Abstract
There is still a paucity of research on the relationship between triglyceride-glucose-body mass index (TyG-BMI) and long-term all-cause and cardiovascular disease (CVD) mortality in patients with chronic kidney disease (CKD). The objective of this study was to explore the relationship between the TyG-BMI index and mortality rate and to determine valuable predictive factors for the survival status of this population. Data were obtained from the National Health and Nutrition Examination Survey (NHANES 2001–2018) and the National Death Index (NDI). We used multivariate Cox regression and restricted cubic spline (RCS) to analyze the link between the TyG-BMI index and all-cause and CVD mortality. Subgroup analysis was conducted according to age, gender, race, education and poverty. In addition, receiver operating characteristic (ROC) curves were utilized to assess the differentiation of the TyG-BMI index in predicting mortality. A total of 3089 individuals were enrolled. Over a median follow-up period of 81 months, 1097 individuals passed away. The RCS analysis revealed a U-shaped link between the TyG-BMI index and all-cause and CVD mortality. The ROC curve indicated that the TyG-BMI index has a stronger diagnostic effect than the TyG index. Subgroup analysis results demonstrated that the TyG-BMI index was more significantly correlated with all-cause and CVD mortality rates in elderly patients. In the American population, a U-shaped association was discovered between the baseline TyG-BMI index and all-cause and cardiovascular mortality rates in CKD patients. The thresholds for all-cause and CVD mortality were found to be 299.31 and 294.85, respectively.
Similar content being viewed by others
Introduction
As a serious health issue, chronic kidney disease (CKD) affects 15–20% of the global population. This condition poses a substantial threat to public health, presenting a formidable challenge for both the international community and healthcare systems worldwide1. Due to its irreversible progression, the burden of CKD rapidly increases, with CKD having the highest disability and mortality rates among chronic diseases2. Identifying and intervening early on hazard factors that influence prognosis is essential for alleviating the global burden of cardiovascular disease (CVD) among CKD patients.
Insulin resistance (IR) is a key characteristic of metabolic syndrome, marked by a decreased ability of insulin to effectively promote glucose utilization3. IR is acknowledged as a hazard factor contributing to the development of macrovascular lesions4. Although the high insulin normal glucose clamp test is considered the gold standard for measuring insulin resistance (IR), it is a invasive procedure, making it unsuitable for clinical research5. A practical alternative evaluation metric is the Homeostatic Model Assessment of Insulin Resistance (HOMA-IR), which is derived from measurements of fasting blood glucose and insulin levels6. However, circulating insulin concentration is not conventionally measured in primary healthcare settings, prompting the development of various simple and feasible alternative indicators for evaluating insulin resistance (IR). Notable among these are the triglyceride glucose index (TyG-index) and the triglyceride glucose body mass index (TyG-BMI index)7,8. Several studies have displayed an link between the TyG index and the onset and progression of chronic kidney disease9,10. However, few studies have assessed the link between TyG-BMI and all-cause and cardiovascular mortality in patients with chronic kidney disease. Currently, there remains a lack of evidence to support the TyG-BMI index as a long-term predictor of all-cause mortality risk in patients with CKD.
This study utilized a large sample of American adults to probe into the link between the TyG-BMI index and long-term all-cause mortality, as well as CVD mortality, in patients with chronic kidney disease (CKD). The goal is to identify valuable predictive factors for the survival status of this population.
Methods
Study design
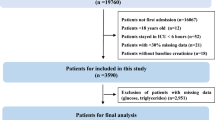
The data for this study are sourced from the NHANES database, which is designed to evaluate the health and nutritional status of Americans across diverse demographics. The NHANES protocol has obtained informed written consent from all participants in the study. This cross-sectional study included 91,351 adult participants from NHANES (2001–2018). The study process is shown in Fig. 1. The exclusion criteria were: (1) Missing data on diagnosis of CKD; (2) participants with triglyceride glucose index deficiency; (3) participants lacking outcome or covariates.
Diagnosis of chronic kidney disease
The diagnosis of chronic kidney disease (CKD) is established based on international guidelines. According to these guidelines, CKD is defined by an estimated glomerular filtration rate (eGFR) of less than 60 mL/min/1.73 m2, or proteinuria of at least 30 mg/g, or both11. The glomerular filtration rate is calculated using the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation, which estimates GFR based on serum creatinine as a marker of renal function12. The classification of CKD stages follows international guidelines as well: CKD Stage I: eGFR ≥ 90 mL/min/1.73 m2, CKD Stage II: 60 ≤ eGFR < 90 mL/min/1.73 m2, CKD Stage III: 30 ≤ eGFR < 60 mL/min/1.73 m2, CKD Stage IV: 15 ≤ eGFR < 30 mL/min/1.73 m2, CKD Stage V: eGFR < 15 mL/min/1.73 m213.
Exposure variables and outcomes
The TyG-BMI index is calculated by multiplying BMI with the TyG index, where BMI is defined as weight divided by height squared (weight/height2), and the TyG index is computed as Ln [fasting triglycerides (mg/dL) × fasting blood glucose (mg/dL)/2]. Triglycerides and fasting blood glucose concentrations were measured using enzymatic methods on Roche Modular P and Roche Cobas 6000 biochemical analyzers. The primary endpoint of this study is all-cause mortality, while the secondary endpoint focuses on cardiovascular mortality. To track these outcomes, subjects were followed until December 31, 2019. The mortality status of each participant was determined by linking their records to the National Death Index, ensuring accurate and comprehensive data on the causes and timing of deaths.
Covariates
Information on various demographic and health-related factors, including age, gender, race/ethnicity, education level, household income, smoking status, disease status, and biochemical parameters, were collected from NHANES family interviews. The specific classification is shown in Table 1.
Statistical analysis
Given the complex sampling design of NHANES, the analysis incorporated minimum sample weights. Participants were categorized into two groups based on their survival status to depict the characteristics of the study population. Continuous variables are represented by weighted means and standard error (SE), with hypothesis testing conducted using either the t-test or the Kruskal–Wallis rank sum test, depending on the distribution of the data. Categorical variables are represented by weighted proportions, and their differences between groups were tested using the chi-square test. The multivariate Cox proportional hazards model was used to evaluate the relationship between the TyG-BMI index and survival status. The dependent variables were survival time and survival status. We constructed three regression models by adjusting for different covariates. In Model 1, no adjustments were made. Model 2 was adjusted for age, gender, race, education level, poverty status (PIR), and smoking status. Model 3 is a comprehensive adjustment model, which further adjusted for hypertension, diabetes, cardiovascular disease, blood lipids, uACR, and eGFR levels based on Model 2. We calculated the hazard ratio (HR) for each covariate and its 95% confidence interval to quantify the strength of the association between the TyG-BMI index and mortality. Restricted Cubic Splines (RCS) analysis, a flexible nonlinear regression method, was employed to explore the nonlinear relationship between continuous variables and outcome variables. We selected three nodes (5th, 50th, and 95th percentiles) and constructed an RCS function to capture the nonlinear effects of the TyG-BMI index. The model with the RCS term was compared to the linear model using a likelihood ratio test to determine the significance of the nonlinear relationship. An RCS curve was drawn to illustrate the relationship between the TyG-BMI index and mortality rate. Receiver operating characteristic (ROC) curves were applied to assess the recognition ability and accuracy of the TyG and TyG-BMI indices. Furthermore, stratified analyses were conducted based on age, gender, race, education and poverty. Data analysis was performed applying R software (version 4.2.1), and a P-value of less than 0.05 was considered statistically significant.
Results
Participant characteristics
A total of 3089 participants were included. The baseline characteristics revealed that participants who died during the follow-up period tended to be older, single, and had lower education levels and incomes. They were also more likely to be smokers and patients with diabetes, hypertension, and heart disease. Detailed baseline characteristics are presented in Table 1.
Association between TyG-BMI index and survival status
To evaluate the correlation between the TyG-BMI index and mortality rates, three models were developed. Participants were grouped into quartiles (Q1–Q4) based on the TyG-BMI index. In a fully adjusted model, the highest quartile group was significantly associated with lower all-cause mortality (HR 0.74; 95% CI 0.57–0.93) compared to the lowest quartile group (Tables 2, 3). RCS regression analysis was conducted to elucidate the specific relationship between the TyG-BMI index and mortality risk in CKD patients. The RCS results indicated a U-shaped link between TyG-BMI and the risk of all-cause and CVD mortality, with inflection points at 299.31 and 294.85, respectively. Both high and low TyG-BMI values increased the risk of all-cause and cardiovascular mortality in this population (Fig. 2). In addition, we divided CKD stages into two groups: stages I–II and stages III–V. Our analysis indicates that there remains a U-shaped relationship between the TyG BMI index and both all-cause mortality and cardiovascular mortality in CKD patients across these different groupings (Fig. 3).
Subgroup analysis results
In subgroup analyses, the association between the TyG-BMI index and all-cause and cardiovascular mortality was consistent across subgroups stratified by sex, race, education and poverty. However, the correlation was more significant in elderly CKD patients for both all-cause and CVD mortality (Table 4).
Sensitivity and specificity analysis
The receiver operating characteristic (ROC) curve (Fig. 4) was utilized to access the sensitivity and specificity of the TyG-BMI index as a prognostic diagnostic tool. Compared to the TyG index alone, the TyG-BMI index demonstrated higher predictive ability for all-cause and cardiovascular mortality, with values of 0.578 and 0.550, respectively. In addition, we determined the optimal cutoff point for TyG-BMI using the Youden index. As shown in Table 5, the optimal cutoff values for TyG-BMI in predicting all-cause mortality and cardiovascular disease (CVD) mortality in patients with chronic kidney disease are 272.57 and 280.34, respectively.
Discussion
This study aims to reveal how the combination of triglyceride-glucose index and body mass index (TyG-BMI) affects the mortality outcomes of this specific population of patients with chronic kidney disease. By employing multivariate Cox regression and RCS analysis, we identified the TyG-BMI index as a reliable predictor of both all-cause and CVD mortality risk in CKD patients. The analysis revealed a U-shaped association between the TyG-BMI index and mortality risk, indicating that both high and low TyG-BMI values are linked to an increased risk of mortality in this population. In addition, the results of subgroup analysis indicated that the association between the TyG-BMI index and all-cause mortality and cardiovascular mortality was consistent across subgroups defined by gender, race/ethnicity, poverty rate, and education level (P for interaction > 0.05). However, subgroup analysis stratified by age revealed that in elderly CKD patients, the correlation between the TyG-BMI index and both all-cause mortality and CVD mortality was more significant. This finding suggests that the TyG-BMI index has broad applicability as a predictor of all-cause mortality and CVD mortality. The TyG-BMI index can serve as an effective risk assessment tool regardless of gender, race, economic status, or education level. This discovery holds significant implications for public health policy and clinical practice, as it indicates that the TyG-BMI index can be widely used for risk assessment among CKD patients in diverse populations. However, the stronger correlation between the TyG-BMI index and mortality in elderly CKD patients suggests that special attention should be given to changes in the TyG-BMI index within this specific population. As age increases, the decline in metabolic function and the associated rise in chronic disease risk may render elderly patients more sensitive to changes in the TyG-BMI index. Therefore, in clinical management, elderly CKD patients may require more frequent and careful monitoring and intervention.
Insulin resistance (IR) is broadly acknowledged for its significant role in the development of type 2 diabetes, dyslipidemia, and obesity within the general population14,15. The TyG-BMI index integrates triglycerides (TG), fasting blood glucose, and BMI, making it a simple and feasible IR evaluation tool16. This comprehensive indicator is considered a more accurate IR marker than a single index. Many studies support that the TyG-BMI index is closely related to non-alcoholic fatty liver disease (NAFLD), cardiovascular events, prehypertension, and diabetes17,18,19,20. These pieces of evidence suggest that the TyG-BMI index may become an important prognostic indicator. In our study, the TyG-BMI index exhibited comparable predictive value for both all-cause and cardiovascular mortality in patients with CKD. Specifically, a U-shaped relationship between the baseline TyG-BMI index and all-cause mortality was identified. This indicates that both low and high values of the TyG-BMI index are associated with increased mortality risk in patients with chronic kidney disease (CKD), suggesting that an optimal range of the TyG-BMI index may be crucial for reducing mortality risk in this population. This U-shaped association between insulin resistance indicators and poor prognosis has been confirmed in previous studies using NHANES data. The mechanism behind this U-shaped association is still uncertain, but it may involve several factors.
On the one hand, elevated TyG-BMI is linked to higher insulin resistance and metabolic disorders, such as increased levels of triglycerides and glucose, which are known risk factors for cardiovascular diseases (CVD)21,22. Patients with high TyG-BMI often exhibit increased levels of LDL cholesterol, lower HDL cholesterol, and higher levels of inflammatory markers like hs-CRP, which contribute to the development and progression of atherosclerosis and subsequent cardiovascular events23,24. Additionally, high TyG-BMI is associated with conditions like hypertension and diabetes, which further elevate the risk of cardiovascular mortality25. On the other hand, low TyG-BMI may be indicative of malnutrition, sarcopenia (loss of muscle mass), and frailty, particularly in CKD patients26,27. These conditions can weaken the body’s overall resilience and immune response, making individuals more susceptible to infections, cachexia, and other complications that increase the risk of all-cause mortality. Malnutrition and low muscle mass are critical concerns in CKD patients, as they are linked to poor clinical outcomes and higher mortality rates28,29. In summary, the U-shaped relationship between TyG-BMI and mortality risks highlights the complex interplay of metabolic health, nutritional status, and cardiovascular risk factors. High TyG-BMI exacerbates cardiovascular risks through metabolic disturbances and inflammation, while low TyG-BMI reflects poor nutritional status and physical frailty, both leading to increased mortality in CKD patients.
Our research offers several significant advantages. First, the complex sampling design effectively adjusts the representativeness of different populations, ensuring the wide applicability and reliability of the research results. In addition, TyG-related indicators based on routine clinical care are used to evaluate insulin resistance, making them easily accessible and cost-effective biomarkers with broad clinical application prospects. This method is both economical and efficient, offering convenient and powerful tools for health management and disease prevention in clinical practice. However, our research has several notable limitations. The cross-sectional study design restricts our ability to establish a causal relationship between insulin resistance and CKD. Furthermore, like all observational studies, despite adjusting for potential confounders in multivariate models, residual confounders from unmeasured or uncontrolled factors may still exist. For example, due to database limitations, we are unable to include Medication use as a covariate in Cox regression analysis. Future research should consider using databases containing detailed drug use information or conducting prospective study designs to better control for confounding effects of drug use. Lastly, due to data limitations in the NHANES database, we are unable to investigate the impact of different etiologies on the correlation between TyG-BMI and mortality. Future research should consider using databases that contain detailed etiological information or designing prospective studies for more granular subgroup analysis.
Conclusion
The TyG-BMI index may serve as a valuable predictive marker for assessing the risk of all-cause and cardiovascular mortality in patients with chronic kidney disease. The association between this index and mortality rate is non-linear, exhibiting a U-shaped pattern.
Data availability
Data is provided within the manuscript or supplementary information files.
Abbreviations
- TyG-BMI:
-
Triglyceride-glucose-body mass
- CKD:
-
Chronic kidney disease
- NDI:
-
National death index
- uACR:
-
Urinary albumin to creatinine ratio
- eGFR:
-
Glomerular filtration rate
- TC:
-
Cholesterol
- IR:
-
Insulin resistance
References
Matsushita, K. et al. Epidemiology and risk of cardiovascular disease in populations with chronic kidney disease. Nat. Rev. Nephrol. 18(11), 696–707 (2022).
Romagnani, P. et al. Chronic kidney disease. Nat. Rev. Dis. Primers 3, 17088 (2017).
Zhang, Y. et al. High triglyceride-glucose index is associated with adverse cardiovascular outcomes in patients with acute myocardial infarction. Nutr. Metab. Cardiovasc. Dis. 30(12), 2351–2362 (2020).
Brownlee, M. The pathobiology of diabetic complications: A unifying mechanism. Diabetes 54(6), 1615–1625 (2005).
Tao, L. C., Xu, J. N., Wang, T. T., Hua, F. & Li, J. J. Triglyceride-glucose index as a marker in cardiovascular diseases: Landscape and limitations. Cardiovasc. Diabetol. 21(1), 68 (2022).
Mirjalili, S. R. et al. An innovative model for predicting coronary heart disease using triglyceride-glucose index: A machine learning-based cohort study. Cardiovasc. Diabetol. 22(1), 200 (2023).
Alizargar, J., Bai, C. H., Hsieh, N. C. & Wu, S. V. Use of the triglyceride-glucose index (TyG) in cardiovascular disease patients. Cardiovasc. Diabetol. 19(1), 8 (2020).
Wang, M. et al. Application value of triglyceride-glucose index and triglyceride-glucose body mass index in evaluating the degree of hepatic steatosis in non-alcoholic fatty liver disease. Lipids Health Dis. 22(1), 186 (2023).
Ren, X., Jiang, M., Han, L. & Zheng, X. Association between triglyceride-glucose index and chronic kidney disease: A cohort study and meta-analysis. Nutr. Metab. Cardiovasc. Dis. 33(6), 1121–1128 (2023).
Ye, Z. et al. Association between the triglyceride glucose index and in-hospital and 1-year mortality in patients with chronic kidney disease and coronary artery disease in the intensive care unit. Cardiovasc. Diabetol. 22(1), 110 (2023).
Rovin, B. H. et al. KDIGO 2021 clinical practice guideline for the management of glomerular diseases. Kidney Int. 100(4s), S1–S276 (2021).
Levey, A. S. et al. A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 150(9), 604–612 (2009).
Webster, A. C., Nagler, E. V., Morton, R. L. & Masson, P. Chronic kidney disease. Lancet 389(10075), 1238–1252 (2017).
Santoro, A. & Kahn, B. B. Adipocyte regulation of insulin sensitivity and the risk of type 2 diabetes. N. Engl. J. Med. 388(22), 2071–2085 (2023).
Rohm, T. V., Meier, D. T., Olefsky, J. M. & Donath, M. Y. Inflammation in obesity, diabetes, and related disorders. Immunity 55(1), 31–55 (2022).
Lim, J., Kim, J., Koo, S. H. & Kwon, G. C. Comparison of triglyceride glucose index, and related parameters to predict insulin resistance in Korean adults: An analysis of the 2007–2010 Korean National Health and Nutrition Examination Survey. PLoS ONE 14(3), e0212963 (2019).
Wang, J. et al. The diagnostic and prognostic value of the triglyceride-glucose index in metabolic dysfunction-associated fatty liver disease (MAFLD): A systematic review and meta-analysis. Nutrients 14, 23 (2022).
Dou, J. et al. Association between triglyceride glucose-body mass and one-year all-cause mortality of patients with heart failure: A retrospective study utilizing the MIMIC-IV database. Cardiovasc. Diabetol. 22(1), 309 (2023).
Zeng, Z. Y. et al. Association of triglyceride glucose index and its combination of obesity indices with prehypertension in lean individuals: A cross-sectional study of Chinese adults. J. Clin. Hypertens. 22(6), 1025–1032 (2020).
Ding, L. et al. The impact of triglyceride glucose-body mass index on all-cause and cardiovascular mortality in elderly patients with diabetes mellitus: Evidence from NHANES 2007–2016. BMC Geriatr. 24(1), 356 (2024).
Kang, S. W., Kim, S. K., Kim, Y. S. & Park, M. S. Risk prediction of the metabolic syndrome using TyG Index and SNPs: A 10-year longitudinal prospective cohort study. Mol. Cell. Biochem. 478(1), 39–45 (2023).
Dang, K. et al. The association between triglyceride-glucose index and its combination with obesity indicators and cardiovascular disease: NHANES 2003–2018. Cardiovasc. Diabetol. 23(1), 8 (2024).
Mahdavi-Roshan, M. et al. Evaluating the use of novel atherogenicity indices and insulin resistance surrogate markers in predicting the risk of coronary artery disease: A case–control investigation with comparison to traditional biomarkers. Lipids Health Dis. 21(1), 126 (2022).
Li, F. et al. Association between the cumulative average triglyceride glucose-body mass index and cardiovascular disease incidence among the middle-aged and older population: A prospective nationwide cohort study in China. Cardiovasc. Diabetol. 23(1), 16 (2024).
Hou, X. Z. et al. Association between different insulin resistance surrogates and all-cause mortality in patients with coronary heart disease and hypertension: NHANES longitudinal cohort study. Cardiovasc. Diabetol. 23(1), 86 (2024).
Cheong, C. Y., Yap, P., Yap, K. B. & Ng, T. P. Associations of inflammatory, metabolic, malnutrition, and frailty indexes with multimorbidity incidence and progression, and mortality impact: Singapore longitudinal aging study. Gerontology 69(4), 416–427 (2023).
Jiang, X. et al. Association of triglyceride-glucose index with the prevalence of cardiovascular disease in malnourished/non-malnourished patients: A large cross-sectional study. Front. Cardiovasc. Med. 10, 1306415 (2023).
Zha, Y. & Qian, Q. Protein nutrition and malnutrition in CKD and ESRD. Nutrients 9, 3 (2017).
Lu, Y. et al. Malnutrition risk and kidney function and decline in community-dwelling older adults. J. Renal Nutr. 32(5), 560–568 (2022).
Acknowledgements
The authors express sincere gratitude to the individual who generously shared the original dataset for this study.
Funding
This work was supported by Jiangxi Provincial Natural Science Foundation (20232BAB216020 and 20232BAB206104).
Author information
Authors and Affiliations
Contributions
TC, CLY and HW wrote the main manuscript text and LYX prepared Figs. 1, 2 and 3. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The NCHS Ethics Review Committee has approved NHANES.
Consent to participate
The patient/participant provided written informed consent to participate in this study.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, T., Wan, H., Luo, Y. et al. Association of triglyceride-glucose-body mass index with all-cause and cardiovascular mortality among individuals with chronic kidney disease. Sci Rep 14, 20593 (2024). https://doi.org/10.1038/s41598-024-71579-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-71579-w
- Springer Nature Limited