Abstract
Seed dormancy is one of the most important adaptive mechanisms in plants, optimizing germination, seedling emergence, and establishment to ensure these processes occur when environmental conditions are favorable for plant survival and growth. Endemic to rocky environments of the southern Atacama Desert, the Austral papaya (Carica chilensis) is the papaya species with the southernmost distribution within the Caricaceae, thriving in the most extreme environmental conditions. This threatened plant exhibits low natural regeneration, primarily attributed to low germination, yet no information regarding seed dormancy release is available. In this study, we investigated the dormancy-break and germination requirements of C. chilensis. We hypothesized that if C. chilensis seeds exhibit physiological dormancy, then seeds with reduced moisture content and those treated with chemicals or growth hormones would exhibit higher germination percentages and faster germination than control seeds akin to other members of Caricacea. Our results confirmed this prediction and revealed that ultra-drying (< 3% moisture content) and treating seeds with sulfuric acid, gibberellic acid, or potassium nitrate are the most effective methods for germinating C. chilensis. Consequently, we suggest using these treatments to propagate this threatened papaya species.
Similar content being viewed by others
Introduction
Plants rely on seeds for reproduction, genetic continuity, and colonization1. However, the successful establishment of seedlings depends on the timing of seed germination; consequently, seeds must possess strategies to perceive their surrounding environment and trigger germination2. Seed dormancy is a plant strategy that optimizes germination, seedling emergence, and establishment so that these processes occur when environmental conditions are favorable for plant survival and growth2,3,4. It refers to inhibited germination in mature seeds even under favorable environmental conditions for germination5. Seed dormancy can be particularly important for plants in harsh and highly unpredictable environments (e.g., in arid ecosystems), where windows for recruitment are few and sparse; hence, it is more prevalent in these environments than in those with more benign conditions6.
Seed dormancy is classified into five general classes5: (1) morphological dormancy (MD), where a seed is immature when the fruit falls and requires a period of growth and embryo differentiation for germination to occur; (2) physical dormancy (PY), where the seed coat is impermeable and prevents water from entering the seed, thus requiring mechanical or chemical scarification for germination to occur; (3) physiological dormancy (PD), where a physiological inhibiting mechanism in the embryo results in low growth potential, which prevents the emergence of the radicle through covering layers, hence seeds require a specific set of conditions (i.e., often a combination of temperature, moisture, and light) to initiate germination; (4) morphophysiological dormancy (MPD), in which an underdeveloped embryo has physiological dormancy, and (5) combinational dormancy, where multiple mechanisms, including physiological, morphological and ecological factors, as well as environmental triggers prevent germination. Most families of plants have seeds with PD3, and depending on the strength of the physiological inhibitory mechanism, they can exhibit one of three levels of PD: (1) non-deep (dormancy can be broken with chemicals, gibberellic acid (GA3), warm or cold stratification, after-ripening in dry storage, and mechanical or chemical scarification), (2) intermediate (dormancy can be broken after a long period of cold stratification, and GA3 may or may not break dormancy), or (3) deep (dormancy can be broken after a long period of cold or warm stratification, and GA3 does not break dormancy)2,3,7,8,9.
Carica chilensis (Planch. ex A.DC.) Solms., commonly known as the Austral Papaya, is an endemic papaya species of Chile distributed along the southern limit of the Atacama Desert10,11,12. This species belongs to the Caricaceae, a predominantly tropical plant family that includes the common papaya (Carica papaya). The family comprises 35 species distributed across six genera13, most with seed dormancy3. Despite the economic importance of the Caricaceae, studies regarding its germination requirements are scarce, primarily focusing on C. papaya14,15,16 or a few Vasconcella species7,8,17,18. Results from these studies suggest that seeds of this plant family exhibit PD2,3 and that low seed germination is due to inhibitory substances, mainly phenolic compounds14,19,20,21, present in both the sarcotesta (the gel-like coating around the seeds) and the sclerotesta (the tough central layer of the testa)8,9,17. However, further research is needed to ascertain the kinds of dormancy in Caricaceae, aiming to enhance our comprehension of the factors influencing germination in this family.
This study aimed to investigate the seed dormancy-break and germination requirements of the Austral papaya, the southernmost species of the Caricaceae. We hypothesized that if C. chilensis seeds exhibit PD, then seeds with reduced moisture content (MC) and those treated with chemicals or growth hormones would exhibit higher germination percentages and faster germination than control seeds akin to other members of Caricacea7. Understanding the mechanisms and factors that govern seed germination and dormancy in C. chilensis holds great importance for several reasons. Firstly, the presence of papain in its fruit makes this wild papaya a plant of interest for cultivation22. Secondly, C. chilensis is classified as Vulnerable due to its limited presence in a few fragmented, low-density populations, and to the decline of natural populations resulting from habitat destruction11,12,22. Thus, studies examining its propagation have been recommended as a conservation strategy23. Lastly, the lack of knowledge regarding germination requirements of most native non-tree species in Chile is considered one of the major bottlenecks for fulfilling the country’s restoration commitements24. Therefore, there is an urgent need for studies assessing the dormancy-break requirements of these species.
Results
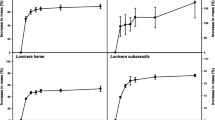
The moisture content of fresh C. chilensis seeds was 33.5% ± 0.98 (mean ± sd). Most essays successfully released seed dormancy; however, the germination percentage remained generally low, varying between 0 and 10% in the pre-sowing treatments for fresh seeds and from 0 to 40% for ultra-dry seeds (Table 1). The germination risk of ultra-dry seeds was 77% higher than that of fresh seeds (Table 2). Pre-sowing treatments significantly influenced the timing of seed germination (χ2 = 243.6, gl = 9, P < 0.0001; Fig. 1). Specifically, three treatments increased germination probability compared to the baseline germination hazard of control seeds: sulfuric acid (H2SO4) increased germination by 200%, while both gibberellic acid (GA3) and potassium nitrate (KNO3) enhanced it by 176% (Table 2).
Forest plot of the Cox proportional hazards regressions of germination clustered by Petri dish replicate. The plot shows seed germination probabilities (hazard ratio, HR) and 95% CI from seeds (N) with different moisture content and treated with different pre-sowing treatments. The HR for control and fresh seeds is standardized to 1 and denoted by the dashed vertical line. An HR > 1 indicates an increased germination probability, whereas an HR < 1 indicates a decreased probability.
The GLM analysis revealed significant interactive effects of MC and pre-sowing treatments on the mean number of germinated seeds per Petri dish (Table 3). Overall, ultra-dry seeds treated with H2SO4, GA3, or KNO3 exhibited the highest germination (Fig. 2).
Mean number of germinated seeds per Petri dish based on GLM model for the combined effects of pre-sowing treatments with moisture content (MC). We sowed each Petri dish with 10 C. chilensis seeds. Error bars indicate ± SE. Small cap letters indicate differences between the number of germinated seeds among different treatment combinations.
Discussion
Seed dormancy is an adaptive trait that allows plants to delay germination until environmental conditions become favorable for seedling establishment and growth1,25; in arid ecosystems, such conditions are generally associated with rainfall events, which are highly variable and largely unpredictable26,27. In the plant family Caricaceae, seed dormancy -particularly in the form of PD- is a common characteristic2,3. Our findings reveal that over 95% of C. chilensis seeds are dormant at maturity and that this dormancy can be partially alleviated by reducing seed MC and treating the seeds with H2SO4, GA3, or KNO3. These results suggest that, as predicted, seeds of the Austral papaya have non-deep PD.
Germination was 77% higher in ultra-dry than in fresh seeds. Similar to our results, ultra-drying promoted germination in Vasconcellea quercifolia7. Reducing seed MC can promote germination via several mechanisms. For example, it can alter seed water potential28. In this regard, seeds with lower MC have more negative matric water potentials than seeds with higher MC; as a result, they absorb water more quickly29. In contrast, solution uptake by fresh or fully imbibed seeds is driven by differences in osmotic potential between the seed and the surrounding solution30. Decreasing MC can also alter seeds’ hormonal balance, reducing abscisic acid (ABA) levels and increasing gibberellins (GA)31,32,33. These two hormones regulate seed germination in opposite manners: ABA promotes seed dormancy and inhibition of germination, whereas GA promotes germination34.
A reduction in seed MC is likely to occur naturally in C. chilensis because the species produces mature fruits between August and October, prior to the onset of summer. Since both temperature and the gradual loss of MC are integrated over time to alter the depth of the dormancy2, the hot and dry conditions of the Mediterranean summer in central Chile likely contribute to the gradual release of dormancy throughout the season. Consequently, seeds can germinate in the field at the onset of the winter rain pulses35 and take advantage of the moist soils and warmer temperatures during the spring to promote seedling establishment. Nonetheless, given that germination patterns and seed dormancy can vary geographically36 and that we collected seeds from a population located towards the southern end of C. chilensis’ distribution, which has drier and warmer summers, it will be informative to confirm if seeds from other localities show similar behavior.
Dormancy was released by treating C. chilensis seeds with H2SO4, GA3, or KNO3. The seed coat of Caricaceae is multiplicative, consisting of a fleshy outer layer known as the sarcotesta, enclosing a hard, lignified portion of the testa37. Hence, scarification with H2SO4 can enhance water absorption and uptake, essential for initiating germination, by breaking down the seed coat. Gibberellins are a family of plant hormones that control many aspects of plant growth, including germination2. GA3 stimulates germination through several mechanisms, including producing hydrolytic enzymes that weaken the seed coat38, mobilization of nutrient reserves, and stimulation of plant embryo expansion and hypocotyl elongation33,39. Numerous studies have documented enhanced germination in C. papaya with the application of GA340. Similarly, GA3 promotes germination in a few species of Vasconcellea7,8. However, apart from these findings, there is currently a lack of information regarding the effects of GA3 in other Caricaceae species. Finally, KNO3 can break seed dormancy and promote germination in at least two ways. First, it has hygroscopic properties; when seeds are imbibed in a KNO3 solution, they absorb moisture from the surrounding environment, weakening the seed coat. Second, it inhibits ABA synthesis while stimulating the synthesis of GA, which promotes germination by enhancing the growth potential of the embryo and overcoming the mechanical barriers imposed by the testa2.
One of the major impediments to the potential use of wild species germplasm for habitat restoration is the need for more knowledge about techniques for breaking seed dormancy and caring for germinating seeds41. Thus, improving our understanding of seed biology is crucial to restoring a broader and more representative range of species42. Chile is one of 115 countries that has subscribed to restoration commitments43. However, dormancy-break and germination requirements of seeds of most non-tree species in Chile remain poorly known. This lack of knowledge hinders the propagation of threatened plants and is recognized as a bottleneck for fulfilling Chile’s restoration commitments33. This study contributes to filling this gap with the aim of promoting more extensive use of species diversity in ecological restoration and helping to conserve endangered plant species.
Methods
Study species
Carica chilensis is an endemic shrub of northern Chile distributed along the southern limit of the Atacama Desert, between the Atacama (28° 39′ S; 71° 42′ W) and Valparaiso (33° 09′ S; 71° 42′ W) Regions. It is classified as threatened, and due to its slow growth and low germination percentages, C. chilensis experiences minimal natural regeneration. Like most Caricaeae13, this species is polygamous with male, female, and hermaphrodite plants. It produces mature fruits from July to October, with peak fruiting in September. Each fruit contains an average of six seeds enveloped by a gelatinous layer. As the southernmost species in the family, C. chilensis is subject to extreme environmental conditions, such as drought and saline soils, across its distribution range.
Fruit collection site and procedure
We collected mature C. chilensis fruits from 40 individuals in the Pupío basin in North Central Chile (32° 07 S, 71° 26 W, 190 masl) between July and September 2021. The area has a Mediterranean climate with warm, dry summers and cool, wet winters44. The mean annual temperature is ~ 22 °C; July is the coldest month (~ 9–8 °C, mean coldest temperature), and February is the warmest (~ 22.7 °C, mean warmest temperature). Mean annual precipitation is 227 mm, 86% of which falls from May to August (data from Los Vilos Weather Station, Dirección General de Aguas, 1982–2019).
We collected between five and eight fruits per plant and placed them in paper bags in a cooler until they were processed a maximum of two days later in the laboratory at Universidad de La Serena. We manually removed the seeds from the fruits and cleaned them with a toothbrush under running water to remove the sarcotesta. Then, we pooled the seeds together for the experiments.
Dormancy-break requirements
We determined the dormancy-break requirements of C. chilensis seeds by examining the combined effects of MC and pre-sowing treatments. To establish the MC treatment, we first assessed initial seed MC by weighing 100 seeds before and after drying at 130 °C for three h45. Seeds for the ultra-dry treatment were then placed in Eppendorf tubes with blue silica gel and dried to < 3% of their initial MC. We assessed germination of fresh and ultra-dry seeds in each of nine pre-sowing treatments: (1) Control (no pre-sowing treatment); (2) seeds soaked in a 400 ppm solution of gibberellic acid (GA3) for 24 h at room temperature; (3) seeds soaked in a 1 M solution of potassium nitrate (KNO3) for 30 min; (4) seeds soaked in distilled water changed daily for seven days at room temperature (hydro conditioning); (5) seeds soaked first in distilled water for 24 h and then in hot water (35 °C) for four h (Temperature shock); (6) seeds shaken in a glass jar for 10 min (percussion), (7) seeds cold (moist) stratified, kept at 5 °C for 7 days; (8) seeds soaked in a 100% solution of hydrogen peroxide (H2O2) for 20 min and; (9) seeds soaked in a 10% solution of sulfuric acid (H2SO4) for 30 min.
We replicated each treatment combination in 10 Petri dishes and sowed each dish with ten seeds in sterilized sand (60 g), which was hydrated to carrying capacity (ca. 12 ml of water). Petri dishes were incubated in germination chambers with 16 h of light at 25 °C7, and we monitored germination periodically for 94 days. We considered a seed germinated when the radicle emerged (> 2 mm). At the end of the experiment, we calculated the number of germinated seeds, and the seed germination percentage and rate for each Petri dish.
Statistical analyses
Initially, we evaluated the independent effects of MC and pre-sowing treatments on seed germination using a Cox Proportional hazards model (‘survival’ package46) nested within each Petri dish to consider the non-independence of seeds. In these models, the dependent variable is a hazard function, which represents how the instantaneous “risk” of experiencing the event (i.e., germination) changes with time. This risk function was compared to a "baseline hazard function," representing the risk of the event occurring at a specific time in a reference individual or group47. To establish a baseline, we utilized the germination rates of fresh and control seeds as the baseline hazard because this is the natural state of the seeds. By comparing the hazard ratios, we could identify the conditions promoting the fastest germination. Subsequently, we investigated how the combined effects of MC and pre-sowing treatments affect the overall number of seeds germinating using a Generalized Linear Model. In this model, the independent variables were MC and pre-sowing treatment, and the dependent variable was the number of germinated seeds per Petri dish (GLM, family: Poisson, link: log). We performed all statistical analyses using the R statistical environment48.
Relevant legislations, permitting and consent
Plant material collection was carried out per relevant institutional, national, and international guidelines and legislation, including the IUCN Policy Statement on Research Involving Species at Risk of Extinction and the Convention on the Trade in Endangered Species of Wild Fauna and Flora. Seeds were obtained from plants on private lands, and we obtained permission from the landowners to access the areas and collect the seeds. Given that C. chilensis is Chile’s sole wild Caricacea species in Chile and easily identifiable in the field, there was no need to collect voucher specimens.
Data availability
The datasets used in the current study are available in the Excel file included in the supplementary material section.
References
Bewley, J. D., Bradford, K. & Hilhorst, H. Seeds: Physiology of Development, Germination and Dormancy (Springer Science & Business Media, 2012).
Finch-Savage, W. E. & Leubner-Metzger, G. Seed dormancy and the control of germination. New Phytol. 171, 501–523 (2006).
Baskin, C. C. & Baskin, J. M. Seeds: Ecology, Biogeography, and Evolution of Dormancy and Germination 2nd edn. (Elsevier, 2014).
Benech-Arnold, R. L., Sánchez, R. A., Forcella, F., Kruk, B. C. & Ghersa, C. M. Environmental control of dormancy in weed seed banks in soil. Field Crop Res. 67, 105–122 (2000).
Baskin, J. M. & Baskin, C. C. A classification system for seed dormancy. Seed Sci. Res. 14, 1–16 (2004).
Jurado, E. & Flores, J. Is seed dormancy under environmental control or bound to plant traits?. J. Veg. Sci. 16, 559–564 (2005).
Urtasun, M. M. et al. Dormancy release, germination and ex situ conservation of the southern highland papaya (Vasconcellea quercifolia, Caricaceae), a wild crop relative. Sci. Hortic. 263, 109134 (2020).
Benítez, S. P., Mario, L., Delgado, O. A. & Medina, C. I. Estudios de germinación y remoción de latencia en semillas de papayuelas Vasconcellea cundinamarcensis y Vasconcellea goudotiana. Cienc. y Tecnol. Agropecu. 14, 187–197 (2013).
Freitas, S. D. J. et al. Métodos de remoção da sarcotesta na germinação de sementes de jaracatiá. Rev. Árvore 35, 91–96 (2011).
Rodríguez Ríos, R. & Marticorena Garri, A. Catálogo de las plantas vasculares de Chile. Gayana. Bot. 75, 1–430 (2018).
MMA. Carica chilensis. (Ministerio de Medio Ambiente, Chile, 2005). https://clasificacionespecies.mma.gob.cl/wp-content/uploads/2019/10/Carica_chilensis_FINAL.pdf
Squeo, F. A., Arancio, G. & Gutierrez, J. R. Libro Rojo de la Flora Nativa y de los Sitios Prioritarios para su Conservación: Región de Coquimbo. (Ediciones Universidad de La Serena, 2001).
Carvalho, F. A. & Renner, S. S. in Genetics and genomics of papaya. 81–92 (Springer, 2013).
Salomão, A. N. & Mundim, R. C. Germination of papaya seed in response to desiccation, exposure to subzero temperatures, and gibberellic acid. Hortic. Sci. 35, 904–906 (2000).
Ellis, R. H., Hong, T. D. & Roberts, E. H. Effect of storage temperature and moisture on the germination of papaya seeds. Seed Sci. Res. 1, 69–72 (1991).
Shankaralingappa, Y. H., Vasugi, C. & Naik, L. B. Ultra dry seed storage-a cost effective method for conservation of germplasm and commercial seeds of papaya. Indian J. Hortic. 76, 423–429 (2019).
Urtasun, M. M., Giamminola, E. M. & Viana, M. L. D. Southern highland papaya (Vasconcellea quercifolia A. St.-Hil.): Do fruit ripening and harvesting time affect seed germination? Acta Scientiarum. Agronomy 42 (2019).
Vélez-Mora, D. P., González, R. A. & Zimmermann, M. J. Mejoramiento de la germinación, control de la hiperhidricidad y formación de brotes en Vasconcellea stipulata Badillo. Rev. Colomb. de Biotecnol. 17, 16–21 (2015).
Chow, Y. J. & Lin, C. H. p-Hydroxybenzoic acid as the major phenolic germination inhibitor of papaya seed. Seed Sci. Technol. 19, 167–174 (1991).
Wood, C. B., Pritchard, H. W. & Amritphale, D. Desiccation-induced dormancy in papaya (Carica papaya L.) seeds is alleviated by heat shock. Seed Sci. Res. 10, 135–145 (2000).
Kari, Z. A. Effect of strorage temperature and seed mouisture contents on papaya (Carica papaya L.) seed viability and germination. J. Sustain. Sci. Manag. 8, 87–92 (2013).
Carrasco, B., García-Gonzáles, R., Díaz, C., Avila, P., Cáceres, P., Lobos, G.A., Silva, H. & Caligari, P.D. Genetic and morphological characterization of the endangered Austral papaya Vasconcellea chilensis (Planch. ex A. DC.) Solms (2014).
Hechenleitner, V. P., Gardner, F. M., Thomas, P. I., Echeverría, C., Escobar, B., Brownless, P., & Martínez A. C. Plantas amenazadas del Centro-Sur de Chile. Distribución, conservación y propagación. Universidad Austral de Chile, Real Jardín Botánico de Edimburgo, Valdivia, Chile, p. 188 (2005).
León-Lobos, P. et al. Lack of adequate seed supply is a major bottleneck for effective ecosystem restoration in Chile: Friendly amendment to Bannister et al. (2018). Restor. Ecol. 28, 277–281 (2020).
Foley, M. E. Seed dormancy: An update on terminology, physiological genetics, and quantitative trait loci regulating germinability. Weed Sci. 49, 305–317 (2001).
Schwinning, S. & Sala, O. E. Hierarchy of responses to resource pulses in arid and semi-arid ecosystems. Oecologia 141, 211–220 (2004).
Noy-Meir, I. Desert ecosystems: Environment and producers. Annu. Rev. Ecol. Syst. 4, 25–51 (1973).
Kigel, J. Seed Development and Germination Vol. 41 (CRC Press, 1995).
Wolk, W. D., Dillon, P. F., Copeland, L. F. & Dilley, D. R. Dynamics of imbibition in Phaseolus vulgaris L. in relation to initial seed moisture content. Plant Physiol. 89, 805–810 (1989).
Roberts, E. & Ellis, R. Water and seed survival. Ann. Bot. 63, 39–39 (1989).
Skubacz, A. & Daszkowska-Golec, A. Seed dormancy: The complex process regulated by abscisic acid, gibberellins, and other phytohormones that makes seed germination work. Phytohormones: Signaling Mechanisms and Crosstalk in Plant Development and Stress Responses. Rijeka Croatia: In Tech, pp. 77–100 (2017).
Finkelstein, R., Reeves, W., Ariizumi, T. & Steber, C. Molecular aspects of seed dormancy. Annu. Rev. Plant Biol. 59, 387–415 (2008).
Kucera, B., Cohn, M. A. & Leubner-Metzger, G. Plant hormone interactions during seed dormancy release and germination. Seed Sci. Res. 15, 281–307 (2005).
Karssen, C. & Lacka, E. in Plant Growth Substances 1985: Proceedings of the 12th International Conference on Plant Growth Substances, Held at Heidelberg, August 26–31, 315–323 (Springer, 1985).
Chesson, P. et al. Resource pulses, species interactions, and diversity maintenance in arid and semi-arid environments. Oecologia 141, 236–253 (2004).
Wagmann, K. et al. Seed dormancy distribution: Explanatory ecological factors. Ann. Bot. 110, 1205–1219 (2012).
Kubitzki, K. Caricaceae. Flowering Plants· Dicotyledons: Malvales, Capparales and Non-betalain Caryophyllales, pp. 57–61 (2003).
Rodríguez, S. et al. Analysis of the effect of scarification process on papaya (Carica papaya Lin.) seeds germination. In Seed Dormancy and Germination (eds Koornneef, M. et al.) (IntechOpen, 2019).
Thomas, S. G., Rieu, I. & Steber, C. M. Gibberellin metabolism and signaling. Vitam. Horm. 72, 289–338 (2005).
Anjanawe, S., Kanpure, R., Kachouli, B. & Mandloi, D. Effect of plant growth regulators and growth media on seed germination and growth vigour of papaya. Ann. Plant Soil Res. 15, 31–34 (2013).
Hay, F. R. & Probert, R. J. Advances in seed conservation of wild plant species: a review of recent research. Conserv. physiol. 1, 030 (2013).
Broadhurst, L. M., Jones, T. A., Smith, F. S., North, T. O. M. & Guja, L. Maximizing seed resources for restoration in an uncertain future. BioScience 66, 73–79 (2016).
Sewell, A., Van Der Esch, S. & Löwenhardt, H. Goals and Commitments for the Restoration Decade (PBL Netherlands Environmental Assessment Agency, 2020).
Luebert, F. & Pliscoff, P. Sinopsis Bioclimática y Vegetacional de Chile (Editorial Universitaria, 2006).
ISTA. (International Seed Testing Association Bassersdorf, Switzerland).
Therneau, T. M. (New York, NY, USA, 2023).
Fox, J. & Weisberg, S. Cox proportional-hazards regression for survival data. An R and S-PLUS companion to applied regression (2002).
R Foundation for Statistical Computing. R: A Language and Environment for statistical Computing (R Foundation for Statistical Computing, 2022).
Acknowledgements
We would like to express our gratitude to the anonymous reviewers for their valuable feedback and insights, which significantly improved the quality of this research. Additionally, we thank Cristian Delpiano and José Reyes for assisting us in collecting seeds in the field. Likewise, we would like to acknowledge Enrique Ostria and Máximo Gonzalez for providing us with germination chambers for the experiments. This study was supported by Proyecto INCO - Investigación científica en Carica chilensis y Porlieria chilensis: GCP-1001-01-CS-0374 and Proyecto Basal FB210006 (Instituto de Ecología y Biodiversidad, IEB).
Author information
Authors and Affiliations
Contributions
A.P.L. designed experiments, analyzed the results, and wrote the first draft of the manuscript; G.C. set up the experiment and collected the data. P.G-G and D.E.C. provided a general overview, assisted with data analysis, and contributed to preparing the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Loayza, A.P., García-Guzmán, P., Carozzi-Figueroa, G. et al. Dormancy-break and germination requirements for seeds of the threatened Austral papaya (Carica chilensis). Sci Rep 13, 17358 (2023). https://doi.org/10.1038/s41598-023-44386-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-44386-y
- Springer Nature Limited