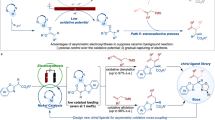
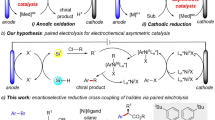
Abstract
Catalytic asymmetric electrosynthesis combines the unique features of an electrochemical addition or removal of electrons with the catalytic asymmetric synthesis of enantioenriched molecules. However, identifying suitable catalysts that are compatible with electrochemical conditions and provide a high stereocontrol is a formidable challenge. Here we introduce a versatile electricity-driven chiral Lewis acid catalysis for the oxidative cross-coupling of 2-acyl imidazoles with silyl enol ethers. Powered by an electric current, this work provides a sustainable avenue to synthetically useful non-racemic 1,4-dicarbonyls, which include products that bear all-carbon quaternary stereocentres. A chiral-at-metal rhodium catalyst activates a substrate towards anodic oxidation by raising the highest occupied molecular orbital on enolate formation, which enables mild redox conditions, high chemo- and enantioselectivities (up to >99% enantiomeric excess) and a broad substrate scope. This work demonstrates the potential of combining asymmetric Lewis acid catalysis with electrochemistry and we anticipate that it will spur the further development of catalytic asymmetric electrosynthesis.





Similar content being viewed by others
Data availability
The X-ray crystallographic coordinates for the structures of Λ-Rh2, 3g, 6n and Rh2-1a reported in this article have been deposited at the Cambridge Crystallographic Data Centre (CCDC) under deposition numbers CCDC 1866726, 1866727, 1868792 and 1866728, respectively. The data can be obtained free of charge from the CCDC via https://www.ccdc.cam.ac.uk/structures/. All other data are available from the authors upon reasonable request.
References
Degner, D. in Electrochemistry III (ed. Steckchan, E.) 1–95 (Springer, Berlin, 1988).
Yoshida, J., Kataoka, K., Horcajada, R. & Nagaki, A. Modern strategies in electroorganic synthesis. Chem. Rev. 108, 2265–2299 (2008).
Yan, M., Kawamata, Y. & Baran, P. S. Synthetic organic electrochemical methods since 2000: on the verge of a renaissance. Chem. Rev. 117, 13230–13319 (2017).
Moeller, K. D. Using physical organic chemistry to shape the course of electrochemical reactions. Chem. Rev. 118, 4817–4833 (2018).
Nutting, J. E., Rafiee, M. & Stahl, S. S. Tetramethylpiperidine N-oxyl (TEMPO), phthalimide N-oxyl (PINO), and related N-oxyl species: electrochemical properties and their use in electrocatalytic reactions. Chem. Rev. 118, 4834–4885 (2018).
Sauer, G. S. & Lin, S. An electrocatalytic approach to the radical difunctionalization of alkenes. ACS Catal. 8, 5175–5187 (2018).
Sauermann, N., Meyer, T. H., Qiu, Y. & Ackermann, L. Electrocatalytic C–H activation. ACS Catal. 8, 7086–7103 (2018).
Trost, B. M. Asymmetric catalysis: an enabling science. Proc. Natl Acad. Sci. USA 101, 5348–5355 (2004).
Walsh, P. J. & Kozlowski, M. C. Fundamentals of Asymmetric Catalysis (University Science Books, Herndon, 2009).
Klotz-Berendes, B., Schäfer, H. J., Grehl, M. & Fröhlich, R. Diastereoselective coupling of anodically generated radicals bearing chiral amide groups. Angew. Chem. Int. Ed. Engl. 34, 189–191 (1995).
Kise, N., Ozaki, H., Moriyama, N., Kitagishi, Y. & Ueda, N. Electroreductive intramolecular coupling of chiral α-imino esters: stereoselective synthesis of mixed ketals of cis-2,4-disubstituted azetidine-3-ones. J. Am. Chem. Soc. 125, 11591–11596 (2003).
Kise, N., Hamada, Y. & Sakurai, T. Electroreductive coupling of optically active α,β-unsaturated carbonyl compounds with diaryl ketones: asymmetric synthesis of 4,5,5-trisubstituted γ-butyrolactones. Org. Lett. 16, 3348–3351 (2014).
Cao, Z.-Y. & Zhou, J. in Multicatalyst System in Asymmetric Catalysis (ed. Zhou, J.) 475–500 (John Wiley & Sons, Hoboken, 2014).
Hammerich, O. & Speiser, B. Organic Electrochemistry 5th edn (CRC Press, Boca Raton, 2016).
Page, P. C. B. et al. Enantioselective organocatalytic epoxidation driven by electrochemically generated percarbonate and persulfate. Adv. Synth. Catal. 350, 1149–1154 (2008).
Jensen, K. L., Franke, P. T., Nielsen, L. T., Daasbjerg, K. & Jørgensen, K. A. Anodic oxidation and organocatalysis: direct regio- and stereoselective access to meta-substituted anilines by α-arylation of aldehydes. Angew. Chem. Int. Ed. 49, 129–133 (2010).
Fu, N., Li, L., Yang, Q. & Luo, S. Catalytic asymmetric electrochemical oxidative coupling of tertiary amines with simple ketones. Org. Lett. 19, 2122–2125 (2017).
Kashiwagi, Y.et al. Enantioselective electrocatalytic oxidation of racemic amines using a chiral 1-azaspiro[5.5]undecane N-oxyl radical. Chem. Commun. 1983–1984 (1999)
Kashiwagi, Y. et al. Asymmetric electrochemical lactonization of diols on a chiral 1-azaspiro[5.5]undecane N-oxyl radical mediator-modified graphite felt electrode. Chem. Commun. 114–115 (2003).
Moutet, J.-C., Duboc-Toia, C., Ménage, S. & Tingry, S. A Chiral poly(2,2′-bipyridyl rhodium(iii) complex) film electrode for asymmetric induction in electrosynthesis. Adv. Mater. 10, 665–667 (1999).
Moutet, J.-C. et al. Heterogeneous and homogeneous asymmetric electrocatalytic hydrogenation with rhodium(iii) complexes containing chiral polypyridyl ligands. New J. Chem. 23, 939–944 (1999).
Franco, D., Riahi, A., Hénin, F., Muzart, J. & Duñach, E. Electrochemical reduction of a racemic allyl β-keto ester catalyzed by nickel complexes: asymmetric induction. Eur. J. Org. Chem. 2257–2259 (2002)
Francke, R. & Little, R. D. Redox catalysis in organic electrosynthesis: basic principles and recent developments. Chem. Soc. Rev. 43, 2492–2521 (2014).
Torii, S., Liu, P., Bhuvaneswari, N., Amatore, C. & Jutand, A. Chemical and electrochemical asymmetric dihydroxylation of olefins in I2−K2CO3−K2OsO2(OH)4 and I2−K3PO4/K2HPO4−K2OsO2(OH)4 systems with Sharpless’ ligand. J. Org. Chem. 61, 3055–3060 (1996).
Nguyen, B. H., Redden, A. & Moeller, K. D. Sunlight, electrochemistry, and sustainable oxidation reactions. Green Chem. 16, 69–72 (2014).
Tanaka, H., Kuroboshi, M., Takeda, H., Kanda, H. & Torii, S. Electrochemical asymmetric epoxidation of olefins by using an optically active Mn–salen complex. J. Electroanal. Chem. 507, 75–81 (2001).
Chen, B.-L. et al. Asymmetric electrocarboxylation of 1-phenylethyl chloride catalyzed by electrogenerated chiral [Coi(salen)]− complex. Electrochem. Commun. 42, 55–59 (2014).
Yuan, R., Watanabe, S., Kuwabata, S. & Yoneyama, H. Asymmetric electroreduction of ketone and aldehyde derivatives to the corresponding alcohols using alcohol dehydrogenase as an electrocatalyst. J. Org. Chem. 62, 2494–2499 (1997).
Kawabata, S., Iwata, N. & Yoneyama, H. Asymmetric electrosynthesis of amino acid using an electrode modified with amino acid oxidase and electron mediator. Chem. Lett. 29, 110–111 (2000).
Hollmann, F., Hofstetter, K., Habicher, T., Hauer, B. & Schmid, A. Direct electrochemical regeneration of monooxygenase subunits for biocatalytic asymmetric epoxidation. J. Am. Chem. Soc. 127, 6540–6541 (2005).
Höllrigl, V., Otto, K. & Schmid, A. Electroenzymatic asymmetric reduction of rac‐3‐methylcyclohexanone to (1S,3S)‐3‐methylcyclohexanol in organic/aqueous media catalyzed by a thermophilic alcohol dehydrogenase. Adv. Synth. Catal. 349, 1337–1340 (2007).
Bui, N.-N., Ho, X.-H., Mho, S.-i. & Jang, H.-Y. Organocatalyzed α-oxyamination of aldehydes using anodic oxidation. Eur. J. Org. Chem. 5309–5312 (2009)
Ho, X.-H., Mho, S.-i., Kang, H. & Jang, H.-Y. Electro-organocatalysis: enantioselective α-alkylation of aldehydes. Eur. J. Org. Chem. 4436–4441 (2010)
DeMartino, M. P., Chen, K. & Baran, P. S. Intermolecular enolate heterocoupling: scope, mechanism, and application. J. Am. Chem. Soc. 130, 11546–11560 (2008).
Tang, S., Liu, Y. & Lei, A. Electrochemical oxidative cross-coupling with hydrogen evolution: a green and sustainable way for bond formation. Chem 4, 27–45 (2018).
Zhu, L. et al. Catalytic asymmetric oxidative enamine transformations. ACS Catal. 8, 5466–5484 (2018).
Næsborg, L. et al. Direct enantio‐ and diastereoselective oxidative homo‐coupling of aldehydes. Chem. Eur. J. 24, 14844–14848 (2018).
Jang, H.-Y., Hong, J.-B. & MacMillan, D. W. C. Enantioselective organocatalytic singly occupied molecular orbital activation: the enantioselective α-enolation of aldehydes. J. Am. Chem. Soc. 129, 7004–7005 (2007).
Tisovský, P., Mečiarová, M. & Šebesta, R. Asymmetric organocatalytic SOMO reactions of enol silanes and silyl ketene (thio)acetals. Org. Biomol. Chem. 12, 9446–9452 (2014).
Zhang, L. & Meggers, E. Steering asymmetric Lewis acid catalysis exclusively with octahedral metal-centered chirality. Acc. Chem. Res. 50, 320–330 (2017).
Horn, E. J. et al. Scalable and sustainable electrochemical allylic C–H oxidation. Nature 533, 77–81 (2016).
Badalyan, A. & Stahl, S. S. Cooperative electrocatalytic alcohol oxidation with electron–proton-transfer mediators. Nature 535, 406–410 (2016).
Fu, N., Sauer, G. S., Saha, A., Loo, A. & Lin, S. Metal-catalyzed electrochemical diazidation of alkenes. Science 357, 575–579 (2017).
Möckel, R. et al. Electrochemical synthesis of aryl iodides by anodic iododesilylation. Angew. Chem. Int. Ed. 57, 442–445 (2017).
Gieshoff, T., Kehl, A., Schollmeyer, D., Moeller, K. D. & Waldvogel, S. R. Insights into the mechanism of anodic N–N bond formation by dehydrogenative coupling. J. Am. Chem. Soc. 139, 12317–12324 (2017).
Xiong, P., Xu, H.-H., Song, J. & Xu, H.-C. Electrochemical difluoromethylarylation of alkynes. J. Am. Chem. Soc. 140, 2460–2464 (2018).
Yang, Q.-L. et al. Copper-catalyzed electrochemical C–H amination of arenes with secondary amines. J. Am. Chem. Soc. 140, 11487–11494 (2018).
Huo, H. et al. Asymmetric photoredox transition-metal catalysis activated by visible light. Nature 515, 100–103 (2014).
Yan, M., Kawamata, Y. & Baran, P. S. Synthetic organic electrochemistry: calling all engineers. Angew. Chem. Int. Ed. 57, 4149–4155 (2017).
Ma, J., Zhang, X., Huang, X., Luo, S. & Meggers, E. Preparation of chiral-at-metal catalysts and their use in asymmetric photoredox chemistry. Nat. Protoc. 13, 605–632 (2018).
König, B. Chemical Photocatalysis (De Gruyter, Berlin 2013).
Kärkäs, M. D., Porco, J. A. & Stephenson, C. R. J. Photochemical approaches to complex chemotypes: applications in natural product synthesis. Chem. Rev. 116, 9683–9747 (2016).
Romero, N. A. & Nicewicz, D. A. Organic photoredox catalysis. Chem. Rev. 116, 10075–10166 (2016).
Twilton, J. et al. The merger of transition metal and photocatalysis. Nat. Rev. Chem. 1, 0052 (2017).
Tan, Y., Yuan, W., Gong, L. & Meggers, E. Aerobic asymmetric dehydrogenative cross-coupling between two –H groups catalyzed by a chiral-at-metal rhodium complex. Angew. Chem. Int. Ed. 54, 13045–13048 (2015).
Evans, D. A., Fandrick, K. R., Song, H.-J., Scheidt, K. A. & Xu, R. Enantioselective Friedel-Crafts alkylations catalyzed by bis(oxazolinyl)pyridine-scandium(III) triflate complexes. J. Am. Chem. Soc. 129, 10029–10041 (2007).
Fu, N., Sauer, G. S. & Lin, S. A general, electrocatalytic approach to the synthesis of vicinal diamines. Nat. Protoc. 13, 1725–1743 (2018).
Acknowledgements
We are grateful for funding from the Deutsche Forschungsgemeinschaft (grant no. ME 1805/13-1).
Author information
Authors and Affiliations
Contributions
E.M. coordinated the project. E.M. and X.H. conceived the project, designed the experiments and wrote the manuscript. X.H. carried out the majority of the synthetic experiments. Q.Z. and J.L. synthesized and characterized Rh2. K.H. collected the crystallographic data, and solved and refined the X-ray crystal structures.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Methods, Supplementary Figures 1–41, Supplementary References
Compound Λ-Rh2
Crystallographic Data for Compound Λ-Rh2
Compound 3g
Crystallographic Data for Compound 3g
Compound 6n
Crystallographic Data for Compound 6n
Compound Rh2-1a
Crystallographic Data for Compound Rh2-1a
Rights and permissions
About this article
Cite this article
Huang, X., Zhang, Q., Lin, J. et al. Electricity-driven asymmetric Lewis acid catalysis. Nat Catal 2, 34–40 (2019). https://doi.org/10.1038/s41929-018-0198-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-018-0198-y
- Springer Nature Limited
This article is cited by
-
Enantioselective nickel-catalyzed anodic oxidative dienylation and allylation reactions
Nature Communications (2024)
-
Critical learning from industrial catalysis for nanocatalytic medicine
Nature Communications (2024)
-
Confluence of asymmetric catalysis and electrosynthesis in sustainable chemical transformations
Science China Chemistry (2024)
-
Radical approach for enantioselective amination
Nature Synthesis (2023)
-
Cycloaddition with asymmetric photoelectrocatalysis
Nature Catalysis (2023)