Abstract
Investigation of a unique and fast method for the determination and separation of a mixture of three drugs viz., ciprofloxacin (CIP), Ibuprofen (IBU), and diclofenac sodium (DIC) in actual samples of human plasma. Also, the technique was used to look at their pharmacokinetics study. Hydrocortisone was chosen as the internal standard (IS). The drugs were chromatographically separated using an Acquity ultra-performance liquid chromatography UPLC ® BEH C18 1.7 µm (2.1 × 150 mm) column with a mobile phase composed of acetonitrile: water (65:35, v/v) adjusted to pH 3 with diluted acetic acid. Plasma proteins were precipitated with acetonitrile. The separated drugs ranged from 0.3 to 10, 0.2–11, and 1–25 µg/mL for CIP, IBU, and DIC, respectively. Calibration curves were discovered to achieve linearity with acceptable correlation coefficients (0.99%). Examination of quality assurance samples showed exceptional precision and accuracy. Following the successful application of this improved technique to plasma samples, the pharmacokinetic characteristics of each selected drug were evaluated using (UPLC) with UV detection at 210 nm. Two green metrics were applied, the Analytical Eco-scale and the Analytical GREEnness Calculator (AGREE). Separation was achieved in only 4-min analysis time. The method's validation agreed with the requirements of the FDA, and the results were sufficient.
Similar content being viewed by others
Introduction
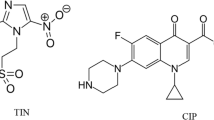
CIP (1-cyclopropyl-6-fluoro-4-oxo-7-piperazin-1-ylquinoline-3-carboxylic acid) (Fig. 1a) is a fluoroquinolone antibiotic that is of the second generation and is widely used to overcome respiratory diseases and urinary tracts in mild to severe severity1,2.
DIC (2-[2-(2,6-dichloroanilino) phenyl] acetic acid) (Fig. 1b) is the salt of sodium version of DIC, a non-steroid anti-inflammatory medication (NSAID) has analgesic, antipyretic, and anti-inflammatory properties and is produced from benzene acetic acid. Diclofenac sodium decreases cyclooxygenase (COX) in a non-selective, reversible, and competitive manner, blocking the formation of prostaglandin precursors from arachidonic acid. As a result, prostaglandin production related to pain, inflammation, and fever is inhibited3.
IBU [2-(4-isobutyl phenyl) propionic acid], naproxen [6-methoxy-α-methyl-2-naphthalene acetic acid,] and tolmetin [1-methyl-5-(4-methyl benzoyl)-1H-pyrrole-2-acetic acid] Fig. 1c are non-steroid anti-inflammatory medicines (NSAIDs) that show favorable anti-inflammatory and analgesic characteristics4.
UPLC is a technique intended to improve sensitivity and accuracy while reducing solvent usage and processing time5. The detection of CIP in biological fluids has been the subject of numerous HPLC methods documented in the literature6,7,8,9,10. Liang et al., 2002 published a paper using reversed-phase HPLC with fluorescence and UV detection to separate and determine CIP and another five drugs in human plasma11. Few HPLC procedures were found to determine CIP, DIC, and IBU simultaneously. However, the number of techniques for separation is registered, and various medications for pain relief, inflammation, and infections are determined12. Green analytical chemistry aims to make it less dangerous, eco-friendly, and more economical for analysts. The bases of analytical chemistry greenness include reducing solvents, using ecologically friendly and low-toxic chemicals, decreasing waste output, analysis duration, and energy usage, and increasing integration, breakdown, and ease of use of analytical tools and procedures13.
The parts of chemical steps that follow the green chemistry principles are described in green chemistry metrics. Metrics are used to evaluate chemical processes' efficiency or environmental performance and measure performance improvements. In recent years, several green metrics have been created to assess how well analytical methods correspond to the green analytical chemistry principles. The national environmental metric index (NEMI) is one example of a qualitative tool14, analytical Eco-scale and green analytical procedure index GAPI15,16 are semi-quantitative, and AGREE (Analytical GREEnness Metric Approach and Software)17 is quantitative.
Separation of the drugs is carried out using UPLC. This study aims to create a unique, fast separation and determination procedure for CIP, IBU, and DIC in pharmaceutical formulations. The investigation' aim was increased to look at the three drugs' pharmacokinetic characteristics. The method's validation agreed with the requirements of the FDA, and the results were sufficient. According to all outcomes, the novel approach is the best in terms of making fixed dose combination available for the three drugs together, how it affects the environment and people's life and how much it costs to analyze data and prepare samples.
Materials and methods
Instruments
Dionex Ultimate 3000 UHPLC. It had an autosampler, a diode array detector, and a tetra solvent delivery pump (Massachusetts, USA). An X-Bridge Acquity UPLC ® BEH C18 1.7µm (2.1 × 150 mm) column was used for separation and quantification. The mobile phase and samples were filtered using 0.22 µm MS disposable syringe filters and 0.22 µm Millipore nylon membrane filters. The mobile phase was a mix of acetonitrile and water (pH 3 by diluted acetic acid). The separation system was 50% acetonitrile − 50% H2O, with a 0.2 mL/min flow rate. During the experiment, a Jenway pH meter was for adjusting the pH of the mobile phase (model 3510, Staffordshire, UK). Sonix TV SS-series ultrasonicator (South Carolina, USA) and a digital balance from Sartorius (Germany) were employed. The mixture was detected at 210 nm. The injection volume was 5 µL and analyzed for 4 min. Also, the heat of the column was 25 °C. A rongtai micropipette with a volume range of 0.1–500.0 µL (Mainland, Shanghai, China). 4000 rpm variable speed centrifuge (Zjmzym, China) with 8 × 20 mL capacity.
Drugs and reagents
Pure samples of the drugs DIC, IBU, and CIB were kindly offered by native companies, viz., Mina-pharm (10th of Ramadan, Egypt), Abbott Healthcare for the Pharmaceutical Industries (90Th Street, New Cairo, Cairo), and ORGANOPHARM for pharmaceutical and chemical industries (Obour City, Egypt), respectively. EPICO for Pharmaceutical Industries kindly offered a pure hydrocortisone sample.
Diclac 75 mg tablets, produced by SANDOZI (Mina-pharm company, Egypt, under license from Hexal AG, Germany). CIP 750 mg tablets made by ORGANOPHARM for pharmaceutical and chemical industries (Obour city, Egypt) and Brufen 600 mg tablets produced by Abbott Healthcare for the Pharmaceutical Industries (90th Street, New Cairo, Cairo), were purchased from the local market. Sigma Aldrich provided HPLC-grade acetonitrile and acetic acid. Plasma samples from the volunteers were obtained to create the calibration curves before the suggested drugs were administered (Approval No: NUB-034-023 by The Committee of Nahda University, Beni-Suef, Faculty of Pharmacy has approved the study protocol from the ethical point of view).
Solutions and calibration samples
From the 1000 µg/mL stock solutions of each CIP, DIC, IBU, and hydrocortisone (IS), 2.5 mL were transferred to 25-mL volumetric flasks. Each is completed to volume with a diluent solution of acetonitrile and H2O (pH 3 by diluted acetic acid) (50/50, v/v) to obtain 100 µg/mL of each.
To construct the calibration curves for each drug, three sets of 10-mL measuring flasks were provided with aliquots of the later working solutions of each of CIP, IBU, and DIC covering the concentrations ranging from 0.3 to 10, 0.2–11, and 1–25 µg/mL, respectively, using the produced working standard solutions (100 µg/mL) and addition of 0.5 mL of the working standard solution of hydrocortisone (IS) to all the samples. Then, a solution composed of a 50/50 v/v mixture of acetonitrile and water was used to complete all flasks to the mark.
Standards for calibration were prepared in-vitro by transferring known concentrations of the working solutions of drugs of interest into three test tubes. Each sample was prepared by adding 200 µL of standard plasma and 50 µL of the working solution of the internal standard and mixing them on a vortex for 1 min. The separation of the plasma protein precipitate required an additional 10 min of centrifugation at 4000 rpm with the addition of 2 mL of acetonitrile. Samples having the concentration range 0.3–10, 0.2–11, and 1–25 µg/mL of CIP, IBU, and DIC, respectively, were obtained by putting them in 200 µL of a mixture of acetonitrile and H2O (pH 3 with diluted acetic acid) (50:50, v/v) after evaporating the solvent in a water bath at 80 °C. Quality control samples at low, moderate, and high values were generated using the same procedure. Concentrations of CIP (1, 4, and 10 µg/mL), DIC (3, 9, and 25 µg/mL), and IBU (0.5, 2.5, and 11 µg/mL), respectively, were put in direct refrigeration at twenty degrees Celsius till testing.
Creating calibration curves and validating a procedure
Pair calibration curves were prepared, one for pure standards and the other for spiked plasma samples, Table 1, and were plotted for each drug. The prepared samples were evaluated in triplicates according to the approved method and under the working conditions of the HPLC instrument. Peak areas for both the internal standard and the analytes were noted. Regression equations were derived by graphing the resulting peak area ratio (the analyte's peak area divided by that of the internal standard's one) vs. the constant concentration. The suggested method also examined the prepared quality control tests, and concentrations were mathematically calculated using the regression equations for calibration curves of spiked plasma. The results have verified the proposed process by adhering to the rules for industry-bioanalytical method validation set forth by the US FDA18.
Greenness profile determination
Various factors must be considered when determining how environmentally friendly an analytical methodology is, including the quantity and toxicity of used chemicals, the produced waste, the used power, the count of the steps in the process, miniaturization, and automation. Two green metrics were applied, the Analytical Eco-scale and the Analytical GREEnness Calculator (AGREE). The Eco-scale score is the initially stated method19,20. The total penalty points (depending on toxicity, reagents, power, and waste) were subtracted from 100. The researcher can measure whether the method is ideally green, acceptable, or not. Another metric is AGREE. A green tool can be considered a rapid quantitative technique that provides a score indicating how closely a technique agrees with the 12 essential rules of green analytical chemistry. It is derived from readily available and accessible software21.
The pharmacokinetic study, medication administration, and extraction protocol
Ethical Approval letter number (NUB-034-023). Approval valid from 15 to 04-2023.
Samples of the target analytes were taken from the plasma of healthy, smoker-free individuals (some of the authors themselves) with an age range of 35–40 years and a weight range of 70–85 kg who had fasted for 8 h except for water without food and were measured using this method after the entire validation process was completed. The study’s potential participants have confessed their complete awareness with information about the goals and risks of the volunteering methodology before being requested to sign a written consent form which was further approved by the institutional ethics committee. ”An informed consent was obtained from all subjects/participants”.
For an oral dose of CIP (750 mg), DIC (75 mg), and IBU (600 mg), 3 groups are formed (n = 3). During the pharmacokinetic analysis, 3.5 mL of blood was drawn from the authors at times of 0.25–9 h. Ethylene diamine tetra acetic acid (EDTA) was used as an anticoagulant during the initial centrifugation of the blood samples at 4000 rpm for 10 min. The collected plasma samples were kept in a fridge until the day of the test. The suggested drugs were extracted from the samples by employing acetonitrile as a protein precipitant. A volume of 50 µL of the internal working standard's prepared solution (100 µg/mL) was added to 200 µL of each sample after placing it in a centrifuge tube. A plasma protein precipitant (2 mL) was added after a 1-min mixing on vortex. A plasma protein precipitant consisting of 0.5 mL of acetonitrile was then added. After that, the steps necessary to prepare the samples that would be utilized to generate the in-vitro calibration curves were carried out. Non-compartmental analysis (NCA) and the pharmacokinetic (PK) solver program were used to calculate the critical pharmacokinetic parameters22. The plasma concentration–time graphs were plotted, and numerous pharmacokinetic metrics were obtained afterward.
Ethics approval and consent to participate
Since the research is involving human participants and human material, it has been performed in accordance with the Declaration of Helsinki and has been approved by an appropriate ethics committee. The named volunteers, who applied for approval for the study protocol from the ethical point of view, have confessed their complete awareness with information about the goals and risks of the volunteering methodology before being requested to sign a written consent form which was further approved by the institutional ethics committee. The Committee of Nahda University, Beni-Suef, Faculty of Pharmacy has approved the study protocol from an ethical point of view. Approval No: NUB-034-023. An informed consent was obtained from all subjects/participants.
Results and discussion
Optimization of the method
A unique and fast method for the determination and separation of a mixture of three drugs viz., ciprofloxacin (CIP), Ibuprofen (IBU), and diclofenac sodium (DIC) in actual samples of human plasma was developed. Separation was achieved within only 4-min analysis time (CIP 2.75 min), (DIC 3.42) and (IBU 3.75 min) Fig. 2. This method is special and gives sharp peaks in low retention time with high selectivity and sensitivity. The method's validation agreed with the requirements of the FDA, and the results were sufficient.
Choice of the appropriate solvent to precipitate proteins
It was attempted to precipitate proteins using many solvents. Ultimately, the protein was precipitated with acetonitrile since it decreases plasma component interference. After the complete dryness of the pure supernatant, the solution was refilled using a mixture of acetonitrile and water (pH 3 with diluted acetic acid). The latter solution was chromatographed using an X-bridge UPLC RP-C18 reversed-phase stationary phase column (2.1 × 150 mm, 1.7 μm). Literature consultation23,24,25 has proved the choice of acetonitrile as a perfect solvent for the separation of CIP, DIC, and IBU.
Mobile phase mixture and flow rate optimization
Several water and acetonitrile mobile phase mixes (from 60:40 to 35:65) and varying flow rates (from 0.1 to 0.2 mL/min) were tested to separate the analyzed drugs and the components of the plasma successfully, taking into consideration the necessity of a high concentration of acetonitrile to obtain a homogeneous, sharp peak with an adequate retention time for CIP, DIC, and IBU. It was observed that low pH was required to produce a perfect peak for CIP. Several pH values between 4 and 8 were explored to improve separation, but CIP was still connected with the plasma peak in every case. For entirely separate CIP and plasma, acetonitrile: water (pH 3 with diluted acetic acid) is used, along with the suitable separation time and sharpness of the peak for DIC and IBU. After applying many separation methods, a solution of 65% acetonitrile and 35% H2O (pH 3 by diluted acetic acid) and a flow rate of 0.2 mL/min for 4 min was selected. To raise the precision and accuracy of the results, internal standards are used to calculate the concentrations of additional compounds by calculating the outcome factor; however, they must not interfere with the sample's main components. Searching for various internal standards, hydrocortisone emerged as the one that performed success through creation and improvement. Procedure optimization followed with total separation Fig. 2.
Validation of the method
The suggested technique was validated for the three drugs under test by US-FDA rules for industrial bioanalytical method validation to determine suitability, stability, LOD, LOQ, linearity, selectivity, accuracy, and precision (intraday and interday)18.
Linearity
With various samples except zero, the method's linearity was evaluated (for pure drugs and spiked plasma standards). A linear regression analysis was utilized to assess the linearity, and calibration curves were used to measure the analyte concentrations. Plots were made against the standard concentrations using the peak area ratios of each medicine to the IS, which revealed the correctness of the computed correlation coefficients' values, slopes, and intercepts. The regression coefficients of the calibration curves applied for the calculation of tested analytes were of the value 0.99 and more, as illustrated in Table 2
The limit of detection (LOD) and quantitation (LOQ)
Based on the findings for three replicates, the LOD and LOQ of CIP, DIC, and IBU were calculated. CIP, DIC, and IBU had LOD values of 0.328, 0.98, and 0.39 µg/mL, respectively, corresponding to 0.439, 2.99, and 2.92 µg/mL LOQ values.
Precision and accuracy
Furthermore, it was concluded that the lower limit of quantification is the fewest sample concentration that can be quantified by sufficient precision and accuracy (LLOQ). CIP, DIC, and IBU were all included in the LLOQ at concentrations of 3, 3, and 0.5 µg/mL, respectively. To evaluate intra-day and inter-day accuracy and precision at four different concentrations (LLOQ (low-quality control (LQC), moderate quality control (MQC), and high-quality control (HQC)), triplicates for each analyte were employed. As calculated by the coefficient of variation, precision was reported to be 0.65, 0.17, and 0.42% for CIP, DIC, and IBU for intraday precision, respectively, and 0.85, 0.35, and 0.81% for interday precision. (Table 3). Comparison of the LOD, LOQ, retention time, and precision of each of the studied drugs and other reported data7,26 was presented in Table 4.
Extraction recovery
Peak areas of the samples loaded into pure plasma at the QC levels (LQC, MQC, and HQC) were compared with peak areas of the standard analytes to determine the extraction recovery of the studied drug using the proposed methods. In Table 5, the obtained extraction recoveries (%RSD 6%) verified the extraction technique's accuracy and stability.
Selectivity
To test selectivity, the spectral data of plasma samples loaded with IS and each tested drug was compared with the ones from plasma samples of fit volunteers. It was tested using the separation among the tested drugs, internal standard, and plasma particles, as in Fig. 2.
Stability
Stability tests have been done to examine the stability of the drugs in plasma samples under a wide range of conditions that can arise during sample analysis. Three freeze cycles, as well as the bench top stability, were finished. All the results in Table 6 demonstrate the stability of the samples. Standard CIP, DIC, and IBU solutions stood stable for one week when frozen.
Greenness profile of the created method
The idea of "green analytical chemistry" (GAC) enables analytical chemists to consider safety, health, and environmental issues while doing the work. A technique is considered a superior green analysis if the Eco-scale score exceeds 75. The procedure will be more environmentally friendly as the score becomes close to 100. The new UPLC method could be seen as a perfect green method because it pointed to a score of 84. The Eco-scale score is shown in Table 7. AGREE is a green tool can be considered a rapid quantitative technique that provides a score indicating how closely a technique agrees with the 12 essential rules of green analytical chemistry. Higher scores indicate greener approaches; the total score is shown in the circle pictogram's center Fig. 3.
Results of a pharmacokinetic study
Table 8 displays the pharmacokinetic parameters determined using non-compartmental analysis and calculated after the oral administration of CIP, DIC, and IBU. It investigated how to measure the given drugs' pharmacokinetic properties individually. Also, the examined drugs' mean plasma concentration–time profiles (n = 3) are displayed in Fig. 4. CIP and DIC took 1 h, and IBU took 2 h to achieve their maximum plasma concentrations (Tmax). The maximum plasma concentrations (Cmax) for CIP27,28,29, DIC30,31, and IBU32,33 were 6.6, 5.25, and 1.37 µg/mL, respectively.
Conclusion
A quick procedure for determining and separating CIP, IBU, and DIC in real human plasma samples has been developed. Hydrocortisone was chosen as the internal standard (IS). The drugs were separated chromatographically using an Acquity UPLC ® BEH C18 1.7µm (2.1 X150 mm) column applying (UPLC) with UV detection at 210 nm. Calibration curves were discovered to be linear with acceptable correlation coefficients (0.99%). Exceptional precision and accuracy, limit of detection (LOD) and quantitation (LOQ), selectivity, and stability have been shown. Separation was achieved in only a 4-min analysis time. Two green metrics were applied, the Analytical Eco-scale and the Analytical GREEnness Calculator (AGREE). The investigation's focus was increased to investigate each of the three drugs' pharmacokinetic characteristics. FDA guidelines were applied in the method's validation, and the results were satisfactory. The new approach is more effective regarding its impact on the natural world and people's health, as well as the budget of analysis and sample preparation, according to comparisons made with the method employing the greenness metrics.
Data availability
This paper contains all data and analyses created or analyzed during the current investigation.
References
Michalska, K., Pajchel, G. & Tysk, S. Determination of ciprofloxacin and its impurities by capillary zone electrophoresis. J. Chromatogr. A. 1051, 267–272. https://doi.org/10.1016/j.chroma.2004.04.048 (2004).
Aksoy, B., Küçükgüzel, I. & Rollas, S. Development and validation of a stability-indicating HPLC method for determination of ciprofloxacin hydrochloride and its related compounds in film-coated tablets. Chroma 66, 57–63. https://doi.org/10.1365/s10337-007-0287-6 (2007).
Patel, K., Verriboina, S. K. & Vasantharaju, S. G. Stability indicating assay method development and validation of simultaneous estimation of chlorzoxazone, diclofenac sodium and paracetamol in bulk drug and tablet by RP-HPLC. Res. J. Pharm. Technol. 14, 5024–5028. https://doi.org/10.52711/0974-360X.2021.00876 (2021).
Okulik, N. & Jubert, A. H. Theoretical study on the structure and reactive sites of three non-steroidal anti-inflammatory drugs: Ibuprofen, naproxen, and tolmetin acids. J. Mol. Struct Theochem. 769, 135–141. https://doi.org/10.1016/j.theochem.2005.10.061 (2006).
Mohamed, M. A. Stability-indicating new RP-UPLC method for simultaneous determination of a quaternary mixture of paracetamol, pseudoephedrine, chlorpheniramine, and sodium benzoate in (Cold–Flu) syrup dosage form. J. AOAC Int. 105, 703–716. https://doi.org/10.1093/jaoacint/qsac002 (2022).
Vybiralova, Z., Nobilis, M., Zoulova, J., Květina, J. & Petr, P. High-performance liquid chromatographic determination of ciprofloxacin in plasma samples. J. Pharm. Biomed. Anal. 37, 851–858. https://doi.org/10.1016/j.jpba.2004.09.034 (2005).
Ashfaq, M. et al. Ecological risk assessment of pharmaceuticals in the receiving environment of pharmaceutical wastewater in Pakistan. Ecotoxicol. Environ. Saf. 136, 31–39. https://doi.org/10.1016/j.ecoenv.2016.10.029 (2017).
Gomez, D. G., Espinosa-Mansilla, A., de la Pena, A. M. & Canada, F. C. HPLC determination of ciprofloxacin, cloxacillin and ibuprofen drugs in human urine samples. J. Pharm. Sci. 29, 1969–1976. https://doi.org/10.1002/jssc.200600126 (2006).
Phyu, A. P. et al. Effects of diclofenac and ibuprofen on pharmacokinetics of ciprofloxacin in myanmar healthy volunteers. Myanmar Health Sci. Res. J. 27, 180–185 (2015).
Solís, R. R., Gómez-Avilés, A., Belver, C., Rodriguez, J. J. & Bedia, J. Microwave-assisted synthesis of NH2-MIL-125 (Ti) for the solar photocatalytic degradation of aqueous emerging pollutants in batch and continuous tests. J. Environ. Chem. Eng. 9, 106230. https://doi.org/10.1016/j.jece.2021.106230 (2021).
Liang, H., Kays, M. B. & Sowinski, K. M. Separation of levofloxacin, ciprofloxacin, gatifloxacin, moxifloxacin, trovafloxacin and cinoxacin by high-performance liquid chromatography: Application to levofloxacin determination in human plasma. J. Chromatogr. B. 772, 53–63. https://doi.org/10.1016/S1570-0232(02)00046-6 (2002).
da Silva, D. C. & Oliveira, C. C. Development of micellar HPLC-UV method for determination of pharmaceuticals in water samples. J. Anal. Methods Chem. 2, 1–12. https://doi.org/10.1155/2018/9143730 (2018).
Gałuszka, A., Migaszewski, Z. & Namieśnik, J. The 12 principles of green analytical chemistry and the SIGNIFICANCE mnemonic of green analytical practices. TrAC Trends Anal. Chem. 50, 78–84. https://doi.org/10.1016/j.trac.2013.04.010 (2013).
Santali, E. Y. et al. Greenness assessment of chromatographic methods used for analysis of empagliflozin: A comparative study. Separations 9, 275. https://doi.org/10.3390/separations9100275 (2022).
Gałuszka, A., Migaszewski, Z. M., Konieczka, P. & Namieśnik, J. Analytical eco-scale for assessing the greenness of analytical procedures. TrAC Trends Anal. Chem. 37, 61–72. https://doi.org/10.1016/j.trac.2012.03.013 (2012).
Płotka-Wasylka, J. A new tool for the evaluation of the analytical procedure: Green analytical procedure index. Talanta 181, 204–209. https://doi.org/10.1016/j.talanta.2018.01.013 (2018).
Pena-Pereira, F., Wojnowski, W. & Tobiszewski, M. AGREE—Analytical GREEnness metric approach and software. Anal. Chem. 92, 10076–10082. https://doi.org/10.1021/acs.analchem.0c01887 (2020).
U.S. Food and Drug Administration. Bioanalytical Method Validation Guidance for Industry. Available at: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/bioanalytical-method-validation-guidance-industry. (2018)
López-Lorente, Á. I. et al. The ten principles of green sample preparation. TrAC Trends Anal. Chem. https://doi.org/10.1016/j.trac.2012.03.013 (2022).
Mohamed, D. & Fouad, M. M. Application of NEMI, Analytical Eco-Scale and GAPI tools for greenness assessment of three developed chromatographic methods for quantification of sulfadiazine and trimethoprim in bovine meat and chicken muscles: Comparison to greenness profile of reported HPLC methods. Microchem. J. 157, 104873. https://doi.org/10.1016/j.microc.2020.104873 (2020).
Sajid, M. & Płotka-Wasylka, J. Green analytical chemistry metrics: A review. Talanta 238, 123046. https://doi.org/10.1016/j.talanta.2021.123046 (2022).
Zhang, Y., Huo, M., Zhou, J. & Xie, S. PKSolver: An add-in program for pharmacokinetic and pharmacodynamic data analysis in Microsoft Excel. Comput. Methods Programs Biomed. 99, 306–314. https://doi.org/10.1016/j.cmpb.2010.01.007 (2010).
Encarnação, T. et al. Development and validation of a RP-HPLC method for the simultaneous analysis of paracetamol, ibuprofen, olanzapine, and simvastatin during microalgae bioremediation. Methods X 7, 101083. https://doi.org/10.1016/j.mex.2020.101083 (2020).
Tantawy, M. A., Wahba, I. A., Saad, S. S. & Ramadan, N. K. Two validated chromatographic methods for determination of ciprofloxacin HCl, one of its specified impurities and fluocinolone acetonide in newly approved otic solution. J. Chromatogr. Sci. 60, 655–662. https://doi.org/10.1093/chromsci/bmab110 (2022).
Alquadeib, B. T. Development and validation of a new HPLC analytical method for the determination of diclofenac in tablets. Saudi Pharm. J. 27, 66–70. https://doi.org/10.1016/j.jsps.2018.07.020 (2019).
da Silva, D. C. & Oliveira, C. C. Development of micellar HPLC-UV method for determination of pharmaceuticals in water samples. J. Anal. Methods Chem. 2, 9143730–9143741. https://doi.org/10.1155/2018/9143730.(2018) (2018).
Szałek, E., Kamińska, A., Gozdzik-Spychalska, J., Grześkowiak, E. & Batura-Gabryel, H. The PK/PD index (CMAX/MIC) for ciprofloxacin in patients with cystic fibrosis. Acta Pol Pharm. 68(5), 777–783 (2011).
Khan, M. K., Khan, M. F., Khan, H. & Mustafa, G. Bioavailability of ciprofloxacin tablets in humans and its correlation with the dissolution rates. Pak. J. Pharm. Sci. 22(3), 329–334 (2009).
Navarro, M. D. S., Marinero, M. L. S. & Navarro, A. S. Pharmacokinetic/pharmacodynamic modelling of ciprofloxacin 250 mg/12 h versus 500 mg/24 h for urinary infections. J. Antimicrob. Chemother. 50, 67–72. https://doi.org/10.1093/jac/dkf079 (2002).
Zhai, X. J., Yu, Y., Chen, F. & Lu, Y. N. Comparative bioavailability and tolerability of single and multiple doses of 2 diclofenac sodium sustained-release tablet formulations in fasting, healthy chinese male volunteers. Curr. Ther. Res. Clin. Exp. 75, 53–58. https://doi.org/10.1016/j.curtheres.2013.09.001 (2013).
Kasperek, R., Zimmer, Ł, Jawień, W. & Poleszak, E. Pharmacokineticsof Diclofenac sodium and Papaverine hydrochloride after oral administration of tablets to rabbits. Acta Pol. Pharm. 72(3), 527–538 (2015).
Dewland, P. M., Reader, S. & Berry, P. Bioavailability of ibuprofen following oral administration of standard ibuprofen, sodium ibuprofen or ibuprofen acid incorporating poloxamer in healthy volunteers. BMC Clin. Pharmacol. 9, 19–28. https://doi.org/10.1186/1472-6904-9-19 (2009).
Weiser, T., Schepers, C., Muck, T. & Lange, R. Pharmacokinetic properties of ibuprofen (IBU) from the fixed-dose combination IBU/caffeine (400/100 mg; FDC) in comparison with 400 mg IBU as acid or lysinate under fasted and fed conditions—Data from 2 single-center, single-dose, randomized crossover studies in healthy volunteers. Clin. Pharmacol. Drug Dev. 8(6), 742–753. https://doi.org/10.1002/cpdd.672 (2019).
Acknowledgements
The authors thank with gratitude the native pharmaceutical companies for their kind providing for pure samples of the drugs DIC, IBU, CIB, and hydrocortisone. Also, they acknowledge the laboratory possibilities provided by Analytical Chemistry Department, Central research laboratory, Nahda University, Beni-Suef, Egypt, throughout the performance of this work.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
A.A.A.-A.: preparing, investigating, outlining, drafting, and revising. M.A.M.: development of a methodology, and writing. R.M. and A.A.F.: rewriting, revising and discussion. M.E.M.H.: supervising, investigating, editing, writing, and conceptualization.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ahmed-Anwar, A.A., Mohamed, M.A., Farghali, A.A. et al. Green UPLC method for estimation of ciprofloxacin, diclofenac sodium, and ibuprofen with application to pharmacokinetic study of human samples. Sci Rep 13, 17613 (2023). https://doi.org/10.1038/s41598-023-44846-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-44846-5
- Springer Nature Limited