Abstract
Overexpression of NorA efflux pumps plays a pivotal role in the multidrug-resistance mechanism in S. aureus. Here, we investigated the activities of prenylated isoflavonoids, present in the legume plant family (Fabaceae), as natural efflux pump inhibitors (EPIs) in fluoroquinolone-resistant S. aureus. We found that four prenylated isoflavonoids, namely neobavaisoflavone, glabrene, glyceollin I, and glyceollin III, showed efflux pump inhibition in the norA overexpressing S. aureus. At sub-inhibitory concentrations, neobavaisoflavone (6.25 µg/mL, 19 µM) and glabrene (12.5 µg/mL, 39 µM), showed up to 6 times more Eth accumulation in norA overexpressing S. aureus than in the control. In addition, these two compounds boosted the MIC of fluoroquinolones up to eightfold. No fluoroquinolone potentiation was observed with these isoflavonoids in the norA knockout strain, indicating NorA as the main target of these potential EPIs. In comparison to the reported NorA EPI reserpine, neobavaisoflavone showed similar potentiation of fluoroquinolone activity at 10 µM, higher Eth accumulation, and less cytotoxicity. Neobavaisoflavone and glabrene did not exhibit membrane permeabilization effects or cytotoxicity on Caco-2 cells. In conclusion, our findings suggest that the prenylated isoflavonoids neobavaisoflavone and glabrene are promising phytochemicals that could be developed as antimicrobials and resistance-modifying agents to treat fluoroquinolone-resistant S. aureus strains.
Similar content being viewed by others
Introduction
Antimicrobial resistance (AMR) has become a fundamental problem worldwide. Recently, the COVID-19 pandemic accelerated the increase of resistance due to unnecessary antimicrobial use in hospitals1. Discovering new antibiotics remains a challenge and among numerous antibiotic candidates, only a few compounds have passed clinical trials in the last decade, including Zemdri (aminoglycoside), Xerava (tetracycline), and Nuzyra (tetracycline)2. To combat the rapid emergence of AMR, the development of novel therapeutic strategies, either with new antibiotics such as teixobactin or with resistance-modifying agents (RMAs), is urgently needed. RMAs are compounds that potentiate antimicrobial activity by perturbing multidrug resistance mechanisms3,4. The use of RMAs in drug combination therapy is gaining more interest because of their increased efficacy to tackle AMR5.
Staphylococcus aureus is one of the most widespread pathogens not only in hospital and community settings but also in livestock farming6,7. Staphylococcus aureus can develop resistance against several antimicrobials including vancomycin, fluoroquinolones, β-lactams, and the latest oxazolidinones (e.g., linezolid)8,9,10,11. Extensive investigations have been conducted to study putative mechanisms underlying resistance in S. aureus, such as the mutation of gyrA in DNA gyrase and the overexpression of efflux pump genes12,13. Efflux pumps play a pivotal role in AMR by extruding a broad range of noxious compounds, such as antibiotics, dyes, biocides, and other organic molecules and contribute to intrinsic and acquired resistance of bacteria14. Among different efflux pumps, members of the Major Facilitator Superfamily (MFS) are the most prevalent efflux pumps in gram-positive bacteria, including in S. aureus, for which the overexpression of NorA MFS efflux pump is observed in 43% of strains, particularly in MRSA strains15.
Considering their important role in the emergence of AMR, NorA efflux pumps are an interesting target for the development of RMAs. The use of EPIs can re-sensitize bacteria resistant to antibiotics16. Moreover, several EPIs are involved in impeding the evolution of resistance genes and decreasing the biofilm formation in S. aureus17,18,19. Unfortunately, there has been very little success in developing EPI as RMAs for clinical use. Whereas several synthetic EPIs have shown potent inhibition of efflux pump activity, they often suffer from off-target effects, cell toxicity and solubility issues20.
Plants can produce a myriad of metabolites as a response to microbial stress, which can be used as antimicrobials or RMAs. Various plant-derived compounds have been reported as NorA EPIs working synergistically with antibiotics, such as reserpine, 5′-methoxy-hydnocarpin (5′-MHC), totarol, epicatechin gallate and epigallocatechin gallate21,22,23,24. Amongst the NorA EPIs are some isoflavones namely biochanin A and genistein, other reported NorA EPIs are orobol, prenylated coumarin osthole and the prenylated flavonoid sophoraflavanone G25,26,27. Prenylated isoflavonoids are mainly found in the plant family Fabaceae28. These compounds are interesting sources for novel antimicrobials due to their diverse antimicrobial mechanisms, including membrane disruption, inhibition of ATP synthesis, reduction of biofilm formation, and inhibition of efflux pumps29. Prenylated isoflavonoids are substituted with hydrophobic prenyl (five-carbon isoprenoid) groups that exist in different configurations in nature (e.g. pyran, furan or chain prenyl)30. Prenylation of isoflavonoids usually occurs as a plant’s defense response to stress against both biotic (e.g., pathogenic microorganisms) or abiotic (e.g., oxidative stress) conditions31,32.
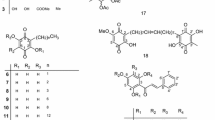
Here, we investigated prenylated isoflavonoids of 5 different subclasses and with different prenyl configurations (Fig. 1). The aim of this research was to evaluate whether prenylated isoflavonoids are effective against fluoroquinolone-resistant S. aureus, either as antimicrobials and/or as NorA EPIs. We hypothesize that prenylated isoflavonoids will have a high chance of acting as EPI since their hydrophobic features facilitate the penetration of compounds into the bacterial cell membrane and the binding of compounds into the binding pocket of the efflux pumps33. Moreover, these hydrophobic molecules possibly act as antimicrobials by disrupting bacterial membrane integrity34. This study aims to identify potential natural EPIs which might be studied further as potential RMAs against resistant S. aureus.
Results
Screening of prenylated isoflavonoids as potential NorA inhibitors
The first screening was performed to narrow down the selection of potential NorA efflux pump inhibitors (Fig. 2A). In this step, we used norA overexpressing strain to select the compounds that potentially act against NorA. EPI activity was plotted as a fold-change of Eth accumulation relative to its corresponding negative control. Intercalation of Eth to the intracellular DNA results in an enhanced quantum yield and therefore enhanced Eth fluorescence. SA-1199B overexpresses the norA efflux pump gene and shows an enhanced Eth efflux and consequently accumulates less dye, resulting in a low level of fluorescence. On the other hand, when EPIs block or competitively bind to NorA, Eth will not be effectively extruded, resulting in higher Eth accumulation and a higher fluorescence signal. Therefore, the fold changes in Eth accumulation correspond to the affinity of the compounds to bind and inhibit NorA resulting in lower efflux activity and higher Eth accumulation.
(A) Schematic diagram of the screening of prenylated isoflavonoids as candidate NorA EPIs; (B) Eth accumulation results with 11 tested compounds in the norA overexpressing strain (SA-1199B). The grey area corresponds to ≤ twofold change of fluorescence intensity (lower than positive control with reserpine); The dots represent the fold change of Eth accumulation at sub-inhibitory concentrations. Glabridin, dehydroglyceollidin IV, and dehydroglyceollidin II showed fold change ≥ 2 at MIC, therefore the data were not plotted in the Fig. 2B; (C) Eth accumulation results of the indicated candidate compounds that were used for further assays. Data were shown as an average of fold changes in Eth accumulation with standard deviations. Double asterisks denote a significant difference of p < 0.01, triple and quadruple asterisks denote p < 0.001 and p < 0.0001, respectively. Statistical significance was calculated with student’s t-test and Welch’s correction35.
Of the 11 prenylated isoflavonoids tested in the Eth accumulation assay, we selected compounds that showed Eth accumulation higher than our positive control reserpine, an indole alkaloid compound and known NorA inhibitor, which showed a twofold fluorescence increase (Fig. 2B)21. Subsequently, antimicrobial activity was measured to exclude false-positive results in the Eth accumulation assay (detailed MIC values in Table S1). Based on our selection criteria (Fig. 2A), four compounds were considered potential EPIs, as they showed promising EPI activity at sub-inhibitory concentrations (up to ½ MIC). Those four compounds were chain-prenylated neobavaisoflavone, ring-prenylated glabrene, pterocarpans glyceollin I, and glyceollin III. Neobavaisoflavone, glabrene, glyceollin I, and glyceollin III reached nearly two times higher Eth accumulation than the positive control reserpine (Fig. 2C).
Evaluating antimicrobial activities of prenylated isoflavonoids in S. aureus
Antimicrobial susceptibility assays were performed with neobavaisoflavone, glabrene, glyceollin I, glyceollin III, all four selected on basis of the results from the Eth accumulation assay (Fig. 2). We evaluated the MICs of the four compounds in the S. aureus wildtype strain SA-1199, the norA overexpressing strain SA-1199B and the norA knockout strain SA-K1758 (Table 1). The MIC values were examined to (1) characterize the activity of the candidate NorA EPIs against the other two strains of S. aureus and (2) check whether candidate NorA EPIs exerted antibacterial activities at the concentrations used in the Eth accumulation assay. Glabrene and neobavaisoflavone showed good antimicrobial activity with MIC values lower than 50 µg/mL in all strains. Neobavaisoflavone showed MIC and MBC values of 12.5 µg/mL and 25 µg/mL, respectively, in all S. aureus strains. Glabrene showed good antimicrobial activity with a MIC of 12.5–25 µg/mL against the wildtype SA-1199 and knockout SA-K1758, and MIC of 25 µg/mL against the overexpressing SA-1199B. Glyceollin I and III were considered moderate or weak antimicrobials due to their high MICs (≥ 50 µg/mL). Among other tested control compounds, ciprofloxacin and norfloxacin showed the highest MIC in norA overexpressing strain SA-1199B and the lowest MIC in norA knockout strain SA-K1758, which corroborates that these fluoroquinolones are substrates of NorA. Unlike for the fluoroquinolones, the overexpression of norA did not remarkably affect the MIC of prenylated isoflavonoids. Erythromycin showed an MIC value of 0.5–1 µg/mL in the norA overexpressing strain SA-1199B and was non-antimicrobial in the norA knockout strain SA-K1758 due to the chromosomally located erm resistance cassette23,36. The dye Eth and the known NorA inhibitor reserpine were found non-antimicrobial up to 50 µg/mL.
Evaluating efflux pump inhibition by prenylated isoflavonoids
To validate the specificity of prenylated isoflavonoids as NorA EPIs, we executed time-resolved Eth accumulation assays in the presence and absence of glabrene with the wildtype SA-1199, norA knockout strain SA-K1758, and the norA overexpressing strain SA-1199B (Fig. 3). For the wildtype and norA knockout strain, Eth is gradually accumulated during time even in the absence of glabrene, whereas the norA overexpressing strain does not show accumulation of Eth, indicating a strong Eth efflux activity. In the presence of glabrene (at ¼ of the MIC value), an enhanced accumulation rate and final Eth accumulation to approx. sixfold increase is shown. The same steady state level is observed for all three strains. The fold changes of the Eth accumulation after 60 min in the presence vs. the absence of glabrene is 6.1, 1.9, and 1.3 for the norA overexpressing, wildtype or norA knockout strain, respectively.
Eth accumulation in the presence of glabrene at a concentration of 6.25 µg/mL (1/4 MIC) in (A) norA overexpressing strain (SA-1199B), (B) wildtype strain (SA-1199), (C) norA knockout strain (SA-K1758). The open circles correspond to the control (cells without glabrene) and the closed (black) circles correspond to cells with glabrene. In these graphs, representative data from three technical replicates of two biological experiments is shown. FC is a fold change in the increase of Eth accumulation after 60 min measurement. The average of two biological repetitions in Eth accumulation graphs were presented in the supplementary Fig. S1 to S3.
Eth accumulation experiments were also conducted in the absence and presence of neobavaisoflavone, glyceollin I and glyceollin III (Fig. S1 to S3). The compounds alone (no cells) in buffer with and without EtBr did not emit fluorescence, as shown in Fig. S4. In Fig. 4, the fold change (FC) of Eth accumulation after 60 min in the presence vs. the absence of the prenylated isoflavonoids for each of the three different S. aureus strains is shown. For all prenylated isoflavonoids, we found a notable FC of Eth accumulation with the norA overexpressing strain, and smaller FCs in the wildtype and the norA knockout strain (fold changes in SA-1199B > SA-1199 > SA-K1758). At the maximum concentration tested in norA overexpressing strain SA-1199B, glabrene showed the highest fold change in Eth accumulation (FC = 5.9 ± 0.8), followed by neobavaisoflavone (FC = 4.5 ± 0.4). Glyceollin I and glyceollin III also displayed higher activity in the norA overexpressing strain with FCs of 2.3 ± 0.5 and 2.4 ± 0.5, respectively. These results denote that NorA is potentially the major target of the tested compounds, particularly glabrene and neobavaisoflavone, suggesting them as the most promising EPI candidates. However, since minor FCs between 0.9 and 1.4 were observed for the norA knockout strain SA-K1758, NorA is potentially not the only efflux pump affected by the tested compounds or the compounds affect the membrane permeability in the norA knockout strain. We therefore performed further assays, such as antibiotic potentiation with NorA substrates and non-NorA substrates and membrane permeabilization assays to address the NorA-specificity of the prenylated isoflavonoids.
Effect of different concentrations of prenylated isoflavonoids (A) glabrene, (B) neobavaisoflavone, (C) glyceollin I, (D) glyceollin III, on the fold change (FC) in Eth accumulation after 1 h measurement with norA overexpressing (SA-1199B), wildtype (SA-1199), and norA knockout (SA-K1758) strains. The glabrene and neobavaisoflavone concentrations used in Fig. 4A and B were chosen based on MIC values in SA-1199B (MIC glabrene 25 µg/mL; MIC neobavaisoflavone 12.5 µg/mL, MIC glyceollin I and glyceollin III > 50 µg/mL).
Evaluating antibiotic potentiation effect of prenylated isoflavonoids
Efflux of fluoroquinolones by NorA has been shown to be an effective resistance mechanism against these antibiotics37. To verify whether prenylated isoflavonoids modulate the antibacterial activity of fluoroquinolones, we performed checkerboard assays in both norA overexpressing and norA knockout strains. Table 2 displays the minimal inhibitory concentration (MIC) of ciprofloxacin, norfloxacin, and erythromycin in absence and presence of prenylated isoflavonoids. Antibiotic potentiation is often described as fold reduction; the extent of the reduction of the MIC indicates the potency of compounds in modulating the efficacy of the antibiotics. We found that glabrene, neobavaisoflavone, glyceollin I, and glyceollin III were able to reduce the MIC of fluoroquinolones in the norA overexpressing strain SA-1199B by 4- to eightfold. When we standardized the antibiotic potentiation at fourfold reduction (to compare with the maximum fold reduction observed with the positive control reserpine, bold rows in Table 2), glabrene and neobavaisoflavone showed synergism with the tested fluoroquinolone antibiotics (FICI = 0.5). All prenylated isoflavonoids reduced the MIC of ciprofloxacin and norfloxacin fourfold (except for norfloxacin in combination with glabrene), matching the fold reduction inflicted by reserpine (Table 2). The modulators were used at different concentrations to elicit this effect, neobavaisoflavone was used at a concentration of 3.125 µg/mL (10 µM), glabrene at a concentration of 6.25 µg/mL (19 µM), and reserpine at a concentration of 6.25 µg/mL (10 µM). There was a slight potentiation by twofold observed in norA knockout strain SA-K1758 at the highest concentrations tested for glabrene and neobavaisoflavone. The twofold reduction in the norA knockout could be attributed to the more susceptible properties of the norA knockout towards antibiotics compared to the norA overexpressing strain. Moreover, both prenylated isoflavonoids and antibiotics can give an additive effect when applied at ½ the MIC values each, resulting in twofold reduction. The FICI values showed that glabrene acted in synergy (FICI = 0.5) with ciprofloxacin and neobavaisoflavone synergized with both fluoroquinolone antibiotics. Based on the fourfold reduction at 10 µM and apparent synergism with both fluoroquinolones, neobavaisoflavone showed the most promising activities among other tested prenylated isoflavonoids as a NorA EPI, in addition to its antimicrobial properties.
Pterocarpans glyceollin I and III showed a fourfold reduction of MICs of both fluoroquinolone antibiotics albeit at higher concentrations (89–118 µM) compared to glabrene and neobavaisoflavone. Pterocarpans were considered partial potentiators (0.5 < FICI < 1) in SA-1199B, while the effect was only additive or indifferent in SA-K1758 (1 ≤ FICI < 4).
A minor antibiotic potentiation (maximum two-fold reduction) was observed for prenylated isoflavonoids and reserpine with the non-fluoroquinolone antibiotic erythromycin. We did not assess the potentiation of erythromycin in the norA knockout strain SA-K1758 because the introduction of erm cassette to this strain resulted in resistance towards erythromycin23,36. To conclude, neobavaisoflavone and to a lesser extent glabrene, stand as the most promising antibiotic potentiator among the tested compounds in the checkerboard assay, in which the potentiation effect might be due to the inhibition of NorA efflux pumps.
Evaluating membrane permeabilization effect of prenylated isoflavonoids
To check the membrane permeabilization effect of prenylated isoflavonoids, we conducted propidium iodide (PI) assays in the three S. aureus strains. PI is a fluorescent dye that produces high fluorescence when it binds to DNA. This dye is commonly used to assess the cell viability in mammalian cells, but also widely utilized to check bacterial membrane integrity38,39. When the cell membrane is not intact, like in heated cells, PI will enter the cells and produce an increase in fluorescence. According to Fig. 5, prenylated isoflavonoids showed some low levels of PI uptake at their maximum concentration tested in the Eth accumulation assay and antibiotic potentiation assay. We also performed the PI assay in the presence of the uncoupler carbonyl cyanide-m-chlorophenyl hydrazone (CCCP) affecting the proton motive force (PMF), as the extent of the PMF may influence propidium ion permeability40. As propidium has a similar structure in terms of the phenanthridine ring as Eth, which is one of the substrates for diverse efflux pumps, we wanted to confirm that the low PI uptake was not due to the extrusion of PI by the efflux pumps (after permeabilization of the membrane)41,42. By dissipating the PMF and therefore inactivating the efflux PMF (uncoupling the pumps), we were able to exclude propidium’s extrusion (Fig S5). Based on our results, prenylated isoflavonoids did not show permeabilization properties up to ½ MIC with and without CCCP in all S. aureus strains.
Effect of prenylated isoflavonoids and reserpine on membrane permeability at their maximum concentration tested in Eth accumulation assay. Data were expressed as relative propidium iodide (PI) uptake, which was obtained by taking final fluorescence after 2 h measurement and calculated relative to the negative control. The definitions of non-permeabilizers (relative PI uptake ≤ 1), poor permeabilizers (relative PI uptake 1–2), good permeabilizers (relative PI uptake > 2) are shown by dotted lines.
Evaluating cytotoxicity and cell viability of prenylated isoflavonoids
The cytotoxicity of glabrene and neobavaisoflavone was determined against Caco-2 cells. According to the LDH assay, these prenylated isoflavonoids did not exhibit profound cell cytotoxicity after 4 h and 24 h incubation, where the cell cytotoxicity was established (in comparison to the positive control) at concentrations up to 25 µg/mL (78 µM) and 12.5 µg/mL (39 µM), respectively (Fig. 6). In addition, no notable cytotoxicity effect was seen with glabrene and neobavaisoflavone after 4 h incubation at concentrations up to 50 µg/mL (155 µM) and 25 µg/mL (78 µM), respectively. However, cytotoxicity was observed after 24 h incubation at the same concentrations.
We carried out a cell viability assay to further demonstrate the effect of the compounds on the metabolic activity of cells (Fig. 7). Glabrene and neobavaisoflavone did not affect cell viability up to ½ MIC, which corresponds to the LDH assay results (Fig. 6). However, the cell viability was slightly reduced to 80% at MIC and further reduction was observed at 2 × MIC. According to these findings, neither glabrene nor neobavaisoflavone had any discernible cytotoxic effects at the tested EPI concentrations.
Discussion
Prenylated isoflavonoids act as potential NorA efflux pump inhibitors in S. aureus
According to the criteria described by Lomovskaya et al. and Opperman et al., prenylated isoflavonoids, particularly glabrene and neobavaisoflavone, seem to be promising EPI candidates based on (1) increased level of Eth accumulation in norA overexpressing strain, (2) antibiotic potentiation in norA overexpressing strain, (3) no significant antibiotic potentiation effect in norA knockout strain, (4) no remarkable antibiotic potentiation with an antibiotic which is not the main substrate for NorA43,44. To ensure the EPI activity towards NorA, we performed antibiotic potentiation assays by combining prenylated isoflavonoids with fluoroquinolones (substrates of NorA) and erythromycin (non-substrate of NorA). The lower potentiation effect that was found in the norA knockout and higher potentiation in the norA overexpressing strain indicates that the mode of action was associated with the NorA efflux pump. Our potentiation results with neobavaisoflavone are in line with those of Abreu et al., where a FICI of 0.50 was observed with fourfold ciprofloxacin MIC reduction45. However, we found up to eightfold ciprofloxacin reduction at ½ MIC. Compared to Abreu et al., we used a more comprehensive approach to confirm the activity of neobavaisoflavone as a NorA inhibitor by performing the assay with the norA knockout strain as well45. Glyceollin I and glyceollin III showed comparable inhibitory behavior, where fourfold to eightfold reduction of ciprofloxacin and norfloxacin MIC were observed in the norA overexpressing strain at concentrations up to 50 µg/mL. Yet, glyceollin I and glyceollin III required higher concentrations than glabrene and neobavaisoflavone. Furthermore, glyceollin I can only reduce the MIC of fluoroquinolones by a maximum of fourfold. We tested the potentiation effect of these four prenylated isoflavonoids with the non-fluoroquinolone antibiotic erythromycin to ensure the specificity of prenylated isoflavonoids as NorA EPI. Erythromycin can be pumped out by ABC transporter MsrA and MFS transporter Mef in S. aureus46,47. Our tested prenylated isoflavonoids did not show any erythromycin potentiation in norA overexpressing strain. Therefore, MsrA and/or Mef appear not to be the main targets of our tested compounds. Based on the notable potentiation observed with fluoroquinolones as verified NorA substrates, we hypothesize that NorA is the main target of these prenylated isoflavonoids.
With our findings, we postulate that glabrene and neobavaisoflavone mainly act as efflux pump inhibitors. In terms of potentiating fluoroquinolones, neobavaisoflavone and glabrene demonstrated promising activities in comparison with previously reported phytochemicals. Both neobavaisoflavone and glabrene reduced the effectiveness of ciprofloxacin by eightfold at concentrations of 19 and 39 µM, respectively, while non-prenylated isoflavonoids genistein and biochanin A gave the same fold reduction at concentrations of 222 µM and 105 µM, respectively45. Osthol, a prenylated coumarin, potentiated ciprofloxacin by fourfold at 25 µM, whereas sophoraflavanone G, a prenylated flavanone, was reported to potentiate norfloxacin by 16-fold at ¼ MIC (2.4 µM)26,27. Neobavaisoflavone showed similar potentiation of fluoroquinolone activity with the known NorA EPI reserpine at 10 µM, and eightfold reduction was observed at two times concentration.
Neobavaisoflavone and glabrene also showed antimicrobial activities against S. aureus. According to a previous classification of antimicrobial activities of natural compounds, neobavaisoflavone can be classified as a very good antibacterial against all three S. aureus strains (MIC ≤ 15 µg/mL), whereas glabrene can be classified as very good to good antibacterial (12.5 < MIC < 25 µg/mL)30. We postulate that glabrene and neobavaisoflavone potentially have dual mechanisms of action by acting as EPIs and antibacterials at specific concentrations.
Glabrene and neobavaisoflavone did not show permeabilization and cytotoxicity at the tested EPI concentrations
We evaluated the secondary effect of the two most active compounds as membrane permeabilizers and their cytotoxicity properties. By performing the PI assay, we infer that the increase of fluorescence in the Eth assay was mainly due to efflux inhibition rather than membrane permeabilization. Yet, we should take into account that some prenylated isoflavonoids are good permeabilizers when they reach their MIC values48. The PI uptake is also facilitated when the cells have elevated PMF40. One putative explanation for some low levels of PI uptake indicated with neobavaisoflavone and glabrene is that by inhibiting the NorA efflux pump, less PMF is consumed and might have an effect on the steady-state PMF level. In other words, these two compounds might not have a permeabilization effect, but by inhibiting the EP, they elevate the PMF steady-state level, leading to enhanced PI uptake.
In addition to the PI assay, we assessed the cytotoxicity effect of glabrene and neobavaisoflavone in Caco-2 cell lines. According to our cytotoxicity assessment, glabrene and neobavaisoflavone did not show notable cytotoxic effects at a concentration up to 25 µg/mL (78 µM) and 12.5 µg/mL (39 µM), respectively. Despite that no IC50 was determined, the cytotoxicity of glabrene and neobavaisoflavone were lower compared to the cytotoxicity of the known NorA EPI reserpine (IC50 30.07 ± 7.57 μM against HCT116 (p53+/+) colon cancer cells)49. One of the disadvantages of the Caco-2 cell lines is that, when compared to normal intestinal enterocytes, these cell lines have absorptive enterocytes only and no other epithelial cells, such as goblet cells and enteroendocrine cells50. Moreover, Caco-2 cell lines have different expressions of metabolic enzymes, leading to unrepresentative situations compared to normal intestinal cells51. To further evaluate the cytotoxicity properties of the compounds, experiments in a more representative model of normal intestinal cells should be performed, for example, primary cell lines, co-cultures (Caco-2 in combination with HT29-I cells), and intestinal organoids50,52,53.
Predicted key molecular properties of prenylated isoflavonoids as potential NorA EPIs
Based on the antibiotic potentiation activity (fourfold reduction of ciprofloxacin MIC), some molecular properties were found highly-correlated (r ≥ 0.95) with EPI activities of glabrene, neobavaisoflavone, glyceollin I, and glyceollin III (Fig. 8, Table S2). Hydrophobic surface area and volume-related descriptors (together 41%) and hydrophobic/hydrophilic balance descriptors (24%) accounted for the majority of descriptors highly-correlated with EPI activity. Partial charge-related descriptors (12%) and molecular size (12%) followed in importance. These results agree with our previous quantitative structure–activity relationships (QSAR) study where hydrophobicity, the balance of hydrophobicity/hydrophilicity, and (partial) charge were involved in the anti-MRSA (methicillin resistant S. aureus) activity of a larger set of prenylated isoflavonoids54. It seems that the same key molecular properties influence antibacterial activity and NorA EPI activity. Nonetheless, as noticed in our initial screening (Fig. 2B and Table S1), not all good NorA EPI candidates showed good antibacterial activity (and vice versa).
Hydrophobicity has been proposed to facilitate the interaction of prenylated isoflavonoids with bacterial membranes, whereas hydroxyl groups help keeping the hydrophobic/hydrophilic balance and facilitate intermolecular hydrogen bonds54,55. The presence of phenylalanine residues in the substrate binding pocket of NorA confers hydrophobicity to this site and may offer π–π stacking possibilities between aromatic rings of prenylated isoflavonoids and these residues56. In the future, a larger set of prenylated isoflavonoids is required to understand the structure–activity relationships of prenylated isoflavonoids as NorA EPIs. Furthermore, protein–ligand binding assays and co-crystallization of NorA with prenylated isoflavonoids will provide definitive confirmation of their binding and shed light into their mechanism of action as EPIs.
Conclusions
The interplay of prenylated isoflavonoids as natural antimicrobials and efflux pump inhibitors against fluoroquinolone-resistant S. aureus makes these compounds interesting hits to develop new and potent RMAs. At concentrations up to ½ MIC in the norA overexpressing strain, glabrene and neobavaisoflavone showed fold changes in Eth accumulation of 5.9 and 4.5 times higher than the control, respectively, indicating an effective reduction of efflux pump activity. In addition, both compounds pronouncedly potentiated fluoroquinolones at a low concentration in the norA overexpressing S. aureus strain, resulting in an up to eightfold reduction of ciprofloxacin and norfloxacin MIC. This potentiation was not observed in the norA knockout strain. Neobavaisoflavone demonstrated a better activity than reserpine by showing higher Eth accumulation and less cytotoxicity. Moreover, neither notable membrane permeabilization nor cytotoxic effects of glabrene and neobavaisoflavone were observed at sub-inhibitory concentrations up to ½ MIC. These findings suggest that the prenylated isoflavonoids glabrene and neobavaisoflavone are potential NorA EPIs and have promising activities for use in combination therapy to tackle NorA-mediated fluoroquinolone resistance in S. aureus. Establishing the (quantitative) structure–activity relationships of prenylated isoflavonoids as NorA EPIs and elucidating their molecular mechanism of inhibition are essential steps for follow up studies.
Methods
Bacterial strains
S. aureus (SA)-1199B (a fluoroquinolone-resistant strain overexpressing norA), SA-1199 (Wild-type clinical isolate strain), and SA-K1758 (norA knockout strain) from BEI Resources were kindly provided by Prof. Michael J. Rybak (Wayne State University, USA) and Prof. Glenn W. Kaatz (Wayne State University, USA). The norA knockout strain (SA-K1758) is a derivative of S. aureus NCTC 8325-4, in which the norA gene was deleted and replaced with an erm cassette carrying the erythromycin resistance gene23,36. The bacteria were stored in a glycerol stock at − 80 °C.
Media, reagents, and phytochemicals
Ethidium bromide (EtBr), propidium iodide (PI), phosphate buffer saline (pH 7.4), reserpine, erythromycin, and ciprofloxacin were purchased from Sigma Aldrich (St. Louis, MO, USA). Peptone physiological salt solution (PPS) was purchased from Tritium Microbiologie (Eindhoven, The Netherlands). Tryptone soy agar (TSA) and tryptone soy broth (TSB) were purchased from Oxoid Ltd (Basingstoke, UK). Dimethyl sulfoxide (DMSO) was purchased from Brunschwig Chemie B.V. (Amsterdam, The Netherlands) and ethyl acetate absolute was from Biosolve B.V. (Valkenswaard, The Netherlands). Neobavaisoflavone (≥ 95% purity) was supplied by PhytoLab GmbH & Co.KG (Vestenbergsgreuth, Germany). Glabridin (97% purity) was purchased from Wako (Osaka, Japan). Glabrene, glyceollin I, glyceollin III, glyceollin IV, glyceollidin II, dehydroglyceollin I, dehydroglyceollin III, dehydroglyceollin IV, and dehydroglyceollidin II were previously purified from roots of Glycyrrhiza glabra and from soybeans (Glycine max (L.) Merrill)57,58. All the tested phytochemicals have ≥ 90% purity.
Antimicrobial susceptibility test
The minimum inhibitory concentrations (MIC) and minimum bactericidal concentrations (MBC) of compounds were determined by using broth microdilution method. Bacteria were streaked from − 80 °C glycerol stock onto TSA agar plates and incubated at 37 °C for 24 h. After overnight incubation, one colony was transferred in 10 mL TSB and grown for another 24 h at the same temperature. Afterwards, the inoculum was diluted in TSB to reach an average inoculum of 5.31 ± 0.16, 5.33 ± 0.22, 5.04 ± 0.21 log10 colony forming units (CFU)/mL for SA-1199B, SA-1199, and SA-K1758, respectively. Stock solutions of compounds were prepared in DMSO and subsequently diluted in TSB. Antibiotics were prepared in phosphate-buffered saline (PBS) (0.1% v/v acetic acid). Next, tested compounds (100 µL) and inoculum (100 µL) were mixed in a 96-well plate, where the final concentration of compounds ranged from 6.25 to 50 µg/mL (solvent concentration was at max. 2% v/v). The negative control (inoculum in TSB with 2% DMSO) and blank (TSB medium without bacteria) were included in each assay. Microbial growth was measured spectrophotometrically every 10 min at OD600 (Tecan-Infinite 200 Pro M nanoplate reader) at 37 °C for 24 h under continuous shaking.
When there was no change in OD600 (flat line) after 24 h measurement, cell viability was measured by plate counting to obtain the MIC and MBC values48. The MIC was defined as the lowest concentration of compound (drug) that resulted in equal or a lower cell count compared to the initial inoculum. MBC was defined as the lowest concentration of compound that showed more than 99.9% bacterial killing from the initial inoculum59. The activities of prenylated isoflavonoids against SA-1199B, SA-1199, and SA-K1758 were tested in technical triplicates with at least two biological repetitions each.
Ethidium accumulation assay
Ethidium accumulation assays were performed according to Coldham et al. with a slight modification60. One colony from an overnight TSA plate was grown in 3 mL TSB medium for approximately 4 h (shaking 200 rpm, at 37 °C) until reaching an OD600 1.2–1.4. The inoculum was aliquoted into 1 mL microtubes and centrifuged at 4000 g for 5 min. The supernatant was discarded, and the cell pellet washed once with PBS buffer (pH 7.4), supplemented with 1 mM MgSO4. The cell pellet was resuspended in the same buffer at OD600 = 0.6. The average inoculum sizes were 9.19 ± 0.17, 9.22 ± 0.14, and 9.00 ± 0.10 log10 CFU/mL for SA-1199B, SA-1199 and SA-K1758, respectively.
Compound stock solutions were prepared in 100% DMSO and subsequently PBS was used to dissolve the compounds and EtBr. The cell suspension (150 μL) was placed into a black 96-well plate with 10 μL EtBr (final concentration 3 μg/mL or 7.6 µM) and 40 μL of compounds (max. 2% v/v DMSO). Negative control (cells with 2% DMSO and EtBr) and blanks (compounds with both buffer and EtBr) were prepared for each assay. The addition of reserpine (20 µg/mL) was used as a positive control21. The ethidium fluorescence was measured by using Spectramax ID3 for 60 min (37 °C), with excitation 520 nm and emission 620 nm. Blanks (each tested compound in buffer with and without EtBr) were measured for each sample to check and correct background fluorescence. The fold change in Eth accumulation was calculated by taking the final fluorescence unit (RFU) relative to the negative control at t = 60 min, as described by Šimunović et al.61.
The first screening with EPI candidates was performed in norA overexpressing strain (SA-1199B) with concentrations ranging from 6.25 to 50 µg/mL. The fold changes in Eth accumulation were shown as means with standard deviation. Statistical significance was analyzed by a student’s t-test with Welch’s correction35. All samples were measured in technical triplicates with two biological repetitions each.
Antibiotic potentiation study (checkerboard assay)
The antibiotic potentiation effect was assessed by using the same procedure as described in the antimicrobial susceptibility test. Bacteria were diluted in TSB to obtain an initial inoculum of 5.37 ± 0.13 and 5.10 ± 0.01 log10 CFU/mL for SA-1199B and SA-K1758, respectively. The 96-well plate was filled with two-fold serial dilution of the antibiotics in combination with prenylated isoflavonoids, starting from ½ to 1/8 MIC. For those compounds with MIC values higher than 50 µg/mL, such as the pterocarpans (Fig. 1), a series of 20, 30, 40, and 50 µg/mL was used. In a 96-well plate, 50 µL of antibiotics and prenylated isoflavonoids were mixed with 100 µL of inoculum (max. 2% DMSO and 0.01% v/v acetic acid). All experiments were done in technical duplicates with at least two biological repetitions each. The fractional inhibitory concentration index (FICI) was calculated according to the formula below:
FICI ≤ 0.5 was defined as “synergy”; 0.5 < FICI < 1 was considered as “partial synergy”; FICI = 1 was categorized as additive; 1 < FICI < 4 was observed as “indifferent” and FICI ≥ 4.0 was indicated as “antagonistic”62. In case the prenylated isoflavonoids did not exhibit an antimicrobial effect itself (no detectable MIC at the maximum concentration tested), the FICI was estimated with the highest concentration of the compound examined.
Membrane permeability assay
Propidium iodide (PI) was used to assess membrane permeabilization, as previously reported with slight modifications48. In brief, bacteria were grown on TSA plates incubated overnight at 37 °C. One colony was transferred in 25 mL TSB and cells were grown for 20 h at 37 °C. The cell cultures were harvested by centrifugation at 4,696 g, 4 °C for 5 min. The cell pellet was washed twice with 5 mL PPS. Subsequently, the pellet was resuspended in 5 mL of PPS to obtain average inoculum sizes of 9.75 ± 0.01, 9.74 ± 0.35 and 9.30 ± 0.06 log10 CFU/mL for SA-1199B, SA-1199, and SA-K1758, respectively. The inoculum (100 μL) and tested compounds (50 µL) at final concentrations of up to ½ MIC, were added to each well in a black 96-well plate with a transparent bottom. A positive control was prepared by heating the cells for 10 min at 95 °C. The negative control contained cells and PI with a final volume of 200 µL. Fluorescence of the initial samples (tested compounds with PI; PI only, without compounds) were checked for the intrinsic fluorescence of prenylated isoflavonoids. Experiments were performed at least in technical duplicates with two biological repetitions each. The relative PI uptake was calculated by taking the final fluorescence unit (RFU) relative to the negative control at t = 120 min.
Cytotoxicity assay
Following 4- and 24-h incubation periods, the cytotoxic effects of glabrene and neobavaisoflavone were tested at a range of concentrations starting from ½ to 2 × MIC. The cytotoxic effects on human colonic carcinoma cell line Caco-2 were assessed by measuring intracellular lactate dehydrogenase (LDH) leakage in the supernatant and analyzing the results using an LDH cytotoxicity detection kit (Roche Applied Science, Almere, The Netherlands)63. Compounds were dissolved in DMSO and the maximum DMSO concentration used was 1.56% (v/v), which reportedly did not affect cell cytotoxicity. LDH activity in the supernatant was expressed as a percentage of the maximum releasable LDH in cells (Caco-2 cells treated with 1% Triton X-100) and calculated with Eq. (4).
Here, exp.value is UV absorbance at 492 nm (Spectramax M2, Molecular Devices). Low control is the spontaneous LDH release in untreated cells (Caco-2 cells in culture medium), and high control is the maximum releasable LDH in cells (Caco-2 cells treated with 1% Triton X-100). The compounds were considered cytotoxic when > 20% cytotoxicity was observed.
Effects on cell viability in Caco-2 cells were assessed after incubation of 4 h. The Caco-2 cell viability was evaluated by measuring the cleavage of the tetrazolium salt WST-1 to formazan by cellular mitochondrial dehydrogenases. The cell viability was examined by using a WST-1 cell viability kit (PromoKine, Heidelberg, Germany), as per the manufacturer’s instructions. Cell viability was expressed as percentage of the control cells (Caco-2 cells in culture medium) and calculated with Eq. (5).
Here, exp.value is UV absorbance at 450 nm (Multiskan Ascent, Thermo Fisher Scientific), and low control is spontaneous cleavage of WST-1 to formazan by mitochondrial dehydrogenases in untreated cells. All data were presented in GraphPad Prism version 9.
Prediction of molecular properties
The 3D optimized structures of prenylated isoflavonoids and molecular properties (descriptors) were calculated using Molecular Operating Environment (MOE) software (version 2019.0102, Chemical Computing Group) after MMFF94x energy minimization (gradient 0.01). Correlation between molecular descriptors and antibiotic potentiation activity (i.e. concentration required for fourfold reduction of ciprofloxacin MIC) was assessed based on the Pearson correlation coefficient (r). List of highly significantly correlated (r ≥ 0.95, p < 0.05) molecular descriptors is in Table S2 in the supplementary file.
Data availability
Upon reasonable request, the corresponding author will provide the datasets used and/or analyzed during the study.
Abbreviations
- EPIs:
-
Efflux pump inhibitors
- AMR:
-
Antimicrobial resistance
- RMAs:
-
Resistance-modifying agents
- MFS:
-
Major facilitator superfamily
- 5′-MHC:
-
5′-Methoxy-hydnocarpin
- Eth:
-
Ethidium
- EtBr:
-
Ethidium bromide
- PI:
-
Propidium iodide
- MIC:
-
Minimum inhibitory concentration
- MBC:
-
Minimum bactericidal concentrations
- FICI:
-
Fractional inhibitory concentration index
- CFU:
-
Colony forming units
- RFU:
-
Relative fluorescence unit
- PMF:
-
Proton-motive force
- PPS:
-
Peptone physiological salt solution
- TSA:
-
Tryptone soy agar
- TSB:
-
Tryptone soy broth
- DMSO:
-
Dimethyl sulfoxide
- LDH:
-
Lactate dehydrogenase
- MOE:
-
Molecular operating environment
- CCCP:
-
Cyanide-m-chlorophenyl hydrazone
- QSAR:
-
Quantitative structure–activity relationships
References
Castro-Lopes, A. et al. Increase of antimicrobial consumption in a tertiary care hospital during the first phase of the COVID-19 pandemic. Antibiotics 10, 778. https://doi.org/10.3390/antibiotics10070778 (2021).
Hutchings, M. I., Truman, A. W. & Wilkinson, B. Antibiotics: Past, present and future. Curr. Opin. Microbiol. 51, 72–80. https://doi.org/10.1016/j.mib.2019.10.008 (2019).
Piddock, L. J. Teixobactin, the first of a new class of antibiotics discovered by iChip technology?. J. Antimicrob. Chemother. 70, 2679–2680. https://doi.org/10.1093/jac/dkv175 (2015).
Gibbons, S. Anti-staphylococcal plant natural products. Nat. Prod. Rep. 21, 263–277. https://doi.org/10.1039/b212695h (2004).
Abreu, A. C., McBain, A. J. & Simoes, M. Plants as sources of new antimicrobials and resistance-modifying agents. Nat. Prod. Rep. 29, 1007–1021. https://doi.org/10.1039/c2np20035j (2012).
Noskin, G. A. et al. The burden of Staphylococcus aureus infections on hospitals in the United States: An analysis of the 2000 and 2001 Nationwide Inpatient Sample Database. Arch. Intern. Med. 165, 1756–1761. https://doi.org/10.1001/archinte.165.15.1756 (2005).
Smith, T. C. Livestock-associated Staphylococcus aureus: The United States experience. PLoS Pathog. 11, e1004564. https://doi.org/10.1371/journal.ppat.1004564 (2015).
Centers for Disease, C. & Prevention. Staphylococcus aureus resistant to vancomycin. MMWR Morb. Mortal. Wkly. Rep. 51, 565–567 (2002)
Harnett, N., Brown, S. & Krishnan, C. Emergence of quinolone resistance among clinical isolates of methicillin-resistant Staphylococcus aureus in Ontario, Canada. Antimicrob. Agents Chemother. 35, 1911–1913. https://doi.org/10.1128/AAC.35.9.1911 (1991).
Sabath, L. D. Mechanisms of resistance to beta-lactam antibiotics in strains of Staphylococcus aureus. Ann. Intern. Med. 97, 339–344. https://doi.org/10.7326/0003-4819-97-3-339 (1982).
Tsiodras, S. et al. Linezolid resistance in a clinical isolate of Staphylococcus aureus. Lancet 358, 207–208. https://doi.org/10.1016/S0140-6736(01)05410-1 (2001).
Trucksis, M., Wolfson, J. S. & Hooper, D. C. A novel locus conferring fluoroquinolone resistance in Staphylococcus aureus. J. Bacteriol. 173, 5854–5860. https://doi.org/10.1128/jb.173.18.5854-5860.1991 (1991).
Kaatz, G. W., Seo, S. M. & Ruble, C. A. Efflux-mediated fluoroquinolone resistance in Staphylococcus aureus. Antimicrob. Agents Chemother. 37, 1086–1094. https://doi.org/10.1128/aac.37.5.1086 (1993).
Hernando-Amado, S. et al. Multidrug efflux pumps as main players in intrinsic and acquired resistance to antimicrobials. Drug Resist. Updat. 28, 13–27. https://doi.org/10.1016/j.drup.2016.06.007 (2016).
Patel, D., Kosmidis, C., Seo, S. M. & Kaatz, G. W. Ethidium bromide MIC screening for enhanced efflux pump gene expression or efflux activity in Staphylococcus aureus. Antimicrob. Agents Chemother. 54, 5070–5073. https://doi.org/10.1128/AAC.01058-10 (2010).
Cernicchi, G., Felicetti, T. & Sabatini, S. Microbial efflux pump inhibitors: A journey around quinoline and indole derivatives. Molecules 26, 6996. https://doi.org/10.3390/molecules26226996 (2021).
Kvist, M., Hancock, V. & Klemm, P. Inactivation of efflux pumps abolishes bacterial biofilm formation. Appl. Environ. Microbiol. 74, 7376–7382. https://doi.org/10.1128/AEM.01310-08 (2008).
Sabatini, S. et al. Investigation on the effect of known potent S. aureus NorA efflux pump inhibitors on the staphylococcal biofilm formation. RSC Adv. 7, 37007–37014. https://doi.org/10.1039/C7RA03859C (2017).
Papkou, A., Hedge, J., Kapel, N., Young, B. & MacLean, R. C. Efflux pump activity potentiates the evolution of antibiotic resistance across S. aureus isolates. Nat. Commun. 11, 3970. https://doi.org/10.1038/s41467-020-17735-y (2020).
Opperman, T. J. & Nguyen, S. T. Recent advances toward a molecular mechanism of efflux pump inhibition. Front. Microbiol. 6, 421. https://doi.org/10.3389/fmicb.2015.00421 (2015).
Schmitz, F. J. et al. The effect of reserpine, an inhibitor of multidrug efflux pumps, on the in-vitro activities of ciprofloxacin, sparfloxacin and moxifloxacin against clinical isolates of Staphylococcus aureus. J. Antimicrob. Chemother. 42, 807–810. https://doi.org/10.1093/jac/42.6.807 (1998).
Stermitz, F. R., Lorenz, P., Tawara, J. N., Zenewicz, L. A. & Lewis, K. Synergy in a medicinal plant: Antimicrobial action of berberine potentiated by 5 ’-methoxyhydnocarpin, a multidrug pump inhibitor. Proc. Natl. Acad. Sci. USA 97, 1433–1437 (2000).
Smith, E. C. et al. The phenolic diterpene totarol inhibits multidrug efflux pump activity in Staphylococcus aureus. Antimicrob. Agents Chemother. 51, 4480–4483. https://doi.org/10.1128/AAC.00216-07 (2007).
Gibbons, S., Moser, E. & Kaatz, G. W. Catechin gallates inhibit multidrug resistance (MDR) in Staphylococcus aureus. Planta Med. 70(12), 1240–1242. https://doi.org/10.1055/s-2004-835860 (2004).
Waditzer, M. & Bucar, F. Flavonoids as inhibitors of bacterial efflux pumps. Molecules 26, 6904. https://doi.org/10.3390/molecules26226904 (2021).
Joshi, P. et al. Osthol and curcumin as inhibitors of human Pgp and multidrug efflux pumps of Staphylococcus aureus: Reversing the resistance against frontline antibacterial drugs. Medchemcomm 5, 1540–1547. https://doi.org/10.1039/C4MD00196F (2014).
Sun, Z. L. et al. Synergism of sophoraflavanone G with norfloxacin against effluxing antibiotic-resistant Staphylococcus aureus. Int. J. Antimicrob. Agents 56, 106098. https://doi.org/10.1016/j.ijantimicag.2020.106098 (2020).
Popa, D.-S. & Rusu, M. E. Isoflavones: Vegetable sources, biological activity, and analytical methods for their assessment. (2017).
Górniak, I., Bartoszewski, R. & Króliczewski, J. Comprehensive review of antimicrobial activities of plant flavonoids. Phytochem. Rev. 18, 241–272. https://doi.org/10.2174/1568026622666220117104709 (2019).
Araya-Cloutier, C. Antibacterial prenylated isoflavonoids and stilbenoids: quantitative structure-activity relationships and mode of action, Wageningen University and Research (2017).
Yazaki, K., Sasaki, K. & Tsurumaru, Y. Prenylation of aromatic compounds, a key diversification of plant secondary metabolites. Phytochemistry 70, 1739–1745. https://doi.org/10.1016/j.phytochem.2009.08.023 (2009).
Kalli, S., Araya-Cloutier, C., de Bruijn, W. J. C., Chapman, J. & Vincken, J. P. Induction of promising antibacterial prenylated isoflavonoids from different subclasses by sequential elicitation of soybean. Phytochemistry 179, 112496. https://doi.org/10.1016/j.phytochem.2020.112496 (2020).
Palazzotti, D. et al. Deciphering the molecular recognition mechanism of multidrug resistance Staphylococcus aureus NorA efflux pump using a supervised molecular dynamics approach. Int. J. Mol. Sci. 20, 4041. https://doi.org/10.3390/ijms20164041 (2019).
Araya-Cloutier, C. et al. QSAR-based molecular signatures of prenylated (iso)flavonoids underlying antimicrobial potency against and membrane-disruption in Gram positive and Gram negative bacteria. Sci. Rep. 8, 1–14. https://doi.org/10.1038/s41598-018-27545-4 (2018).
Ahad, N. A. & Yahaya, S. S. S. in AIP Conference proceedings. 888–893 (American Institute of Physics).
Price, C. T., Kaatz, G. W. & Gustafson, J. The multidrug efflux pump NorA is not required for salicylate-induced reduction in drug accumulation by Staphylococcus aureus. Int. J. Antimicrob. Agents 20, 206–213. https://doi.org/10.1016/s0924-8579(02)00162-0 (2002).
Neyfakh, A. A., Borsch, C. & Kaatz, G. Fluoroquinolone resistance protein NorA of Staphylococcus aureus is a multidrug efflux transporter. Antimicrob. Agents Chemother. 37, 128–129. https://doi.org/10.1128/AAC.37.1.128 (1993).
Dengler, W. A., Schulte, J., Berger, D. P., Mertelsmann, R. & Fiebig, H. H. Development of a propidium iodide fluorescence assay for proliferation and cytotoxicity assays. Anticancer Drugs 6, 522–532. https://doi.org/10.1097/00001813-199508000-00005 (1995).
Stiefel, P., Schmidt-Emrich, S., Maniura-Weber, K. & Ren, Q. Critical aspects of using bacterial cell viability assays with the fluorophores SYTO9 and propidium iodide. BMC Microbiol. 15, 1–9 (2015).
Kirchhoff, C. & Cypionka, H. Propidium ion enters viable cells with high membrane potential during live-dead staining. J. Microbiol. Methods 142, 79–82. https://doi.org/10.1016/j.mimet.2017.09.011 (2017).
Blair, J. M. & Piddock, L. J. How to measure export via bacterial multidrug resistance efflux pumps. MBio 7, e00840-e1816. https://doi.org/10.1128/mBio.00840-16 (2016).
Paixão, L. et al. Fluorometric determination of ethidium bromide efflux kinetics in Escherichia coli. J. Biol. Eng. 3, 1–13. https://doi.org/10.1186/1754-1611-3-18 (2009).
Lomovskaya, O. et al. Identification and characterization of inhibitors of multidrug resistance efflux pumps in Pseudomonas aeruginosa: novel agents for combination therapy. Antimicrob. Agents Chemother. 45, 105–116. https://doi.org/10.1128/AAC.45.1.105-116.2001 (2001).
Opperman, T. J. et al. Characterization of a Novel Pyranopyridine Inhibitor of the AcrAB efflux pump of Escherichia coli. Antimicrob. Agents Chemother. 58, 722–733. https://doi.org/10.1128/AAC.01866-13 (2014).
Abreu, A. C. et al. Looking to nature for a new concept in antimicrobial treatments: Isoflavonoids from Cytisus striatus as antibiotic adjuvants against MRSA. Sci. Rep. 7, 1–16. https://doi.org/10.1038/s41598-017-03716-7 (2017).
Reynolds, E., Ross, J. I. & Cove, J. H. Msr(A) and related macrolide/streptogramin resistance determinants: Incomplete transporters?. Int. J. Antimicrob. Agents 22, 228–236. https://doi.org/10.1016/s0924-8579(03)00218-8 (2003).
Poole, K. Efflux pumps as antimicrobial resistance mechanisms. Ann. Med. 39, 162–176. https://doi.org/10.1080/07853890701195262 (2007).
Araya-Cloutier, C., Vincken, J. P., van Ederen, R., den Besten, H. M. W. & Gruppen, H. Rapid membrane permeabilization of Listeria monocytogenes and Escherichia coli induced by antibacterial prenylated phenolic compounds from legumes. Food Chem. 240, 147–155. https://doi.org/10.1016/j.foodchem.2017.07.074 (2018).
Abdelfatah, S. A. & Efferth, T. Cytotoxicity of the indole alkaloid reserpine from Rauwolfia serpentina against drug-resistant tumor cells. Phytomedicine 22, 308–318. https://doi.org/10.1016/j.phymed.2015.01.002 (2015).
Wikman-Larhed, A. & Artursson, P. Co-cultures of human intestinal goblet (HT29-H) and absorptive (Caco-2) cells for studies of drug and peptide absorption. Eur. J. Pharm. Sci. 3, 171–183. https://doi.org/10.1016/0928-0987(95)00007-Z (1995).
Bertrand, M., Jackson, P. & Walther, B. Rapid assessment of drug metabolism in the drug discovery process. Eur. J. Pharm. Sci. 11(2), S61-72. https://doi.org/10.1016/s0928-0987(00)00165-2 (2000).
Dutton, J. S., Hinman, S. S., Kim, R., Wang, Y. & Allbritton, N. L. Primary cell-derived intestinal models: Recapitulating physiology. Trends Biotechnol. 37, 744–760. https://doi.org/10.1016/j.tibtech.2018.12.001 (2019).
Takahashi, Y. et al. Organoid-derived intestinal epithelial cells are a suitable model for preclinical toxicology and pharmacokinetic studies. iScience 25, 104542. https://doi.org/10.1016/j.isci.2022.104542 (2022).
Kalli, S., Araya-Cloutier, C., Hageman, J. & Vincken, J. Insights into the molecular properties underlying antibacterial activity of prenylated (iso)flavonoids against MRSA. Sci. Rep. 11, 1–14. https://doi.org/10.1038/s41598-021-92964-9 (2022).
Li, J., Beuerman, R. W. & Verma, C. S. Molecular insights into the membrane affinities of model hydrophobes. ACS Omega 3, 2498–2507. https://doi.org/10.1021/acsomega.7b01759 (2018).
Brawley, D. N. et al. Structural basis for inhibition of the drug efflux pump NorA from Staphylococcus aureus. Nat. Chem. Biol. https://doi.org/10.1038/s41589-022-00994-9 (2022).
Van de Schans, M. G. et al. Involvement of a hydrophobic pocket and helix 11 in determining the modes of action of prenylated flavonoids and isoflavonoids in the human estrogen receptor. Chembiochem 16, 2668–2677. https://doi.org/10.1002/cbic.201500343 (2015).
Van De Schans, M. G. et al. Glyceollins and dehydroglyceollins isolated from soybean act as SERMs and ER subtype-selective phytoestrogens. J. Steroid Biochem. Mol. Biol. 156, 53–63. https://doi.org/10.1016/j.jsbmb.2015.11.020 (2016).
Andini, S., Araya-Cloutier, C., Waardenburg, L., den Besten, H. M. W. & Vincken, J. P. The interplay between antimicrobial activity and reactivity of isothiocyanates. Lwt-Food Sci. Technol. 134, 109843. https://doi.org/10.1016/j.lwt.2020.109843 (2020).
Coldham, N. G., Webber, M., Woodward, M. J. & Piddock, L. J. A 96-well plate fluorescence assay for assessment of cellular permeability and active efflux in Salmonella enterica serovar typhimurium and Escherichia coli. J. Antimicrob. Chemother. 65, 1655–1663. https://doi.org/10.1093/jac/dkq169 (2010).
Šimunović, K. et al. Efflux pump inhibition and resistance modulation in Mycobacterium smegmatis by Peucedanum ostruthium and Its Coumarins. Antibiotics 10, 1075. https://doi.org/10.3390/antibiotics10091075 (2021).
Zimmermann, S. et al. Clinically approved drugs inhibit the Staphylococcus aureus multidrug NorA efflux pump and reduce biofilm formation. Front. Microbiol. 10, 2762. https://doi.org/10.3389/fmicb.2019.02762 (2019).
van Dinteren, S. et al. Valorisation of liquorice (Glycyrrhiza) roots: Antimicrobial activity and cytotoxicity of prenylated (iso)flavonoids and chalcones from liquorice spent (G. glabra, G. inflata, and G. uralensis). Food Funct. 13, 12105–12120. https://doi.org/10.1039/D2FO02197H (2022).
Acknowledgements
We would like to thank Prof. Michael Rybak and Prof. Glenn W Kaatz (Wayne State University, USA) for providing us the S. aureus strains, Prof. Paolo Ruggerone (University of Cagliari, Italy) for his valuable feedback during discussions, and Hainan Wang, MSc for her contribution on performing antibiotic potentiation assays with erythromycin and norfloxacin against S. aureus 1199B and K-1758.
Funding
NWO (Nederlandse Organisatie voor Wetenschappelijk Onderzoek,Netherlands) with grant number VI.Veni.192.095.
Author information
Authors and Affiliations
Contributions
Conceptualization, writing (original draft preparation and editing), data curation, investigation and data analysis, methodology, M.I.I.; Funding acquisition, writing (review and editing), supervision, J.P.V.; Data curation, investigation, and data analysis, S.V.D.; Data curation, investigation, and data analysis, E.T.B.; Supervision, writing (review and editing), K.M.P.; Funding acquisition, supervision, conceptualization, writing (review and editing), C.A.C.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ika Irianti, M., Vincken, JP., van Dinteren, S. et al. Prenylated isoflavonoids from Fabaceae against the NorA efflux pump in Staphylococcus aureus. Sci Rep 13, 22548 (2023). https://doi.org/10.1038/s41598-023-48992-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-48992-8
- Springer Nature Limited