Abstract
This study aimed to assess the efficacy of dual T-cell suppression using individually tailored doses of antithymocyte globulin (ATG) and attenuated dose of post-transplant cyclophosphamide (PTCy) in haploidentical hematopoietic stem cell transplantation (haplo-HSCT). We conducted a retrospective analysis of 78 adults with acute leukemia or myelodysplastic syndrome who underwent haplo-HSCT using intravenous busulfan and fludarabine conditioning. Thirty-two patients received attenuated ATG/PTCy, while 46 patients received ATG (7.5 mg/kg) as GVHD prophylaxis. The 100-day cumulative incidence of grade III-IV (9.7% vs. 32.4%, P = 0.018) acute GVHD, as well as 2-year moderate-severe chronic GVHD (13.9% vs. 43.9%, P = 0.018) in the ATG/PTCy group were significantly lower than those in the ATG group. The 2-year overall survival was comparable between the two groups. However, 2-year GVHD-free, relapse-free survival in the ATG/PTCy group was significantly higher compared to that in the ATG group (38.9% vs. 21.7%, P = 0.021). Moreover, during post-engraftment period, the ATG/PTCy group exhibited lower incidences of life-threatening bacterial (12.5% vs. 37%, P = 0.033) and viral infection (0% vs. 17.4%, P = 0.035) than the ATG group. In conclusion, the combination of individually tailored ATG and low-dose PTCy appears to be a promising strategy in haplo-HSCT.
Similar content being viewed by others
Introduction
Allogeneic hematopoietic stem cell transplantation provides a chance for cure in patients with hematologic malignancies, and familial haplo-identical hematopoietic stem cell transplantation (haplo-HSCT) is increasingly being performed in light of the donor’s immediate availability1. However, due to HLA mismatches2,3, graft-versus-host disease (GVHD) remains a significant challenge, and there is no consensus on the optimal GVHD prophylaxis regimen for haplo-HSCT.
Antithymocyte globulin (ATG) has been widely used in haplo-HSCT for GVHD prevention, offering advantages such as reduced rates of graft rejection and relapse4,5,6,7. However, the acute GVHD (aGVHD) rate did not drop below 40%, and the benefits are compromised by the high risk of infections, especially viral infections including cytomegalovirus (CMV) reactivation, due to delayed immune reconstitution5,8,9,10,11. On the other hand, Luznik et al. reported that high-dose post-transplant cyclophosphamide (PTCy) effectively reduces GVHD rates in haplo-HSCT12, with subsequent reports confirming its efficacy in allogeneic HSCT with mismatched donor13,14. However, challenges arise with high-dose PTCy, including excessive cytotoxicity leading to a risk of acute cardiotoxicity and potential attenuation of the graft-versus-leukemia effect15,16. Therefore, attempts have been made to address these issues by exploring the efficacy of low-dose PTCy17.
Considering the demand for optimal GVHD prophylaxis in haplo-HSCT, efforts have been made to combine low-dose PTCy and ATG to mitigate their respective disadvantages. This study aims to report on the efficacy of dual T-cell suppression using individually tailored ATG and attenuated doses of PTCy, comparing it to ATG alone as GVHD prophylaxis in haplo-HSCT for patients with acute leukemia and myelodysplastic syndrome (MDS).
Patients and methods
Participants and study design
This is a retrospective study of patients with acute leukemia and MDS who were over 18 years old and underwent haplo-HSCT following identical busulfan and fludarabine (BuFlu) conditioning between June 2017 and 2023 at Seoul National University Hospital, Seoul, South Korea. All patients received either ATG (ATG group) or a combination of ATG and PTCy (ATG/PTCy group) as T-cell depletion for GVHD prophylaxis. In the consideration of the choice between ATG and ATG/PTCy, some of the staff physicians implemented ATG/PTCy for research purposes, while the remainder adhered to the conventional institutional practice of applying ATG. Haploidentical donors were considered in cases where a matched donor was unavailable or when HSCT was urgently needed. In all patients, peripheral blood stem cells (PBSCs) were used as a graft source.
Transplantation procedures
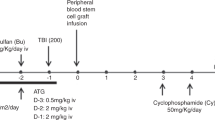
For the ATG group, busulfan 3.2 mg/kg and fludarabine 40 mg/m2 on days -6 through -3 were used as myeloablative conditioning (MAC), and busulfan 3.2 mg/kg on days -7 and -6 with fludarabine 30 mg/m2 on days -7 through -2 were used as reduced-intensity conditioning (RIC). ATG (rabbit, thymoglobulin; Sanofi-Aventis, Korea) 2.5 mg/kg on days -3 through -1, for a total of 7.5 mg/kg was administrated as GVHD prophylaxis. For the ATG/PTCy group, the MAC regimen was the same as in ATG group, while for the RIC, busulfan 3.2 mg/kg on days -6 and -5 with fludarabine 40 mg/m2 on days -6 through -3 were employed. ATG was given from days -3 through -1 and was adjusted per absolute lymphocyte count (ALC) on days -3 based on our previous study18: for ALC > 1,000/μl patients, total of 4.0 mg/kg was used; for ALC 500–1,000/μl, total of 3.5 mg/kg; and for ALC < 500/μl, total of 3.0 mg/kg was used. PTCy was administered at doses of 50 mg/kg on day 3 and 30 mg/kg on day 4 (Supplementary Fig. 1). In addition, calcineurin inhibitors were used to prevent GVHD. The choice of calcineurin inhibitors, either cyclosporine or tacrolimus, was left to the attending physician’s preference. Methotrexate (15 mg/m2 intravenous push on day 1 and 10 mg/m2 on day 3 and day 6) was considered in ATG group, and mycophenolate mofetil (15 mg/kg three times a day from days 5 through 30) was administered in ATG/PTCy group.
All patients received uniform prophylactic regimens; ciprofloxacin for gut decontamination, trimethoprim/sulfamethoxazole for Pneumocystis jirovecii, micafungin for fungus, and acyclovir for herpes simplex virus. The serum CMV polymerase chain reaction test was conducted twice weekly, and preemptive ganciclovir treatment was initiated in cases of CMV reactivation.
Definitions and outcomes
The diagnosis of acute leukemia and MDS was made according to the revised 2016 WHO classification. The modified European Group for Blood and Marrow Transplantation (mEBMT) risk score was used to assess risks of HSCT19. The 2016 MAGIC criteria were used to grade aGVHD20. Chronic GVHD (cGVHD) was classified as mild, moderate, or severe according to the 2014 National Institutes of Health consensus criteria21. Neutrophil engraftment was defined as absolute neutrophil count (ANC) > 500/μl for 3 consecutive measurements. Platelet engraftment was defined as 3 consecutive measurements of > 20,000/μl without transfusion. Non-relapse mortality (NRM) was defined as death without progression of underlying disease. Relapse-free survival (RFS) was defined as the time from stem cell infusion to relapse or death from any cause. Overall survival (OS) was defined as the time from stem cell infusion to death of any cause. GVHD-free, relapse-free survival (GRFS) was defined according to Ruggeri et al. (ie, being alive with no grade 3–4 aGVHD, severe cGVHD, or relapse at any time point after HSCT). The cause of death was evaluated as primary disease, graft failure, GVHD, infection, and other based on a previous report22. Any bacterial, viral, and fungal Infection data were collected retrospectively until the patient’s death or last follow-up, using standardized definitions of severe infections after HSCT23,24. Infections requiring intensive care or vasopressor support were considered life-threatening infections.
Statistical analysis
Differences between groups were assessed using a Student’s t-test or Wilcoxon rank-sum test for continuous variables, and chi-square test for categorical variables, as indicated. Median follow-up duration was calculated by the reverse Kaplan–Meier method. RFS, OS and GRFS were estimated by the Kaplan–Meier method and log-rank test was used for univariate comparisons of survival. Cumulative incidence curves were used in competing risk setting to calculate the probability of GVHD, relapse incidence and NRM. Univariate and multivariate Cox proportional hazards regression models were used to analyze GVHD and survival outcomes with type of GVHD prophylaxis, age, diagnosis, mEBMT score, conditioning intensity, amount of infused CD34 + cell, and HSCT timing. Multivariate analysis was performed using stepwise backward selection, and adjusted hazard ratios (HRs) with 95% confidence intervals (CIs) were calculated. All tests were 2-sided, and P values of < 0.05 were considered statistically significant. For all parts of statistical analyses, the statistical software ‘R’ version 4.3.1 (www.r-project.org) were used.
Ethics declarations
This study was conducted according to Declaration of Helsinki and was approved by the Institutional Review Board of Seoul National University Hospital (IRB no. H-2207–083-1339). The requirement for informed consent was waived by the Institutional Review Board of Seoul National University Hospital due to the retrospective nature of the study.
Results
Patient and disease characteristics
A total of 78 patients were included in this study; 32 belonged to the ATG/PTCy group, while 46 were in the ATG group. Table 1 presents the baseline characteristics of the two groups. There were no differences in baseline characteristics, including disease status and infused CD34 cell dose, between the two groups.
Transplantation outcomes
Neutrophil engraftment (93.8% vs. 95.7%) and platelet engraftment (90.6% vs. 91.3%) did not differ between the ATG/PTCy and ATG groups (Table 2). Regarding immune reconstitution, the ATG/PTCy group exhibited a trend toward faster recovery: time to neutrophil engraftment (median, 16 vs. 18 days, P = 0.002) and time to platelet engraftment (median, 15 vs. 18 days, P = 0.25). The incidence of both CMV antigenemia and disease was lower in the ATG/PTCy group, but the difference did not show significance (53.1% vs. 67.4%; P = 0.3, and 3.1% vs 19.6%; P = 0.073, respectively). Only two patients in the ATG group were found to have post-transplant lymphoproliferative disease as evidenced by biopsy.
Up to December 2023, all of the patients were followed for a median of 38 months (95% CI, 29.4–44); the median follow-up time for ATG/PTCy and ATG groups were 29.4 and 43.3 months, respectively. During follow-up, 37 patients (47.4%) relapsed and 47 (60.3%) died. There was no difference between the groups in terms of causes of death.
Graft-versus-host disease
The 100-day cumulative incidence of grade II-IV (22.1% vs. 42.9%, P = 0.046) and grade III-IV aGVHD (9.7% vs. 32.4%, P = 0.018), as well as 2-year moderate-severe cGVHD (13.9% vs. 43.9%, P = 0.018) in the ATG/PTCy group were significantly lower than those in the ATG group (Fig. 1, Table 2). The 2-year cumulative incidence of severe cGVHD in the ATG/PTCy group showed a trend toward decrease compared to that in the ATG group (6.3% vs. 28.4%, P = 0.072).
As presented in Table 3, the ATG/PTCy combination was a significantly protective factor for grade II-IV (HR 0.40; 95% CI, 0.17–0.96; P = 0.041) and grade III-IV (HR 0.21; 95% CI, 0.06–0.75; P = 0.016), as well as moderate-severe cGVHD (HR 0.25; 95% CI, 0.07–0.87; P = 0.029) in multivariate analysis. Patient age of ≤ 60 years was also associated with a lower grade III-IV aGVHD (HR 0.36; 95% CI, 0.14–0.95; P = 0.038).
Survival outcomes
The median OS, RFS, and GRFS in the entire cohort were 18.5, 8.0, and 5.1 months, respectively. The 2-year probability of OS (52.5% vs. 31.3%, P = 0.14) and RFS (40.5% vs. 30.4%, P = 0.18) for ATG/PTCy and ATG groups were comparable (Fig. 2). The 2-year GRFS in the ATG/PTCy group was significantly higher compared to that in the ATG group (38.9% vs. 21.7%, P = 0.021). The univariate and multivariate analysis revealed that the GVHD prophylaxis protocol did not affect OS and RFS (Supplementary Table 1, Table 3). However, for GRFS, the ATG/PTCy protocol was an independent protective factor (HR 0.55; 95% CI, 0.31–0.96; P = 0.034). Additionally, a low mEBMT score (≤ 3) was marginally protective factor for GRFS compared to a high score (> 3) (HR 0.61; 95% CI, 0.34–1.08; P = 0.089).
The 2-year cumulative incidence of relapse and NRM in the entire cohort were 50.6% and 25.8%, respectively. The 2-year cumulative incidence of relapse (46.4% vs. 54.2%, P = 0.29) and NRM (21.9% vs. 29%, P = 0.49) were similar between the ATG/PTCy group and ATG group (Fig. 2).
Infectious complications
Overall, 218 episodes of infections occurred in 66 of 78 patients (84.6%), with a median of 2 events/patient (mean, 2.8; range, 0–8). Detailed data of infectious events are summarized in Supplementary Table 2. 47.7% were of bacterial, 37.6% viral, and 14.7% fungal origin.
In pre-engraftment period (≤ day 30), there was no significant difference in the incidence of infection between the ATG/PTCy and ATG group (34.4% vs. 54.3%, P = 0.131) (Table 4). The 30-day cumulative incidences of severe bacterial, viral, and fungal infections were comparable between the groups (Supplementary Fig. 2). In the ATG/PTCy group, a lower proportion of patients experienced two or more infectious episodes compared to the ATG group (3.1% vs. 21.7%, P = 0.046).
In post-engraftment period (> day 30), the incidence of life-threatening bacterial (12.5% vs. 37%, P = 0.033) and viral infection (0% vs. 17.4%, P = 0.035) was lower in the ATG/PTCy group than the ATG group. The 1-year cumulative incidence of severe and life-threatening viral infection ([38.9% vs. 58.6%, P = 0.045], [0% vs. 12.4%, P = 0.012], respectively) and life-threatening bacterial infection (11.6% vs. 36.1%, P = 0.026) were significantly lower in the ATG/PTCy group compared to the ATG group (Fig. 3).
Discussion
In patients with acute leukemia and MDS undergoing haplo-HSCT using PBSC and BuFlu conditioning, this study demonstrated the efficacy of our novel GVHD prophylaxis protocol (combining low-dose PTCy and ATG adjusted for days -3 ALC levels) in reducing severe GVHD compared to the standard ATG-based regimen. Dual T-cell depletion with individually tailored ATG and attenuated dose of PTCy did not elevate relapse risk and improved GRFS. Additionally, ATG/PTCy resulted in fewer severe infections than ATG.
Due to PTCy’s potential to enhance the effect of ATG in mitigating GVHD17, recent studies have explored various combinations of ATG and PTCy to improve GVHD prevention in haplo-HSCT (Supplementary Table 3). The cumulative incidence of aGVHD with ATG/PTCy in our study was comparable with other reports, indicating 11.5–34.6% for grade II-IV and 5–9% for grade III-IV aGVHD25,26,27,28. These studies consistently suggest that, despite variations in dosing regimens, dual T-cell depletion with ATG/PTCy can be more effective in reducing severe aGVHD than ATG alone. Additionally, we found a significantly lower cumulative incidence of moderate-severe cGVHD for ATG/PTCy. As the mechanisms of GVHD prevention differ between ATG and PTCy29,30, the synergistic effect of the ATG/PTCy combination can be influenced by the dosage and timing of each. Studies with PTCy at a total dose of only 29 mg/kg26 and ATG used post-transplant at a dose of 2.5 mg/kg25 showed no difference in the incidence of moderate-severe cGVHD between the two groups. In contrast, a study using a total PTCy dose of 80 mg/kg (with a total of 7.5 mg/kg of pre-transplant ATG), similar to ours, demonstrated a fewer extensive cGVHD in the ATG/PTCy group27. Furthermore, in a study using ATG 5 mg/kg and PTCy 50 mg/kg, the ATG/PTCy group had less moderate-severe cGVHD31, consistent with our findings. In our study, ATG/PTCy significantly reduced GVHD but did not impact the relapse risk. Instead, we observed a significantly higher GRFS in the ATG/PTCy group, consistent with previous studies25,26. Our result demonstrated that dual T-cell depletion with ATG/PTCy effectively reduces the risk of severe GVHD without increasing the relapse risk, resulting in a favorable GRFS outcome. Further mechanism studies on the dosing of ATG and PTCy are warranted to achieve the optimal synergistic effect of ATG/PTCy protocol.
Excessive exposure to ATG may increase immunosuppressive toxicity, elevating risks of infections and relapse32. This risk could be heightened when used with PTCy, emphasizing the need for a strategy to judiciously reduce the dose of ATG26. Previous studies reported that weight-based dosing of ATG carries the risk of overdose, and the ALC level on the day of ATG administration could optimize ATG dosing18,33. Our ATG/PTCy protocol tailored the dose of ATG individually for each recipient based on the ALC level at day -3; i.e., if the ALC is low, a small amount of ATG is administered. The combination of low-dose ATG and PTCy did not increase CMV infection compared to the standard dose of ATG, consistent with previous studies25,31. Additionally, we found a significant decrease in life-threatening bacterial and viral infection in the ATG/PTCy group, particularly in the post-engraftment period. As ATG is associated with a delay of the immune reconstitution, an attenuated dose of ATG might ameliorate post-engraftment viral infections34. Moreover, the ATG group exhibited more moderate-severe cGHVD, suggesting the potential for prolonged immunosuppressive therapy. These factors could have contributed to a higher incidence of bacterial infections in the ATG group. The reduced risk of life-threatening infections might have been more attributed to individually tailored ATG dosing than to the synergistic effect of ATG and PTCy. Further research is needed on personalized dosing in GVHD prophylaxis with ATG/PTCy for haplo-HSCT.
This study had several limitations. First, this is a retrospective study. Thus, the two groups were not randomized and characteristics between the two groups were not fully comparable, although generally well-balanced. Although ATG/PTCy was identified as an independently favorable factor in the multivariate analyses, there is a possibility of confounding effects due to patient selection and differences between the groups, such as the use of additional immunosuppressants and timing of HSCT. Second, a relatively small sample size may limit the statistical power. Instead, we minimized the confounding factors; diseases included only MDS and acute leukemia, and all patients received an identical conditioning regimen and graft source, and infection prophylaxis. Third, we did not investigate data on immune reconstitution and ATG concentration. For the optimal dose and schedule of ATG/PTCy, further mechanism studies are warranted.
Despite these limitations, our study implemented a novel personalized dosing strategy by combining low-dose PTCy with a reduced dose of ATG tailored to the ALC level on the day of ATG infusion. We compared its outcomes with those of an ATG-based regimen in haplo-HSCT with the same graft source and conditioning regimen. Additionally, we conducted a comparative analysis of infection episodes, identifying differences between the two groups in terms of timing and etiology of infections.
In conclusion, our results demonstrated that dual T-cell depletion with an individually tailored ATG and attenuated dose of PTCy led to a low incidence of both severe aGVHD and cGVHD, improving GRFS compared to the standard-dose ATG regimen. In addition, ATG/PTCy showed a low incidence of severe infections, making it a feasible protocol. Reducing the PTCy dosage slightly and incorporating an individually tailored small amount of ATG appears to be a promising strategy in haplo-HSCT, emphasizing the need for further research to determine the optimal regimen.
Data availability
The datasets generated during the current study are available from the corresponding authors on reasonable request.
References
Passweg, J. et al. Use of haploidentical stem cell transplantation continues to increase: The 2015 European Society for Blood and Marrow Transplant activity survey report. Bone Marrow Transpl. 52, 811–817 (2017).
Beatty, P. G. et al. Marrow transplantation from related donors other than HLA-identical siblings. N. Engl. J. Med. 313, 765–771 (1985).
Anasetti, C. et al. Effect of HLA compatibility on engraftment of bone marrow transplants in patients with leukemia or lymphoma. N. Engl. J. Med. 320, 197–204 (1989).
Lu, D.-P. et al. Conditioning including antithymocyte globulin followed by unmanipulated HLA-mismatched/haploidentical blood and marrow transplantation can achieve comparable outcomes with HLA-identical sibling transplantation. Blood 107, 3065–3073 (2006).
Chang, Y.-J. & Huang, X.-J. Seminars in hematology. 82–89 (Elsevier).
Baron, F. et al. Anti-thymocyte globulin as graft-versus-host disease prevention in the setting of allogeneic peripheral blood stem cell transplantation: a review from the Acute Leukemia Working Party of the European Society for Blood and Marrow Transplantation. Haematologica 102, 224 (2017).
Santoro, N. et al. Unmanipulated haploidentical stem cell transplantation in adults with acute lymphoblastic leukemia: A study on behalf of the Acute Leukemia Working Party of the EBMT. J. Hematol. Oncol. 10, 1–11 (2017).
Huang, X. et al. Haploidentical hematopoietic stem cell transplantation without in vitro T-cell depletion for the treatment of hematological malignancies. Bone Marrow Transpl. 38, 291–297 (2006).
Wang, Y. et al. Long-term follow-up of haploidentical hematopoietic stem cell transplantation without in vitro T cell depletion for the treatment of leukemia: nine years of experience at a single center. Cancer 119, 978–985 (2013).
Kalra, A. et al. Impact of donor and recipient cytomegalovirus serostatus on outcomes of antithymocyte globulin–conditioned hematopoietic cell transplantation. Biol. Blood Marrow Transpl. 22, 1654–1663 (2016).
Chang, Y. J. et al. Optimal dose of rabbit thymoglobulin in conditioning regimens for unmanipulated, haploidentical, hematopoietic stem cell transplantation: Long-term outcomes of a prospective randomized trial. Cancer 123, 2881–2892 (2017).
Luznik, L. et al. HLA-haploidentical bone marrow transplantation for hematologic malignancies using nonmyeloablative conditioning and high-dose, posttransplantation cyclophosphamide. Biol. Blood Marrow Transpl. 14, 641–650 (2008).
Bashey, A. et al. T-cell–replete HLA-haploidentical hematopoietic transplantation for hematologic malignancies using post-transplantation cyclophosphamide results in outcomes equivalent to those of contemporaneous HLA-matched related and unrelated donor transplantation. J. Clin. Oncol. 31, 1310–1316 (2013).
Jorge, A. S. et al. Single antigen–mismatched unrelated hematopoietic stem cell transplantation using high-dose post-transplantation cyclophosphamide is a suitable alternative for patients lacking HLA-matched donors. Biol. Blood Marrow Transpl. 24, 1196–1202 (2018).
Lin, C. J. et al. Cardiomyopathy in patients after posttransplant cyclophosphamide–based hematopoietic cell transplantation. Cancer 123, 1800–1809 (2017).
Ishida, S. et al. The clinical features of fatal cyclophosphamide-induced cardiotoxicity in a conditioning regimen for allogeneic hematopoietic stem cell transplantation (allo-HSCT). Ann. Hematol. 95, 1145–1150 (2016).
Wang, Y. et al. Low-dose post-transplant cyclophosphamide can mitigate GVHD and enhance the G-CSF/ATG induced GVHD protective activity and improve haploidentical transplant outcomes. Oncoimmunology 6, e1356152 (2017).
Woo, G.-U. et al. Preconditioning absolute lymphocyte count and transplantation outcomes in matched related donor allogeneic hematopoietic stem cell transplantation recipients with reduced-intensity conditioning and antithymocyte globulin treatment. Biol. Blood Marrow Transpl. 26, 1855–1860 (2020).
Gratwohl, A. The EBMT risk score. Bone Marrow Transpl. 47, 749–756 (2012).
Harris, A. C. et al. International, multicenter standardization of acute graft-versus-host disease clinical data collection: A report from the Mount Sinai Acute GVHD International Consortium. Biology Blood Marrow Transpl. 22, 4–10 (2016).
Jagasia, M. H. et al. 2015 National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. The 2014 Diagnosis and Staging Working Group report. Biol. Blood Marrow Transpl. 21, 389–401 (2015).
Copelan, E. et al. A scheme for defining cause of death and its application in the T cell depletion trial. Biol. Blood Marrow Transpl. 13, 1469–1476 (2007).
Cordonnier, C. et al. Definitions of infectious diseases and complications after stem cell transplant. Web site of the EBMT (2001).
Esquirol, A. et al. Severe infections and infection-related mortality in a large series of haploidentical hematopoietic stem cell transplantation with post-transplant cyclophosphamide. Bone Marrow Transpl. 56, 2432–2444 (2021).
Zhang, W. et al. Reduced-dose post-transplant cyclophosphamide plus low-dose post-transplant anti-thymocyte globulin as graft-versus-host disease prophylaxis with fludarabine–busulfan–cytarabine conditioning in haploidentical peripheral blood stem cell transplantation: a multicentre, randomized controlled clinical trial. Br. J. Haematol. 200, 210–221 (2023).
Wang, Y. et al. Low-dose post-transplant cyclophosphamide and anti-thymocyte globulin as an effective strategy for GVHD prevention in haploidentical patients. J. Hematol. Oncol. 12, 1–9 (2019).
Barkhordar, M. et al. Modified combination of anti-thymocyte globulin (ATG) and post-transplant cyclophosphamide (PTCy) as compared with standard ATG protocol in haploidentical peripheral blood stem cell transplantation for acute leukemia. Front. Immunol. 13, 921293 (2022).
Law, A. D. et al. Reduced-intensity conditioning and dual T lymphocyte suppression with antithymocyte globulin and post-transplant cyclophosphamide as graft-versus-host disease prophylaxis in haploidentical hematopoietic stem cell transplants for hematological malignancies. Biol. Blood Marrow Transpl. 24, 2259–2264 (2018).
Bonifazi, F. et al. Rabbit ATG/ATLG in preventing graft-versus-host disease after allogeneic stem cell transplantation: Consensus-based recommendations by an international expert panel. Bone Marrow Transpl. 55, 1093–1102 (2020).
Al-Homsi, A. S., Roy, T. S., Cole, K., Feng, Y. & Duffner, U. Post-transplant high-dose cyclophosphamide for the prevention of graft-versus-host disease. Biol. Blood Marrow Transpl. 21, 604–611 (2015).
Xu, X. et al. Low dose anti-thymocyte globulin with low dose posttransplant cyclophosphamide (low dose ATG/PTCy) can reduce the risk of graft-versus-host disease as compared with standard-dose anti-thymocyte globulin in haploidentical peripheral hematopoietic stem cell transplantation combined with unrelated cord blood. Bone Marrow Transpl. 56, 705–708 (2021).
Soiffer, R. J. et al. Prospective, randomized, double-blind, phase III clinical trial of anti–T-lymphocyte globulin to assess impact on chronic graft-versus-host disease–free survival in patients undergoing HLA-matched unrelated myeloablative hematopoietic cell transplantation. J. Clin. Oncol. 35, 4003 (2017).
Kennedy, V. E. et al. Optimizing antithymocyte globulin dosing for unrelated donor allogeneic hematopoietic cell transplantation based on recipient absolute lymphocyte count. Biol. Blood Marrow Transpl. 24, 150–155 (2018).
Fehse, N. et al. Influence of anti-thymocyte globulin as part of the conditioning regimen on immune reconstitution following matched related bone marrow transplantation. J. Hematother. Stem Cell research 12, 237–242 (2003).
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
D.H.K. collected and analyzed the data, performed the statistical analysis, interpretated the data, wrote the initial draft of the manuscript, and revised the manuscript. DY.S., Y.K., I.K., and SS.Y. interpretated the data and contributed in editing of the manuscript. J.M.B. and J.H. designed the study, interpretated the data, and reviewed the main manuscript. All the authors have read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kim, D.H., Shin, DY., Koh, Y. et al. Dual T-cell depletion with individually tailored anti-thymocyte globulin and attenuated dose of post-transplant cyclophosphamide in haploidentical peripheral stem cell transplantation. Sci Rep 14, 13885 (2024). https://doi.org/10.1038/s41598-024-64361-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-64361-5
- Springer Nature Limited