Abstract
Our main goal in this work is to synthesize valuable sulfonamide compounds according to the principles of green chemistry and also to present a unique convergent paired mechanism for their synthesis. In this study, we introduced a new type of convergent paired electro-organic synthesis of sulfonamide derivatives via a catalyst, oxidant, halogen and amine-free method. In this research, instead of using toxic amine compounds, an innovative mechanism based on the reduction of nitro compounds and in-situ production of amine compounds was used. The mechanism of electrophile generation is the cathodic reduction of the nitro compound to the hydroxylamine compound and then the anodic oxidation of the hydroxylamine to the nitroso compound. On the other hand, the nucleophile generation mechanism involves the two-electron oxidation of sulfonyl hydrazide to related sulfinic acid at the anode surface. The reaction leading to the synthesis of sulfonamides involves a one-pot reaction of the generated nitroso compound with the produced sulfinic compound.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Sulfonamides are known as valuable organic compounds with great biological and pharmaceutical activities1. They are widely used to develop the synthesis of new drugs and other pharmaceutically active components in medicinal and agricultural chemistry. Sulfonamide drugs have clinical applications such as antibacterial, anti-diabetic, anti-tuberculosis, anti-fungal antimalarial, etc2,3,4,5,6,7,8. In addition, they are used as additives in organic dyes to promote stability and lubricity9. To date, researchers have adopted various methods for the synthesis of sulfonamides and these efforts are still ongoing to provide a green method. As a general procedure, the synthesis of sulfonamides involves the coupling between a sulfonyl compound (often arenesulfonyl hydrazide and sodium arenesulfinate) and an amine (mostly of the primary and secondary types)10. The synthetic methods reported so far require special conditions and catalysts such as (I) the utilization of transition metals such as copper11,12, (II) the use of strong oxidants such as hydrogen peroxide (H2O2) or tert-butyl hydroperoxide (TBHP)13,14,15,16 and finally (III) In the presence of halogens such as iodine17. These methods suffered from some disadvantages such as the use of metals and oxidants as toxic/expensive reagents, harsh handling conditions, and the production of toxic waste; which is not in accordance with green chemistry principles18,19. In contrast, in electro-organic synthesis, electrons are used as green and cost-effective reactant, which has advantages such as simplicity, minimizing the production of toxic waste and energy consumption, and being eco-friendly10,20. Consequently, the demand for using metal catalysts and hazardous oxidants is eliminated21,22,23,24.
The unique features mentioned above were related to conventional electrosynthesis reactions. In these methods, when the desired electrochemical reaction is performed on the working electrode, the opposite electrochemical reaction occurs simultaneously at the counter electrode. The reactions that are carried out on the counter electrode are usually reduction or oxidation of the solvent, and their task is to consume the electrons produced in the working electrode or to supply the necessary electrons for the working electrode process. Now, if by changing the counter electrode reaction, to the production of the product or helps to produce the final product, the conventional electrosynthesis process has become “paired electrosynthesis”24,25. This method saves energy and time. Since in this method one mole of electrons can perform useful reactions in the working and counter electrodes and since the total current efficiency is the sum of the reactions that occur in each electrode, the current efficiency can increase up to 200% when cathodic and anodic processes produce the same product. Useful and complete information about the "paired electrosynthesis" can be obtained from the references24,25.
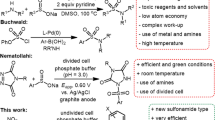
It should be noted that, in chemical synthesis, sulfnyl hydrazides are considered as an alternative to sulfinic acids because these compounds have advantages over other sulfonyl derivatives, including the fact that they are stable, non-corrosive, and moisture compatible20. Figure 1 compares the methods of synthesizing sulfonamides using sulfonyl hydrazides in recent years with the method proposed in this research.
The first two methods are very efficient chemical methods, but they have the disadvantage of using toxic reagents, expensive catalysts, high temperature and amine molecules26,27. Although the third method28, which is an electrochemical method, has solved most of the problems of the first two methods, it still has one problem: the use of amines which are toxic29 as a starting material. In the fourth method, sulfonyl hydrazide is not used and it seems that it should not be here, but because of its unique features and the lack of use of toxic amine, it is included here for comparison with the results of this research30,31. But, in this work, we presented a new strategy based on the activation of sulfonyl hydrazides and nitro compounds at the anode and the cathode without using any kind of catalyst or oxidant. It should be noted that so far no report has been published regarding the chemical or electrochemical reaction of nitro compounds and sulfonyl hydrazides.
In this method, by reducing the nitro compound to hydroxylamine on the cathode surface and then the oxidation of the formed hydroxylamine on the anode surface to the nitroso compound (electrophile formation process) on the one hand, and the oxidation of sulfonyl hydrazide to sulfonic acid on the anode surface (nucleophile formation process) on the other hand, the conditions for their reaction in water and the synthesis of sulfonamides are provided. In this method, the only by-product produced is nitrogen gas, which is a safe by-product. Replacing amines with nitro compounds minimizes the production of toxic waste products, which has created more favourable conditions in terms of green chemistry32.
Experimental data
Reagents and apparatus
All voltammetric experiments including cyclic voltammetry and controlled potential coulometry were carried out by a µ-Autolab model PGSTAT 20 potentiostat/galvanostat (Metrohm-Autolab, Netherland) and nova 1.10 software was used to control of the µ-Autolab instruments with USB connection and collect the data. For electrochemical studies, a three-electrode system in an undivided cell (all electrodes from AZAR Electrodes) consisting of a glassy carbon (GC) disc as a working electrode (2.8 mm diameter), platinum wire as the counter electrode, and Ag/AgCl (3 M) as the reference electrode was used. In macroscale electrolysis, cathode electrodes that are used were the assembly of four carbon rods with a total surface area of 32 cm2, while in the case of the anode, it consisted of a carbon rod (8 cm2 area). It should be noted that before each voltammetric experiment, the GC electrode was polished using alumina slurry and cleaned with acetone. Subsequently, it was completely rinsed with distilled water. Nitrobenzene (NB1), p-nitrotoluene (NB2), acetonitrile, phosphoric acid, n-hexane, and ethyl acetate were purchased from Sigma-Aldrich Company and were used as received, without further purification. Silica gel (SiO2, 60 Å silica gel, Merck Grade, 70–230 mesh) was obtained from Merck Company. p-Chloronitrobenzene (NB3) and p-Iodonitrobenzene (NB4) were synthesized according to previously described methods33,34. The FTIR spectra of products were recorded in the range 500–4000 cm−1 using a Perkin‐Elmer 1760. The 1H and 13C NMR spectra were recorded in CDCl3 on a Bruker Avance 400 MHz spectrometer. Mass spectrometry analysis was performed in electron impact mode at an ionization potential of 70 eV using an Agilent-5973 mass spectrometer. The melting point of the products was determined using a Barnstead Electrothermal 9100 instrument.
Synthetic procedures for arenesulfonyl hydrazides
Benzenesulfononyl hydrazide (SH1) and p-Toluenesulfonyl hydrazide (SH2) were synthesized according to the previous published method35. Briefly, 10 mmol of 4-methyl benzenesulfonyl chloride or benzenesulfonyl chloride was poured into a 25 mL beaker. In the following, 20 mL of THF solvent was added to it and stirred for 5 min at room temperature (RT) until it completely dissolved. On the other hand, in a 50 mL bottom flask, 30 mmol of hydrazine was poured and placed at a temperature of 0–5 °C. The prepared solution was added drop by drop to the flask containing hydrazine and after that, the mixture was stirred for 30 min at a temperature of 0–5 °C. After this time, the mixture was stirred for another 1 h at room temperature. After the completion of the reaction, water and ethyl acetate were added to the reaction mixture, and after decanting, the organic layer was separated and sodium sulphate was added to it. In the last step, the organic layer was filtered and its solvent was evaporated. A pure white product was obtained (product yield is more than 95%).
Electrochemical synthesis of sulfonamide derivatives
A mixture of arensulfonyl hydrazide (0.5 mmol) and nitroaren (0.5 mmol) was electrolyzed at − 0.6 V versus Ag/AgCl in an undivided cell containing a solution (ca. 80 mL) of water (phosphate buffer, pH = 2.0, c = 0.2 M)/acetonitrile (50/50 v/v) at room temperature (RT). The electrolysis was terminated when the decay of the current became more than 95%. At the end of electrolysis, after extracting the reaction solution with ethyl acetate, the organic layer was allowed to dry overnight. Purification was performed using thin layer chromatography (eluent 4:1 n-hexane/ethyl acetate) to afford the sulfonamide as the product. The final product was characterized by H-NMR, FTIR and mass spectroscopy (Supplementary Information). The structures of known sulfonamides were confirmed by the comparison of their spectroscopic data with those previously reported. It should be noted that acetonitrile has been used as a co-solvent for the complete dissolution of the required precursors.
Characteristics of the products
N-Hydroxy-4-methyl-N-phenylbenzenesulfonamide (SU1): Pale yellow; MP = 137–139 °C (Lit. 139–141 °C)30; IR (KBr, cm−1): 3377 (medium, O–H), 2856 (weak, CH3), 1590 (medium C=C), 1447, 1347, 1172 (strong, S=O). 1H-NMR (400 MHz, DCl3) δ 7.40 (d, J = 8.3 Hz, 2H), 7.24 (d, J = 0.9 Hz, 2H), 7.22 (d, J = 3.1 Hz, 2H), 7.20 (s, 1H), 7.17–7.13 (m, 2H), 2.42 (s, 3H).
N-Hydroxy-4-methyl-N-(p-tolyl)benzenesulfonamide (SU2): Pale yellow; MP: 88–90 °C, IR (KBr, cm−1): 3360 (medium, O–H), 2922 (weak, CH3) 1596 (medium C=C), 1452, 1338 and 1162 (strong, S=O). MS (m/z) (EI, 70 eV) (relative intensity): 91.2 (95), 155.2 (75), 276.1 (100). 1H-NMR (400 MHz, CDCl3) δ 7.43 (d, J = 8.0 Hz, 2H), 7.22 (d, J = 7.9 Hz, 2H), 7.03 (q, J = 8.5 Hz, 4H), 2.42 (s, 3H), 2.32 (s, 3H).
N-(4-Chlorophenyl)-N-hydroxy-4-methylbenzenesulfonamide (SU3): Pale yellow; MP: 125–127 °C (Lit. 129–130 °C)27; IR (KBr, cm−1): 3347 (medium, O–H), 2923 (weak, CH3), 1595 (medium C=C), 1482, 1343 and 1163 (strong, S=O). 1H-NMR (400 MHz, CDCl3) δ 7.42 (d, J = 8.1 Hz, 2H), 7.24 (d, J = 2.9 Hz, 2H), 7.22 (d, J = 3.7 Hz, 2H), 7.10 (d, J = 8.8 Hz, 2H), 2.42 (s, 3H).
N-Hydroxy-N-phenylbenzenesulfonamide (SU4): MP = 129–131 °C (Lit. 133–134 °C)30; IR (KBr, cm−1): 3376 (medium, O–H), 1590 (medium C=C), 1486, 1347, 1172 (strong, S=O). 1H-NMR (400 MHz, CDCl3) δ 7.62 (t, J = 7.4 Hz, 1H), 7.54 (d, J = 7.6 Hz, 2H), 7.42 (t, J = 7.7 Hz, 2H), 7.25 (d, J = 6.6 Hz, 2H), 7.17–7.12 (m, 3H).
N-(4-Chlorophenyl)-N-hydroxybenzenesulfonamide (SU5): Pale yellow; MP: 101–103 °C (Lit. 102–104 °C)27; IR (KBr, cm−1): 3370 (medium, O–H), 1578 (medium C=C), 1484, 1347 and 1170 (strong, S=O). 1H-NMR (400 MHz, CDCl3) δ 7.56–7.48 (m, 1H), 7.43 (d, J = 7.4 Hz, 2H), 7.37–7.29 (m, 2H), 7.11 (d, J = 8.3 Hz, 2H), 6.98 (d, J = 8.3 Hz, 2H).
N-Hydroxy-N-(4-iodophenyl)benzenesulfonamide (SU6): Pale yellow; MP: 106–109 °C (Lit. 108–110 °C)27; IR (KBr, cm−1): 3339 (medium, O–H), 1446 (medium C=C), 1340 and 1166 (strong, S=O). 1H-NMR (400 MHz, CDCl3) δ 7.63 (d, J = 7.4 Hz, 2H), 7.55 (d, J = 9.1 Hz, 2H), 7.48–7.41 (m, 3H), 6.90 (d, J = 8.6 Hz, 2H).
Results and discussion
To study the electrochemical behavior of p-nitrotoluene (NB2), cyclic voltammogram (CV) of NB2 at a glassy carbon electrode, in aqueous phosphate buffer (pH = 2.0, c = 0.2 M)/acetonitrile (50/50 v/v) at scan rate of 100 mV s−1 is shown in Fig. 2, part I. When the cathodic scan is performed first, the voltammogram consists of an irreversible well defined cathodic peak (C0) at − 0.60 V versus Ag/AgCl attributed to a four-electron reduction of the nitro group of NB2 to hydroxylamine group in the forward scan, and a reversible couple (A1/C1) at 0.19 and 0.12 V versus Ag/AgCl, respectively, in the reverse scan. The anodic peak of A1 corresponds to the oxidation of cathodically generated N-(p-tolyl)hydroxylamine (HA2) to related nitroso compound, p-nitrosotoluene (NS2) and its cathodic counterpart (C1) which is related to the reduction of NS2 to HA2 (Fig. 3)30,31. It should be noted that the existence of peaks A1 and C1 strongly depends on the direction of the potential scan. So when the potential is initially scanned from 0.0 to 0.5 V versus Ag/AgCl, none of the peaks A1 and C1 are observed.
Part I: Cyclic voltammogram of 1.0 mM NB2 in aqueous phosphate buffer (pH, 2.0, c = 0.2 M)/acetonitrile (50/50 v/v). Scan rate: 100 mV/s at room temperature. Part II: Cyclic voltammograms of HA2/NS2 redox couple (1.0 mM) in aqueous buffer/acetonitrile (50/50, v/v) mixture with different pH values and same ion strength at scan rate of 100 mV s−1. The pH of buffer solutions from (a) to (f) are 2.2, 2.7, 4.0, 4.9, 6.0 and 7.2, respectively. Part III: Pourbaix diagram of HA2/NS2 redox couple. Working electrode: Glassy carbon electrode. All experiments are performed at room temperature. These figures were prepared by Microsoft Excel (OFFICE 2013).
Part I: The redox behavior observed in Fig. 2. Part II: The oxidation mechanism of SH2. Part III: The reaction of produced nucleophile with the generated electrophile.
In the next part of this work, the effect of pH on the HA2/NS2 redox couple was investigated in detail by cyclic voltammetry (Fig. 2, part II). The results showed that E1/2 (half-wave potential) of HA2/NS2 redox couple is pH-dependent and changes to less positive potentials with increasing pH. This shift confirms the presence of the proton(s) in the HA2/NS2 redox process as shown in Fig. 3, part I. Based on this, the Pourbaix diagram related to the HA2/NS2 redox couple has been reported in the range of pH 2 to 7 (Fig. 2, part III). In this pH range, the plot shows a straight line with a slope of 0.053 mV/pH, which corresponds to the theoretical value (0.059 mV/pH) for the two-electron/two-proton process.
To obtain more details on the electrochemical properties of HA2 produced by NB2 reduction, the effect of the potential scan rate on the cyclic voltammogram of NB2 has been investigated (Fig. 4). The findings show that the peak current ratio (IpC1/IpA1) depends on the scan rate. This ratio increases with increasing scan rate, which indicates the occurrence of a chemical reaction after the electron transfer step. In addition, adsorption-diffusion behavior of HA2 was investigated. To achieve this goal, the log IpA1 was plotted against log scan rate (log v) at pH = 2.0. The results lead to a straight line with a slope of 0.65 (Fig. 4). This slope is lower than the theoretical value of 1 for the adsorption-controlled process and higher than the theoretical value of 0.5 for the diffusion-controlled process. Accordingly, adsorption-diffusion behavior has been proposed for HA2 oxidation process at a glassy carbon electrode in aqueous phosphate buffer (pH, 2.0, c = 0.2 M)/acetonitrile (50/50 v/v) mixture. These results show that the oxidation process of hydroxylamine (HA) on the anode surface is not purely adsorptive and therefore the amount of HA on the anode surface during the electrolysis depends on various parameters such as the solution stirring speed, anode–cathode distance, solution viscosity, etc.
Left: The effect of potential scan rate on the cyclic voltammograms NB2 (1.0 mM) in water (phosphate buffer, pH, 2.0, c = 0.2 M)/acetonitrile (50/50, v/v) mixture. Scan rates from (a) to (f) are: 10, 25, 50, 100, 250 and 500 mV s−1. Right: The plot of log IpA1 versus log ν at glassy carbon electrode at room temperature.
Cyclic voltammogram of p-toluenesulfonyl hydrazide (SH2) was shown in Fig. 5, part I, curve a and inset. SH2 has an irreversible anodic peak at 0.62 V versus Ag/AgCl, attributed to two-electron oxidation of SH2 to p-toluenesulfinic acid (SA2) according to Fig. 3, part II36. In the next steps, we use this compound (SA2) as a nucleophile. In almost all electrochemical syntheses, nucleophiles are generated at the cathode by electron injection from cathode to a molecule. This study is one of the few studies in which the generation of nucleophile has been performed at the anode by taking electrons from the molecule. In this Figure, curve b is the cyclic voltammogram of NB2, which was recorded under the same conditions as Fig. 2. Unlike individual voltammograms a and b, under similar conditions, the cyclic voltammogram of NB2 in the presence of SH2 is shown in curve c. This voltammogram is recorded in such a way that SH2 is also oxidized in the anodic scan (switching anodic potential more than EpA2). The effect of oxidized form of SH2 (SA2) on the cyclic voltammogram of NB2 is shown in Fig. 5, part I, curve c. The disappearance of the cathodic peak C1 confirms the chemical reaction between SA2 and NS2.
Part I: (a and inset) Cyclic voltammogram of SH2 (1.0 mM). (b) Cyclic voltammogram of 1.0 mM of NB2 in the absence of SH2. (c) Cyclic voltammogram of 1.0 mM of NB2 in the presence of SH2 (1.0 mM). Part II: Cyclic voltammograms of 1.0 mM of NB2 in the presence of SH2 (1.0 mM) (a) Anodic switching potential + 0.35 V versus Ag/AgCl. (b) Anodic switching potential + 0.90 V versus Ag/AgCl. Solvent: water (phosphate buffer, pH, 2.0, c = 0.2 M) /acetonitrile (50/50, v/v) mixture. Scan rate: 100 mV s−1. Working electrode: Glassy carbon electrode. All experiments are performed at room temperature. These figures were prepared by Microsoft Excel (OFFICE 2013).
To confirm the necessity of oxidation of SH2 to create an active nucleophile (SA2) for reaction with NS2, in another experiment the anodic scan of NB2 in the presence of SH2 was limited to + 0.35 V (before EpA2). The cyclic voltammogram of NB2 in the presence of SH2 with a truncated anodic switching potential (ASP) is shown in Fig. 5, part II, curve a and is compared with the cyclic voltammogram of NB2 in the presence of SH2 with a higher anodic scan range (EASP = 0.90 V) (curve b). The comparison of two voltammograms (a and b) clearly shows that the removal of peak C1 occurs when the anodic switching potential becomes more positive than the EpA2. In other words, the presence of SA2 on the surface of the electrode is necessary for the reaction with NS2.
Based on the obtained electrochemical results as well as the identification of the structure of the final products obtained from the electrolysis of NB in the presence of SH, the following mechanism is proposed for the synthesis of sulfonamides SU1-6 (Fig. 6). According to Fig. 6, in the first step, the NO2 group of NB is reduced at the cathode to the hydroxylamine group by taking four electrons. In the next step, the hydroxylamine compound (HA) is oxidized at the anode to form the corresponding nitroso compound (NS) by losing two electrons and two protons. We refer to these two stages as the “electrophilic generation step”. On the other hand, the sulfonyl hydrazide (SH) is oxidized at the anode by losing two electrons, two protons, and releasing a N2 molecule to the corresponding sulfinic acid (SA). We refer to this stage as the "nucleophilic formation step". In the final stage, the produced nucleophile reacts with the generated electrophile to produce the final product (SU1-6) (Fig. 3, part III).
To improve the yield and purity of the synthesized sulfonamides, the amount of charge passing through the electrolytic cell was optimized. The condition of the experiment are as follows: applied potential: − 0.7 V versus Ag/AgCl, NB2 amount: 0.5 mmol, SH2 amount: 0.5 mmol, solvent: water (0.2 M phosphate buffer, pH = 2.0)/acetonitrile (50/50 v/v) mixture, anode: carbon and cathode: carbon. The results have been shown in Fig. 7. As can be seen, the product yield increases as the passing charge increases in the range of 2–5 F mol−1, but above this range, the yield of the product will be decreased. According to Fig. 6, the theoretical amount of electricity required for the synthesis of sulfonamides SU1-6 is 4 F mol−1. Therefore, it can be expected that with the increase in the amount of electricity consumed, the yield will also increase, and reach the maximum. Nevertheless, since the current efficiency in this cell is not 100%, the maximum efficiency is obtained with the consumption of 5 F mol−1. The decrease in current efficiency from 100 is due to the necessity of using simple cell (undivided cell) and the occurrence of back reactions such as the reduction of NS in the cathode to HA. Based on the yield obtained at 4 F mol−1 and 5 F mol−1, the current efficiency and the amount of back reaction(s) in this cell can be considered equal to 52% and 23%, respectively. A further increase in electricity consumption causes a decrease in yield due to the occurrence of side reactions such as over-oxidation of the final product (SU).
Effect of the charge passed on the yield of SU2. NB2 amount: 0.5 mmol, SH2 amount: 0.5 mmol, solvent: water (0.2 M phosphate buffer, pH = 2.0)/acetonitrile (50/50 v/v) mixture, applied potential: − 0.7 V versus Ag/AgCl. Anode: carbon and cathode: carbon. All experiments are performed at room temperature. These figures were prepared by Microsoft Excel (OFFICE 2013).
Also, the effect of electrode type on the yield of the desired products was investigated. The results have been shown in Table 1 As can be seen, the highest yield is achieved when both anode and cathode are made of carbon.
Under optimized conditions, we investigated the scope of the reaction using arenesulfonyl hydrazides (SH1 and SH2) and some nitroarenes (NB1-4) (Table 2). The results show that we have succeeded in devising an ingenious method for the synthesis of sulphonamides SU1-6 in a one-pot reaction with an overall yield of 59–68%. No data have been reported so far for the electrochemically synthesis of these sulfonamides. The reaction is carried out under mild conditions without using oxidizing or agents or catalysts at room temperature and atmospheric pressure.
Conclusions
For the first time, we reported the electrochemical synthesis of sulfonamide compounds, with a new ping-pong type reaction mechanism. In this method, the activation of nitroarene and arylsulfonyl hydrazide as starting materials was necessary. In the first stage, the nitro group at the cathode surface was reduced to the hydroxylamine group and then it is oxidized at the anode surface to the corresponding nitroso compound (NS). On the other hand, SH compounds are oxidized to the corresponding sulfinic compounds at the anode surface. The chemical reaction between the two electrochemically produced precursors (SA and NS) leads to the corresponding sulfonamide. This procedure has many advantages over other reported methods such as: (I) Use of nitro compounds instead of toxic amine compounds, (II) Using electrons as a cost-effective and green oxidant instead of using chemical oxidants, (III) No need to use a catalyst, (IV) High current efficiency and low energy consumption. In convergent strategy, under ideal conditions, a current efficiency of 200% is achievable. Although our experiments were performed on a relatively small scale, there is little difficulty in producing larger quantities either by using larger cells or by running several cells in series.
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information file.
References
Buathongjan, C., Beukeaw, D. & Yotphan, S. Iodine-catalyzed oxidative amination of sodium sulfinates: A convenient approach to the synthesis of sulfonamides under mild conditions. Eur. J. Org. Chem. 2015, 1575–1582 (2015).
Almalki, A. J. et al. Synthesis, antimicrobial, anti-virulence and anticancer evaluation of new 5(4H)-oxazolone-based sulfonamides. Molecules 27, 671 (2022).
Durgun, M. et al. Synthesis, characterization, in vitro cytotoxicity and antimicrobial investigation and evaluation of physicochemical properties of novel 4-(2-methylacetamide) benzenesulfonamide derivatives. Bioorg. Chem. 70, 163–172 (2017).
Supuran, C. T., Casini, A. & Scozzafava, A. Protease inhibitors of the sulfonamide type: Anticancer, antiinflammatory, and antiviral agents. Med. Res. Rev. 23, 535–558 (2003).
Alaoui, S. et al. Synthesis and anti-cancer activities of new sulfonamides 4-substituted-triazolyl nucleosides. Bioorg. Med. Chem. Lett. 27, 1989–1992 (2017).
Dai, H.-X., Stepan, A. F., Plummer, M. S., Zhang, Y.-H. & Yu, J.-Q. Divergent C-H functionalizations directed by sulfonamide pharmacophores: Late-stage diversification as a tool for drug discovery. J. Am. Chem. Soc. 133, 7222–7228 (2011).
Lal, J., Gupta, S. K., Thavaselvam, D. & Agarwal, D. D. Biological activity, design, synthesis and structure activity relationship of some novel derivatives of curcumin containing sulphonamides. Eur. J. Med. Chem. 64, 579–588 (2013).
Qin, H. L., Zhang, Z.-W., Lekkala, R., Alsulami, H. & Rakesh, K. Chalcone hybrids as privileged scaffolds in antimalarial drug discovery: A key review. Eur. J. Med. Chem. 193, 112215 (2020).
Sayed, A. Z. & El-Gaby, M. S. Synthesis of novel dyestuffs containing sulphonamido moieties and their application on wool and polyamide fibres. Color. Technol. 117, 293–297 (2001).
Jamshidi, M., Amani, A., Khazalpour, S., Torabi, S. & Nematollahi, D. Progress and perspectives of electrochemical insights for C–H and N–H sulfonylation. New J. Chem. 45, 18246–18267 (2021).
Tang, X. et al. Copper-catalyzed sulfonamides formation from sodium sulfinates and amines. Chem. Commun. 49, 6102–6104 (2013).
Yotphan, S. et al. Iodine-catalyzed expeditious synthesis of sulfonamides from sulfonyl hydrazides and amines. Org. Biomol. Chem. 14, 590–597 (2016).
Ji, J., Liu, Z., Liu, P. & Sun, P. Synthesis of sulfonamides via copper-catalyzed oxidative C–N bond cleavage of tertiary amines. Org. Biomol. Chem. 14, 7018–7023 (2016).
Wei, W. et al. Metal-free direct construction of sulfonamides via iodine- mediated coupling reaction of sodium sulfinates and amines at room temperature. Adv. Synth. Catal. 357, 987–992 (2015).
Yu, H. & Zhang, Y. NH4I-catalyzed synthesis of sulfonamides from arylsufonylhydrazides and amines. Chin. J. Chem. 34, 359–362 (2016).
Parumala, S. K. R. & Peddinti, R. K. Metal-free synthesis of sulfonamides via iodine-catalyzed oxidative coupling of sulfonyl hydrazides and amines. Tetrahedron Lett. 57, 1232–1235 (2016).
Yang, K., Ke, M., Lin, Y. & Song, Q. Sulfonamide formation from sodium sulfinates and amines or ammonia under metal-free conditions at ambient temperature. Green Chem. 17, 1395–1399 (2015).
Yuan, Y. et al. Independent phase modulation for quadruplex polarization channels enabled by chirality-assisted geometric-phase metasurfaces. Nat. Commun. 11, 4186 (2020).
Pandit, S. S., Pandit, V. U. & Bandgar, B. P. Rapid and efficient synthesis of sulfonamides from sulfonic acid and amines using cyanuric chloride-DMF adduct. J. Sulphur Chem. 29, 619–622 (2008).
Pollok, D. & Waldvogel, S. R. Electro-organic synthesis: A 21st century technique. Chem. Sci. 11, 12386–12400 (2020).
Sadatnabi, A., Mohamadighader, N. & Nematollahi, D. Convergent paired electrochemical synthesis of azoxy and azo compounds: An insight into the reaction mechanism. Org. Lett. 23, 6488–6493 (2021).
Goljani, H., Tavakkoli, Z., Sadatnabi, A., Masoudi-Khoram, M. & Nematollahi, D. A new electrochemical strategy for the synthesis of a new type of sulfonamide derivatives. Sci. Rep. 10, 17904 (2020).
Sadatnabi, A. & Nematollahi, D. An eco-friendly strategy using a double-current two-phase cell system for electrografting of polyacrylic acid. J. Electroanal. Chem. 927, 116991 (2022).
Aslam, S. et al. Heterocyclic electrochemistry: Renewable electricity in the construction of heterocycles. ACS Omega 8(7), 6175–6217 (2023).
Sbei, N., Hardwick, T. & Ahmed, N. Electrochemical organic transformations via paired electrolysis. ACS Sustain. Chem. Eng. 9, 6148–616918 (2021).
Chung, S. & Kim, J. Cu-catalyzed aerobic oxidative synthesis of sulfonamides from sulfonyl hydrazides and amines. Tetrahedron Lett. 60, 792–795 (2019).
Chen, J. et al. Iodine-catalyzed sulfonylation of sulfonyl hydrazides with tert-amines: A green and efficient protocol for the synthesis of sulphonamides. RSC Adv. 9, 31212–31216 (2019).
Sheykhan, M. et al. An approach to C–N activation: Coupling of arenesulfonyl hydrazides and arenesulfonyl chlorides with tert-amines via a metal-, oxidant- and halogen-free anodic oxidation. Green Chem. 19, 5940–5948 (2017).
Poste, A. E., Grung, M. & Wright, R. F. Amines and amine-related compounds in surface waters: A review of sources, concentrations and aquatic toxicity. Sci. Total Environ. 481, 274–279 (2014).
Mokhtari, B., Nematollahi, D. & Salehzadeh, H. Paired electrochemical conversion of nitroarenes to sulfonamides, diarylsulfones and bis (arylsulfonyl) aminophenols. Green Chem. 20, 1499–1505 (2018).
Mokhtari, B., Nematollahi, D. & Salehzadeh, H. A tunable pair electrochemical strategy for the synthesis of new benzenesulfonamide derivatives. Sci. Rep. 9, 4537 (2019).
Hauk, A., Richartz, H., Schramm, K. W. & Fiedler, H. Reduction of nitrated phenols: A method to predict half-wave-potentials of nitrated phenols with molecular modelling. Chemosphere 20, 717–728 (1990).
Sandmeyer, T. Ueber die Ersetzung der Amid-gruppe durch Chlor, Brom und Cyan in den aromatischen Substanzen. Ber. Dtsch. Chem. Ges. 17, 2650–2653 (1884).
Goljani, H., Tavakkoli, Z., Sadatnabi, A. & Nematollahi, D. Two-phase electrochemical generation of aryldiazonium salts: Application in electrogenerated copper-catalyzed sandmeyer reactions. Org. Lett. 22, 5920–5924 (2020).
Alford, E. & Schofield, K. Cinnolines. Part XXX. The nature of the C (3)-position. The synthesis of 3-methyl-, 3-chloro-, and 3-bromo-cinnoline; also, of cinnoline and the Bz-nitrocinnolines. J. Chem. Soc. (Resumed) https://doi.org/10.1039/JR9530000609 (1953).
Li, W. et al. Catalyst-free synthesis of 3-sulfone nitrile from sulfonyl hydrazides and acrylonitrile in water. Org. Biomol. Chem. 15, 7819–7823 (2017).
Acknowledgements
The authors wish to acknowledge Iran National Science Foundation (INSF) for financial support of this work. The authors also acknowledge the Bu-Ali Sina University Research Council and Center of Excellence in Development of Environmentally Friendly Methods for Chemical Synthesis (CEDEFMCS) for their support of this work.
Author information
Authors and Affiliations
Contributions
P.P. investigation, conceptualization, methodology, writing—original draft. A.S. investigation, conceptualization, methodology, writing—original draft. D.N. writing—review & editing, supervision, project administration.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Patoghi, P., Sadatnabi, A. & Nematollahi, D. A new type of convergent paired electrochemical synthesis of sulfonamides under green and catalyst-free conditions. Sci Rep 13, 17582 (2023). https://doi.org/10.1038/s41598-023-44912-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-44912-y
- Springer Nature Limited