844 (2010).
Eberhart, A. J., Imbriglio, J. E. & Procter, D. J. Nucleophilic ortho allylation of aryl and heteroaryl sulfoxides. Org. Lett. 13, 5882–5885 (2011).
Huang, X. & Maulide, N. Sulfoxide-mediated α-arylation of carbonyl compounds. J. Am. Chem. Soc. 133, 8510–8513 (2011).
Eberhart, A. J. & Procter, D. J. Nucleophilic ortho-propargylation of aryl sulfoxides: an interrupted Pummerer/allenyl thio-claisen rearrangement sequence. Angew. Chem. Int. Ed. 52, 4008–4011 (2013).
Huang, X., Patil, M., Fares, C., Thiel, W. & Maulide, N. Sulfur(iv)-mediated transformations: from ylide transfer to metal free arylation of carbonyl compounds. J. Am. Chem. Soc. 135, 7312–7323 (2013).
Peng, B., Huang, X., Xie, L.-G. & Maulide, N. A Brønsted acid catalyzed redox arylation. Angew. Chem. Int. Ed. 53, 8718–8721 (2014).
Peng, B., Geerdink, D., Fars, C. & Maulide, N. Chemoselective intermolecular α-arylation of amides. Angew. Chem. Int. Ed. 53, 5462–5466 (2014).
Pulis, A. P. & Procter, D. J. C–H coupling reactions directed by sulfoxides: teaching an old functional group new tricks. Angew. Chem. Int. Ed. 55, 9842–9860 (2016).
Fernandez-Salas, J. A., Eberhart, A. J. & Procter, D. J. Metal-free CH–CH-type cross-coupling of arenes and akynes directed by a multifunctional sulfoxide group. J. Am. Chem. Soc. 138, 790–793 (2016).
Cowper, P., Jin, Y., Turton, M. D., Kociok-Köhn, G. & Lewis, S. E. Azulenesulfonium salts: accessible, stable, and versatile reagents for cross-coupling. Angew. Chem. Int. Ed. 55, 2564–2568 (2016).
Li, Y. et al. Aryne 1,2,3-trifunctionalization with aryl allyl sulfoxides. J. Am. Chem. Soc. 138, 10814–10817 (2016).
Yanagi, T. et al. Metal-free approach to biaryls from phenols and aryl sulfoxides by temporarily sulfur-tethered regioselective C–H/C–H coupling. J. Am. Chem. Soc. 138, 14582–14585 (2016).
Shrives, H. J., Fernandez-Salas, J. A., Hedtke, C., Pulis, A. P. & Procter, D. J. Regioselective synthesis of C3 alkylated and arylated benzothiophenes. Nat. Commun. 8, 14801 (2017).
Kaldre, D. et al. An asymmetric redox arylation: chirality transfer from sulfur to carbon through a sulfonium [3,3]-sigmatropic rearrangement. Angew. Chem. Int. Ed. 56, 2212–2215 (2017).
Chen, D. et al. Metal-free O–H/C–H difunctionalization of phenols by o-hydroxyarylsulfonium salts in water. Chem. Sci. 8, 1601–1606 (2017).
Yorimitsu, H. Cascades of interrupted Pummerer reaction-sigmatropic rearrangement. Chem. Rec. 17, 1156–1167 (2017).
Shang, L. et al. Redox-neutral α-arylation of alkyl nitriles with aryl sulfoxides: a rapid electrophilic rearrangement. J. Am. Chem. Soc. 139, 4211–4217 (2017).
He, Z. et al. Synthesis of C2 substituted benzothiophenes via an interrupted Pummerer/[3,3]-sigmatropic/1,2-migration cascade of benzothiophene S-oxides. Angew. Chem. Int. Ed. 57, 5759–5764 (2018).
Zhang, L. et al. Selective [5,5]-sigmatropic rearrangement by assembly of aryl sulfoxides with allyl nitriles. Angew. Chem. Int. Ed. 58, 5316–5320 (2019).
Berger, F. et al. Site-selective and versatile aromatic C–H functionalization by thianthrenation. Nature 567, 223–228 (2019).
He, Z., Pulis, A. P. & Procter, D. J. The interrupted Pummerer reaction in a sulfoxide-catalyzed oxidative coupling of 2-naphthols. Angew. Chem. Int. Ed. 58, 7813–7817 (2019).
He, Z., Pulis, A. P., Perry, G. J. & Procter, D. J. Pummerer chemistry of benzothiophene S-oxides: metal-free alkylation and arylation of benzothiophenes. Phosphorus Sulfur Silicon Relat. Elem. 194, 669–677 (2019).
Šiaučiulis, M., Ahlsten, N., Pulis, A. P. & Procter, D. J. Transition-metal-free cross-coupling of benzothiophenes and styrenes in a stereoselective synthesis of substituted (E,Z)-1,3-dienes. Angew. Chem. Int. Ed. 58, 8779–8783 (2019).
Aukland, M. H., Talbot, F. J. T., Fernandez-Salas, J. A., Ball, M., Pulis, A. P. & Procter, D. J. An interrupted Pummerer/nickel-catalysed cross-coupling sequence. Angew. Chem. Int. Ed. 57, 9785–9789 (2018).
Prier, C. K., Rankic, D. A. & MacMillan, D. W. C. Visible light photoredox catalysis with transition metal complexes: applications in organic synthesis. Chem. Rev. 113, 5322–5363 (2013).
Romero, N. A. & Nicewicz, D. A. Organic photoredox catalysis. Chem. Rev. 116, 10075–10166 (2016).
Cismesia, M. A. & Yoon, T. P. Characterizing chain processes in visible light photoredox catalysis. Chem. Sci. 6, 5426–5434 (2015).
Hedstrand, D. M., Kruizunga, W. H. & Kellogg, R. M. Light induced and dye accelerated reductions of phenacyl onium salts by 1,4-dihydropyridines. Tetrahedron Lett. 14, 1255–1258 (1978).
Otsuka, S., Nogi, K., Rovis, T. & Yorimitsu, H. Photoredox-catalyzed alkenylation of benzylsulfonium salts. Chem. Asian J. 14, 532–536 (2019).
Donck, S., Baroudi, A., Fensterbank, L., Goddard, J.-P. & Ollivier, C. Visible-light photocatalytic reduction of sulfonium salts as a source of aryl radicals. Adv. Synth. Catal. 355, 1477–1482 (2013).
Liu, W. et al. Organocatalysis in cross-coupling: DMEDA-catalyzed direct C–H arylation of unactivated benzene. J. Am. Chem. Soc. 132, 16737–16740 (2010).
Discekici, E. H. et al. A highly reducing metal-free photoredox catalyst: design and application in radical dehalogenations. Chem. Commun. 51, 11705–11708 (2015).
Garrido-Castro, A. F., Salaverri, N., Maestro, M. C. & Aleman, J. Intramolecular homolytic substitution enabled by photoredox catalysis: sulfur, phosphorus, and silicon heterocycle synthesis from aryl halides. Org. Lett. 21, 5295–3000 (2019).
Boyington, A. J., Seath, C. P., Zearfoss, A. M., Xu, Z. & Jui, N. T. Catalytic strategy for regioselective arylethylamine synthesis. J. Am. Chem. Soc. 141, 4147–4153 (2019).
Martinez-Gualda, A. M. et al. Chromoselective access to Z- or E- allylated amines and heterocycles by a photocatalytic allylation reaction. Nat. Commun. 10, 2634 (2019).
Wang, H. & Jui, N. T. Catalytic defluoroalkylation of trifluoromethylaromatics with unactivated alkenes. J. Am. Chem. Soc. 140, 163–166 (2018).
Yanagi, T., Nogi, K. & Yorimitsu, H. Construction of biaryls from aryl sulfoxides and anilines by means of a sigmatropic rearrangement. Chem. Eur. J. https://doi.org/10.1002/chem.201903570 (2019).
Kunfermann, A. et al. Pseudilins: halogenated, allosteric inhibitors of the non-mevalonate pathway enzyme IspD. Angew. Chem. Int. Ed. 53, 2235–2239 (2014).
Preller, M., Chinthalapudi, K., Martin, R., Knölker, H.-J. & Manstein, D. J. Inhibition of myosin ATPase activity by halogenated pseudilins: a structure–activity study. J. Med. Chem. 54, 3675–3685 (2011).
Martin, R. et al. Total synthesis of pentabromo- and pentachloropseudilin, and synthetic analogues—allosteric inhibitors of myosin ATPase. Angew. Chem. Int. Ed. 48, 8042–8046 (2009).
Acknowledgements
We thank the EPSRC (Doctoral Prize Fellowship to M.H.A.; Established Career Fellowship to D.J.P.) and The University of Manchester (Lectureship to G.J.P.P.; Studentship to M.Š.) for their generous support. Additional thanks go to L. Natrajan and F. J. Hernandez for their assistance with the fluorescence quenching, electrochemical studies and quantum yield determination, as well as to D. Leonori for his advice on the design of the experiments.
Author information
Authors and Affiliations
Contributions
M.H.A. and D.J.P. conceived the study and co-wrote the manuscript. M.H.A. designed and performed experiments and M.Š., A.W. and G.J.P.P. performed experiments.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Methods, Figs. 1–12, Tables 1–6 and references.
Compound 2
Crystallographic information for Compound 2.
Compound 60
Crystallographic information for Compound 60.
Compound S12
Crystallographic information for Compound S12.
Rights and permissions
About this article
Cite this article
Aukland, M.H., Šiaučiulis, M., West, A. et al. Metal-free photoredox-catalysed formal C–H/C–H coupling of arenes enabled by interrupted Pummerer activation. Nat Catal 3, 163–169 (2020). https://doi.org/10.1038/s41929-019-0415-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-019-0415-3
- Springer Nature Limited
This article is cited by
-
Metallaphotoredox deuteroalkylation utilizing thianthrenium salts
Nature Communications (2024)
-
Modular assembly of arenes, ethylene and heteroarenes for the synthesis of 1,2-arylheteroaryl ethanes
Nature Chemistry (2024)
-
A general arene C–H functionalization strategy via electron donor–acceptor complex photoactivation
Nature Chemistry (2023)
-
Radical arenes
Nature Chemistry (2023)
-
Anti-Markovnikov ring-opening of sulfonium salts with alkynes by visible light/copper catalysis
Science China Chemistry (2022)